6 1 Introduction to Bonding l Chemical Bond

6 -1 Introduction to Bonding l Chemical Bond ¡Mutual attraction between the nuclei and valence electrons of different atoms that binds them together. l Types of Bonds ¡Ionic Bonds l. Force of attraction between oppositely charged ions. ¡Covalent Bond l. Force of attraction for electrons, that results in a pair of electrons being shared by two atoms.
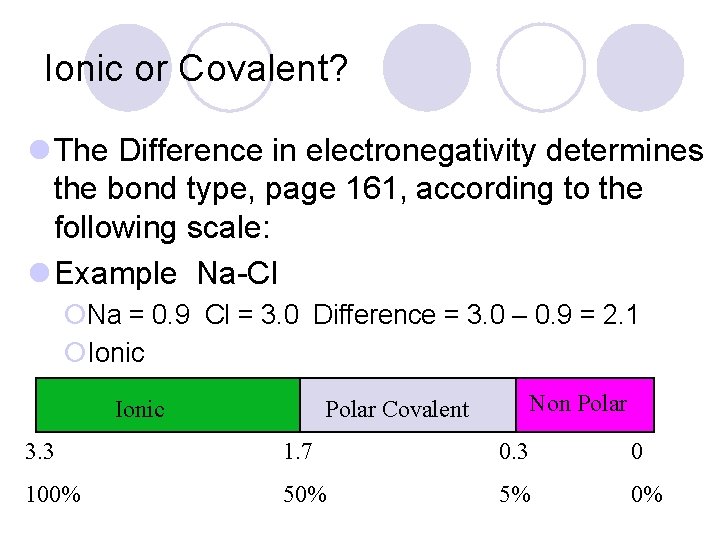
Ionic or Covalent? l The Difference in electronegativity determines the bond type, page 161, according to the following scale: l Example Na-Cl ¡Na = 0. 9 Cl = 3. 0 Difference = 3. 0 – 0. 9 = 2. 1 ¡Ionic Non Polar Covalent 3. 3 1. 7 0. 3 0 100% 5% 0%

Polar Vs. Nonpolar Covalent Bonds Non-Polar – electrons are equally shared Polar – electrons are not equally shared ¡ Balanced Distribution of Electrical Charge ¡ Unbalanced Distribution of Electrical Charge + -

