6 6 NOTES Oxidation Numbers 2 Oxidation Number

6. 6 – NOTES Oxidation Numbers

• 2. Oxidation Number: The charge (real or hypothetical) assigned to an atom. • Describes the placement of electrons across an entire compound. Where are the electrons spending most of their time? • - Charge an atom would have IF e- were transferred completely;

• Oxidation number rules: • Pure elements uncombined state (diatomics, P 4, S 8, He, etc. ) = 0 • Monatomic ions - ions composed of only 1 atom; equals the charge • Specific nonmetals • Oxygen for most compounds is -2; in H 2 O 2 and other peroxides (O 2 -2) = -1 • Hydrogen +1; unless bonded to metals in binary compounds (Li. H, Na. H, Ca. H 2) = -1 • Fluorine -1 in ALL compounds; other halogens will be (-) as halides, when combined w/ oxygen could have (+) oxidation #s • Sum of oxidation numbers in a compound = 0 • Sum of oxidation numbers in a polyatomic ion = charge found on ion

• Example #2: Determine the oxidation number of the transition metal: (Hint: when given a polyatomic ion, use the number and charge of the ion rather than individual atoms. ) • Cu(NO 2)2, Sn(SO 4)2 Pb(NO 3)2 • Cu(NO 2)2 = x + 2(-1) = 0 so x = 2 • Sn(SO 4)2 = x + 2(-2) = 0 so x = 4
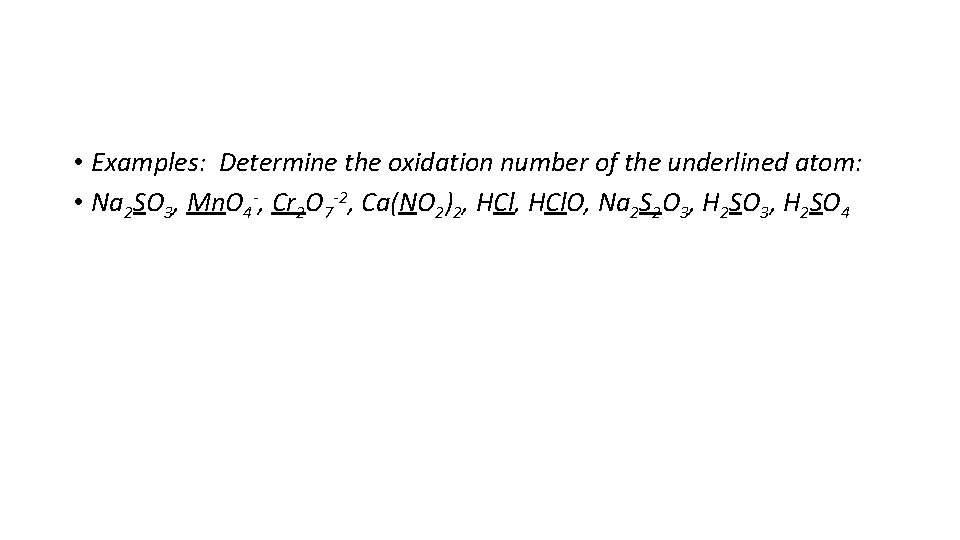
• Examples: Determine the oxidation number of the underlined atom: • Na 2 SO 3, Mn. O 4 -, Cr 2 O 7 -2, Ca(NO 2)2, HCl. O, Na 2 S 2 O 3, H 2 SO 4
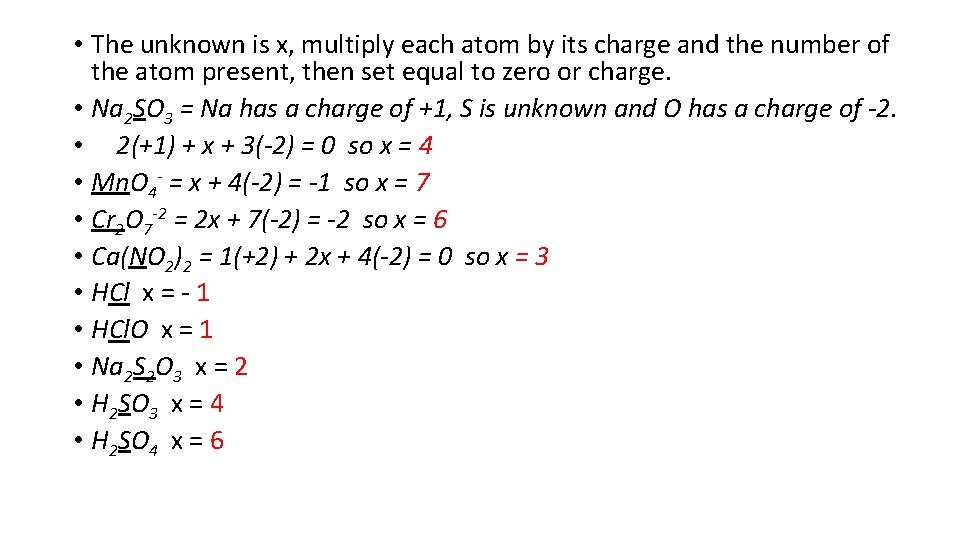
• The unknown is x, multiply each atom by its charge and the number of the atom present, then set equal to zero or charge. • Na 2 SO 3 = Na has a charge of +1, S is unknown and O has a charge of -2. • 2(+1) + x + 3(-2) = 0 so x = 4 • Mn. O 4 - = x + 4(-2) = -1 so x = 7 • Cr 2 O 7 -2 = 2 x + 7(-2) = -2 so x = 6 • Ca(NO 2)2 = 1(+2) + 2 x + 4(-2) = 0 so x = 3 • HCl x = - 1 • HCl. O x = 1 • Na 2 S 2 O 3 x = 2 • H 2 SO 3 x = 4 • H 2 SO 4 x = 6
- Slides: 6