Table of Electronegativities

Identifying Bond Types l Determine if the following are going to form; Ionic, Polar Covalent or Non-polar Covalent Bonds: C-N Ca-F Br-Br H-Br
Summary of Bond Types
Chapter 6. 2 - Covalent Bonding l Molecule (Molecular Substance) ¡Group of atoms that are held together by a covalent bond. l Molecular Formula ¡Describes the composition of atoms in a single molecule of a compound. ¡Number and type of each atom in the molecule. l Diatomic molecule ¡A molecule containing only 2 atoms. ¡Seven Diatomic Elements ¡H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2
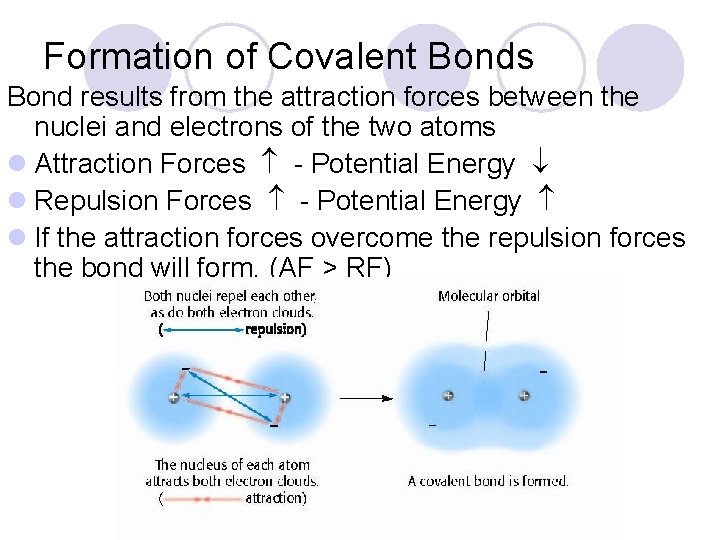
Formation of Covalent Bonds Bond results from the attraction forces between the nuclei and electrons of the two atoms l Attraction Forces - Potential Energy l Repulsion Forces - Potential Energy l If the attraction forces overcome the repulsion forces the bond will form. (AF > RF)
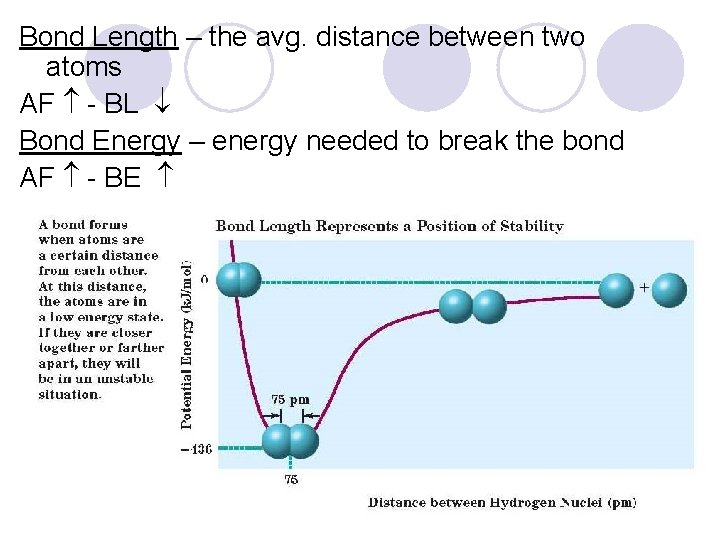
Bond Length – the avg. distance between two atoms AF - BL Bond Energy – energy needed to break the bond AF - BE
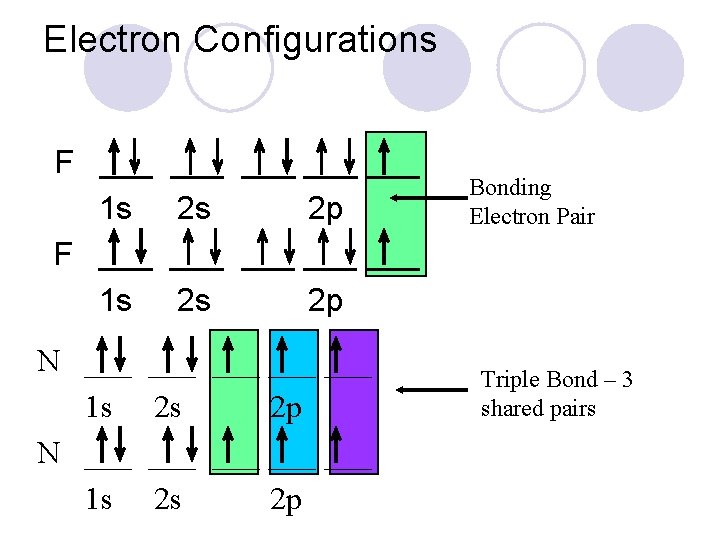
Electron Configurations F ___ 1 s N ___ 1 s ___ ___ ___ ___ 2 s 2 p Bonding Electron Pair Triple Bond – 3 shared pairs

Lewis structures l Formula that specifies which atoms are bonded to each other in a molecule. l Places atoms in a pattern in which they can share electrons to satisfy the octet rule. l Every shared pair of electrons are depicted as a dash ( — ). ¡Shared Pair, are 2 electrons that are shared between 2 atoms. (bonding pairs) l All unshared electrons must be paired. ¡Unshared Pair, are 2 electrons that are NOT shared between 2 atoms. (non-bonding pairs)

Single Covalent Bonds l Draw the Lewis Structure for the molecule that has 1 N and 3 H. ¡Identify which atom has the most free electrons. l. Usually the lowest electronegative element. ¡Place this atom in the center of the structure.

Systematic over Trial-Error l l l Begin with the lowest electronegative element in the center. Count the total number of valence electrons (N) needed to account for the atoms (based on the column of the atom in the periodic table) and charge (add one electrons for each negative charge; subtract one electron for each positive charge). Draw the framework with single bonds. Some knowledge of the way the atoms are connected may be required. Using lone pairs, complete octets around the non-central atoms. Count the number of electrons depicted (two for each bond and two for each lone pair). If this number is less than N, then add electrons to the central atom until the total number of electrons depicted is N. If the octet rule is not satisfied for the central atom and lonepair electrons are nearby, use those electrons to make double or triple bonds to the central atom.

Practice Single Covalent Bonds l Write the Lewis Structure for the following formulas: ¡Cl 2 ¡OF 2 ¡C 2 H 6 ¡C 3 H 7 Cl

Multiple Covalent Bonds l Some structures need more than a single pair of electrons to reach the octet. l Double covalent bonds ¡Covalent bond between 2 atoms in which there a total of 4 electrons being shared. (2 pair) l Triple covalent bonds ¡Covalent bond between 2 atoms in which there a total of 6 electrons being shared. (3 pair) ¡CO 2 and HCN

Polyatomic Ion Structures l Polyatomic ions, are covalently bonded atoms that form a charge due to the gain or loss of electrons. l When drawing the structures of polyatomic ions: ¡(–) ions must have extra electrons in the structure that is equivalent to its charge. ¡(+) ions must lose the number of electrons in the structure that is equivalent to its charge.

Example l NH 4 +1, the ammonium ion. l Starts out like the ammonia molecule NH 3 l But it has an extra H and a +1 charge, meaning the structure has 1 less electron.

Exceptions to the Octet l Incomplete Octet ¡Some atoms that have less than eight electrons. l. Hydrogen only needs two and Boron needs six electrons. l Expanded Octet ¡Atoms that can have more than eight electrons. l Common expanded octets: ¡Cl, Br, I, S, and P l IMPORTANT!!! ¡Only one atom may exceed the standard eight valence. ¡Must be the central atom only!!

Coordinate Covalent Bond l Like a single covalent bond, but a single atom is sharing 2 electrons with another atom, in which it doesn’t increase the # of electrons in the atom sharing the 2 electrons. l This is determined by one atom having more electrons drawn than it originally contained. l Indicated by using an arrow rather than a dashed line. ( ) The arrow must point away from the atom that has the pair of electrons.

Resonance l Bonding in molecules or ions that cannot be correctly represented by a single Lewis Structure. Can be determined by Formal Charges. l FC = V-N-B/2 l Example: Ozone or CO 2 l O = C → O or O = C = O or O←C → O
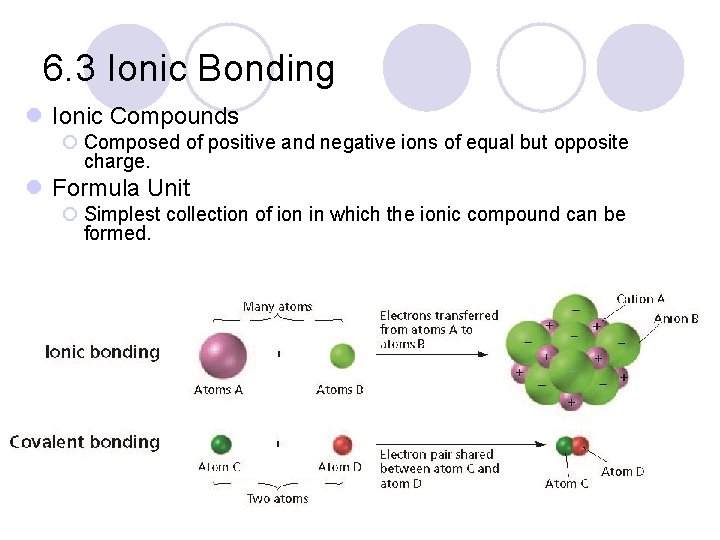
6. 3 Ionic Bonding l Ionic Compounds ¡ Composed of positive and negative ions of equal but opposite charge. l Formula Unit ¡ Simplest collection of ion in which the ionic compound can be formed.

3 D Array of forces is created (Lattice) l. Strength of Array – varies w/ sizes, charges, & number of ions. l Arrangement of ions gives the crystal its strength l To compare the strengths Ca. F 2 ¡Chemists compare energies released when the ionic compound forms. l Lattice Energy – energy released, 1 mole of an ionic compound is formed. (from gaseous ions)
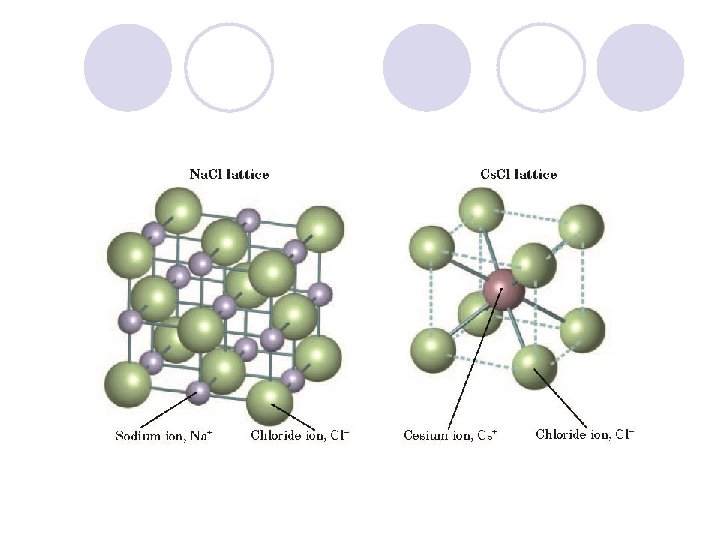
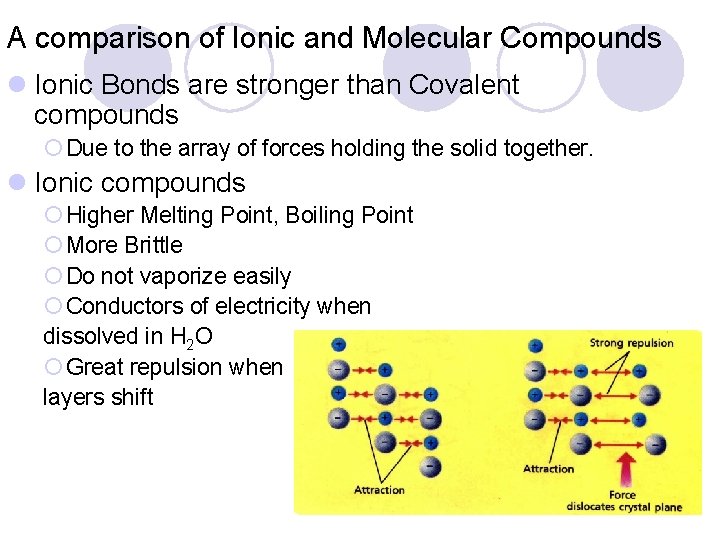
A comparison of Ionic and Molecular Compounds l Ionic Bonds are stronger than Covalent compounds ¡ Due to the array of forces holding the solid together. l Ionic compounds ¡ Higher Melting Point, Boiling Point ¡ More Brittle ¡ Do not vaporize easily ¡ Conductors of electricity when dissolved in H 2 O ¡ Great repulsion when layers shift

Lewis Dot Diagrams l Using electron dot notation, sketch the exchange of valence electrons in atoms as they form ionic bonds.

Class work l Show the electron transfer for the following elements and write the formula unit for the binary ionic compound: 1) Zn + I 2) Al + S 3) K + P 4) Mg + N 5) Ba + Se 6) Fe 2+ + O

6. 4 Metallic Bonding l Bonding resulting from the attraction of positive ions and mobile electrons l Metal Atoms ¡Small Number of Valence Electrons l 2 in the s-Block & 0 in the p-Block ¡Low Ionization Energy & Electronegativity l. Easily give up electrons l. At best – weakly covalent ¡Electrons are Delocalized l. Electrons do not belong to any one ion

Metallic Properties Electron-Sea Model Explains Properties of Metals l Lustrous l Good Conductors of Heat & Electricity l Malleability ¡Bent or Shaped w/ Hammer l Ductility ¡Able to be drawn into a wire

6. 5 Molecular Geometry l Properties of Molecular Compounds depend upon 2 main things ¡Bonding ¡Molecular Geometry – arrangement of atoms in space l. Used to determine Molecular Polarity l VSEPR Theory is used to predict molecular Geometry
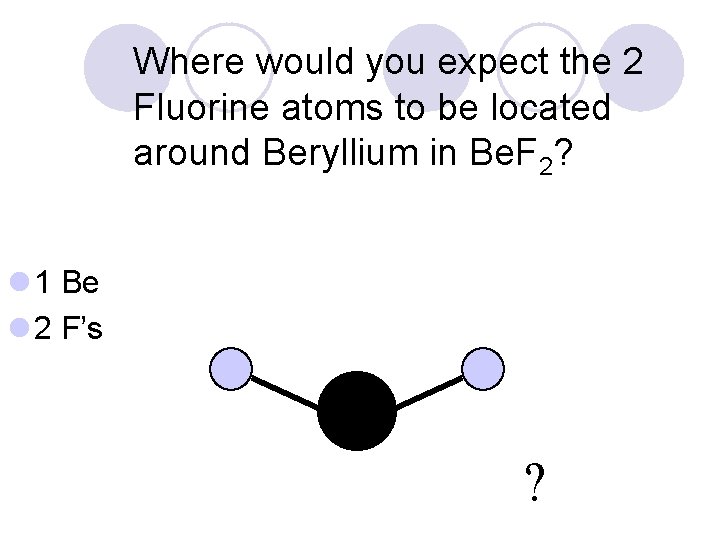
Where would you expect the 2 Fluorine atoms to be located around Beryllium in Be. F 2? l 1 Be l 2 F’s ?
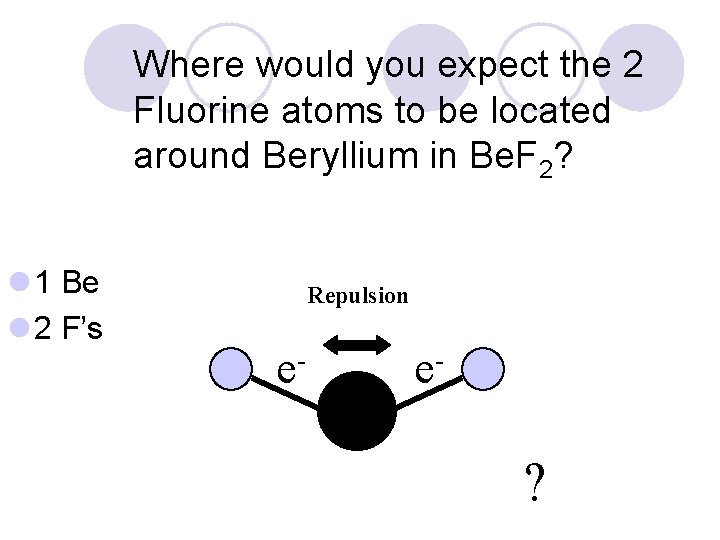
Where would you expect the 2 Fluorine atoms to be located around Beryllium in Be. F 2? l 1 Be l 2 F’s Repulsion e- e- ?
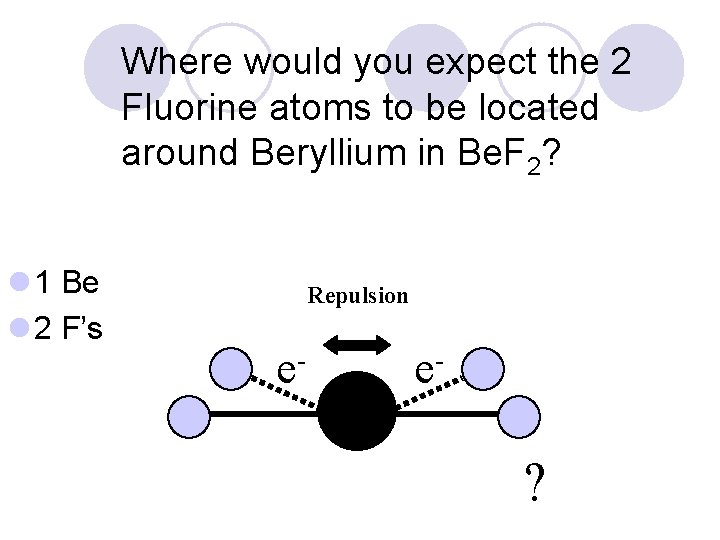
Where would you expect the 2 Fluorine atoms to be located around Beryllium in Be. F 2? l 1 Be l 2 F’s Repulsion e- e- ?
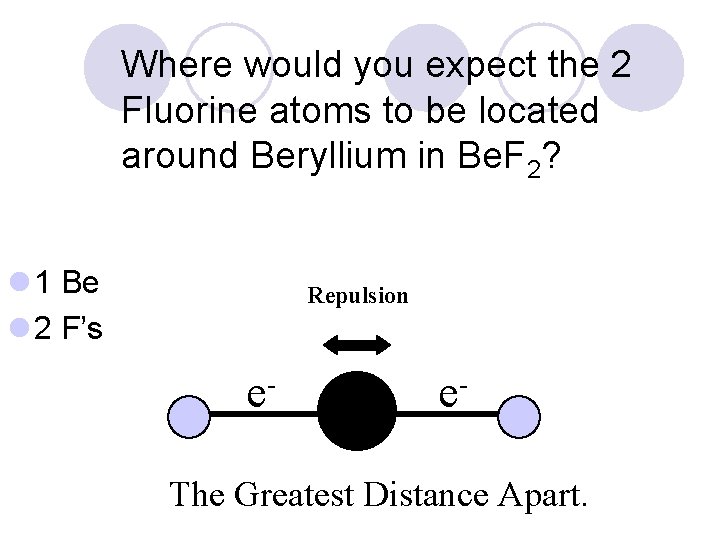
Where would you expect the 2 Fluorine atoms to be located around Beryllium in Be. F 2? l 1 Be l 2 F’s Repulsion e- e- The Greatest Distance Apart.
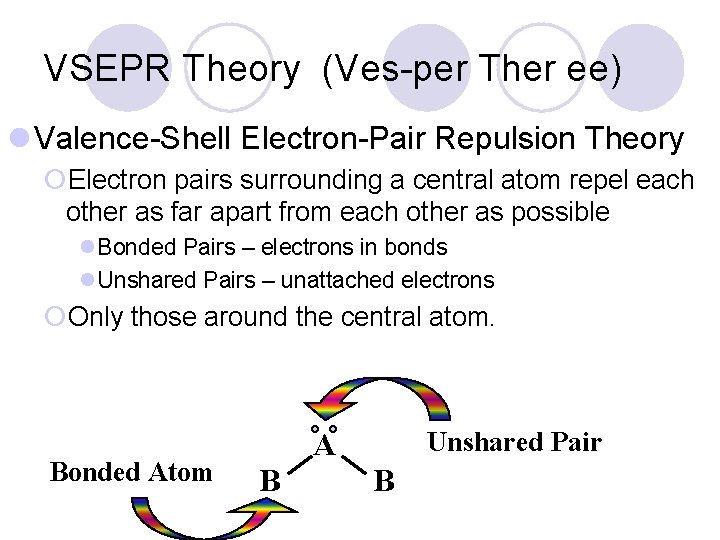
VSEPR Theory (Ves-per Ther ee) l Valence-Shell Electron-Pair Repulsion Theory ¡Electron pairs surrounding a central atom repel each other as far apart from each other as possible l. Bonded Pairs – electrons in bonds l. Unshared Pairs – unattached electrons ¡Only those around the central atom. Bonded Atom Unshared Pair A B B
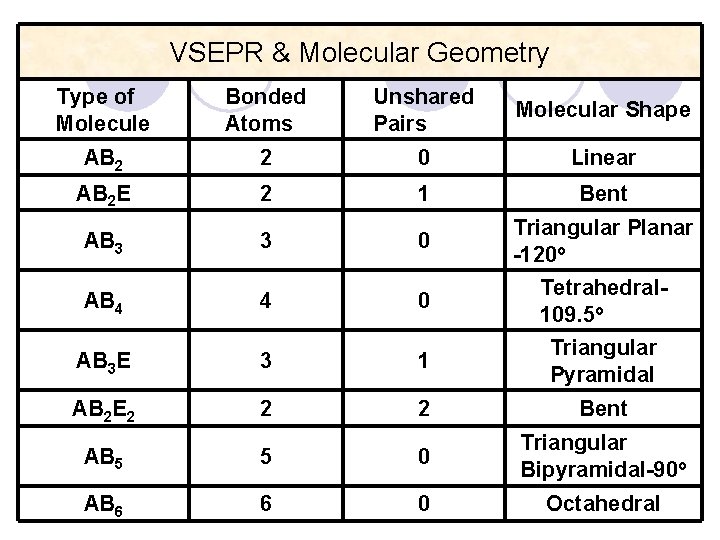
VSEPR & Molecular Geometry Type of Molecule Bonded Atoms Unshared Pairs Molecular Shape AB 2 2 0 Linear AB 2 E 2 1 Bent 0 Triangular Planar -120 o 0 Tetrahedral 109. 5 o AB 3 AB 4 3 4 AB 3 E 3 1 Triangular Pyramidal AB 2 E 2 2 2 Bent AB 5 5 0 Triangular Bipyramidal-90 o AB 6 6 0 Octahedral
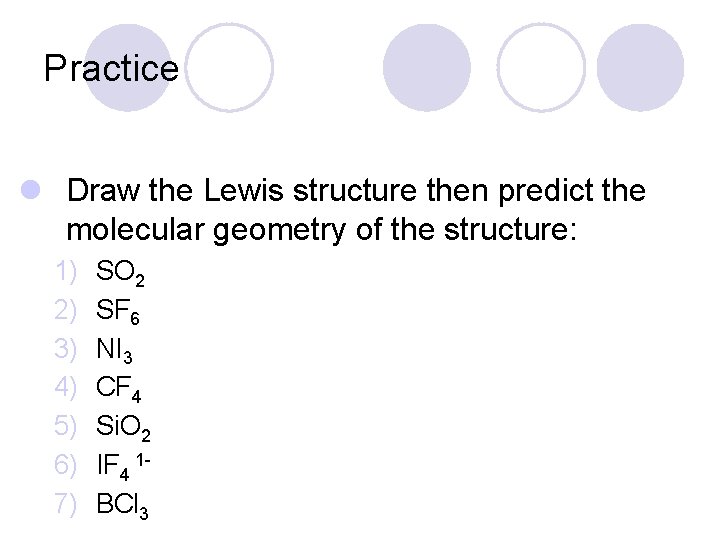
Practice l Draw the Lewis structure then predict the molecular geometry of the structure: 1) 2) 3) 4) 5) 6) 7) SO 2 SF 6 NI 3 CF 4 Si. O 2 IF 4 1 BCl 3
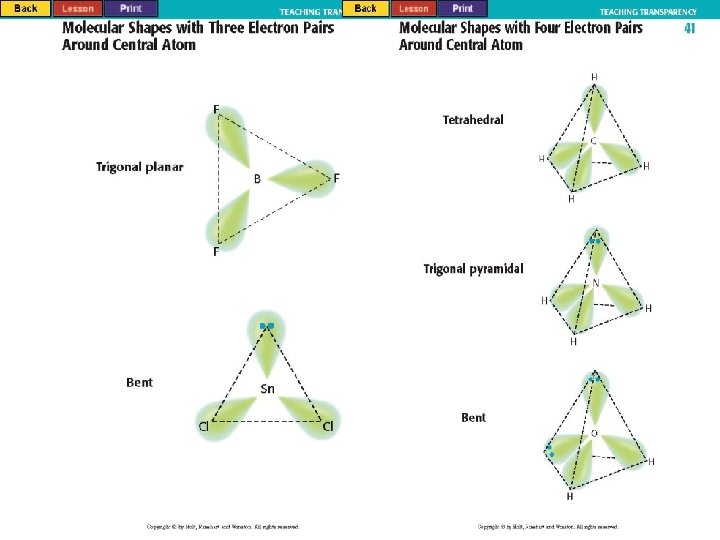
Molecular Polarity l Polar Bond – uneven sharing of electrons C C + O - l Oxygen has a greater electronegativity than carbon
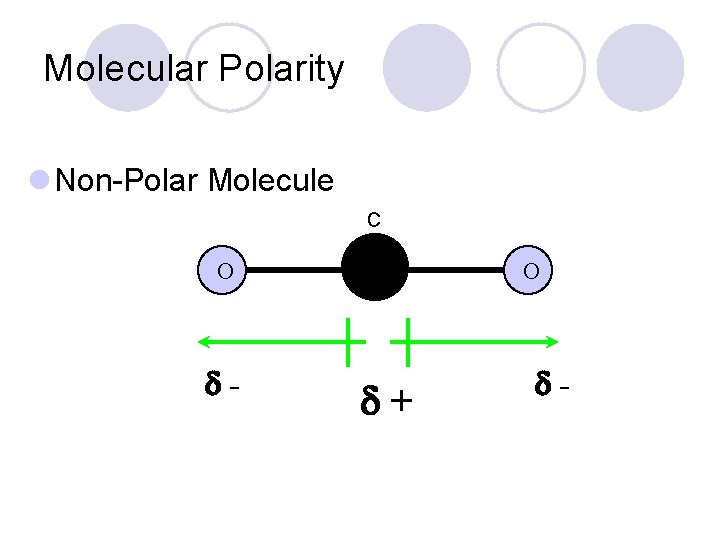
Molecular Polarity l Non-Polar Molecule C O - C + O -
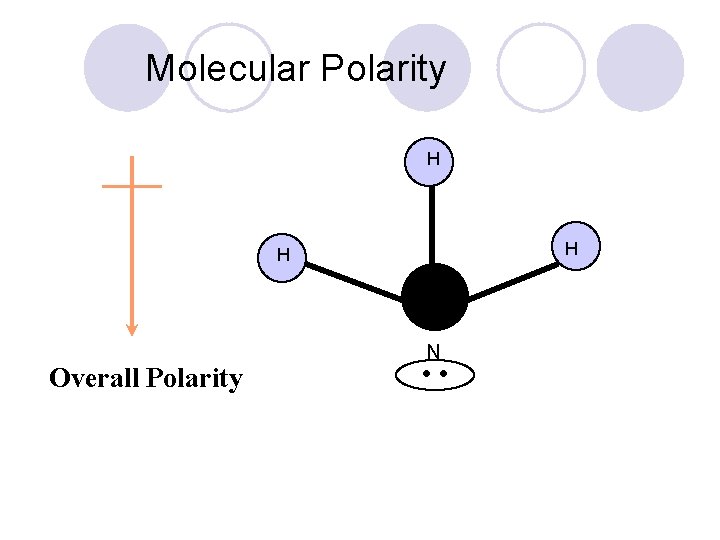
Molecular Polarity H H H Overall Polarity N • •

General Rule for determining Polarity. l. Non-Polar ¡Zero Unshared Pairs ¡If all the bonded atoms are of the same type. l. Polar ¡ 1 or More Unshared Pairs ¡ Unless linear shape
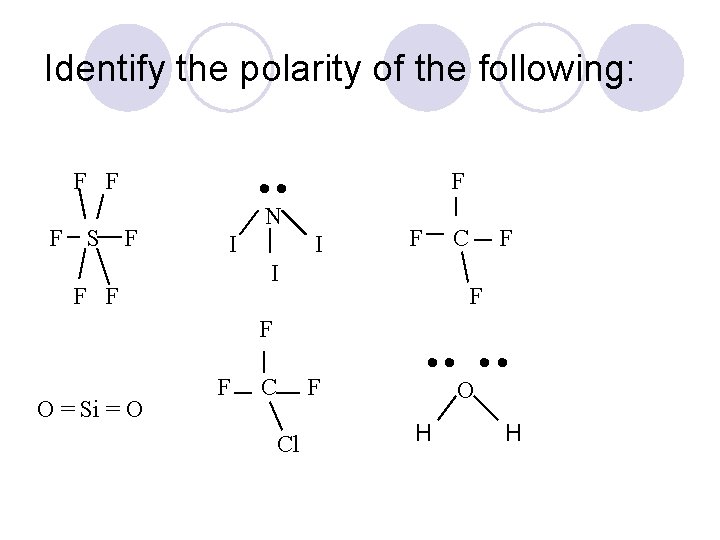
Identify the polarity of the following: F F F S F F N I I F C I F F F O = Si = O F Cl O H H
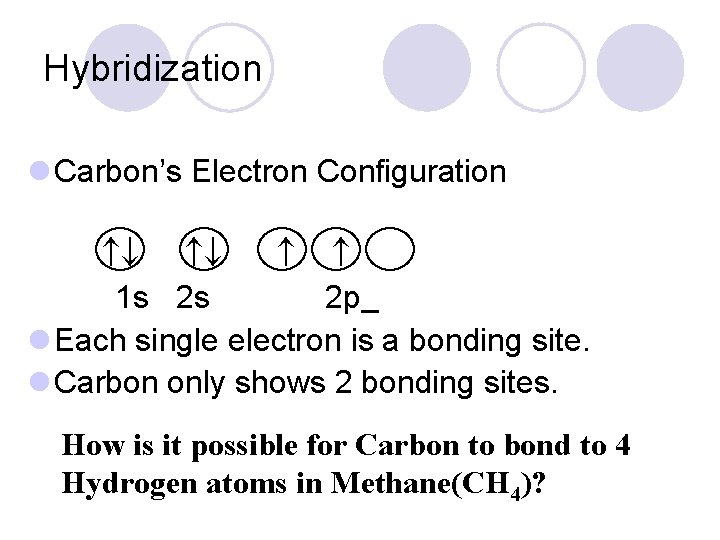
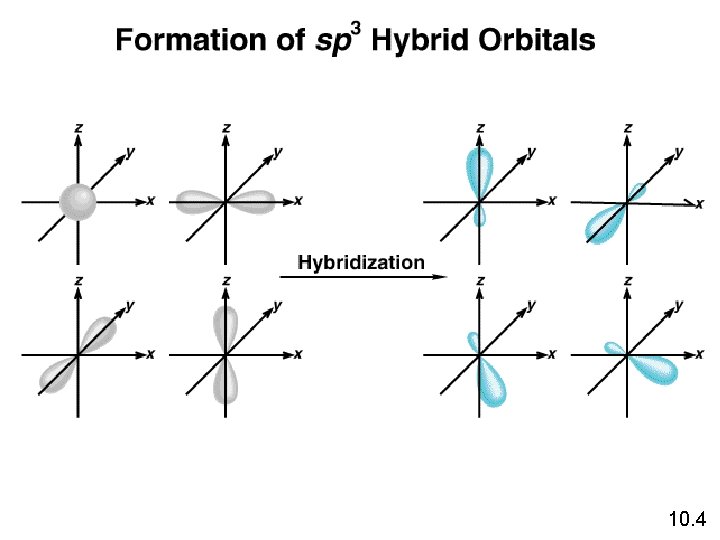
Hybridization l Carbon’s Electron Configuration 1 s 2 s 2 p l Each single electron is a bonding site. l Carbon only shows 2 bonding sites. . How is it possible for Carbon to bond to 4 Hydrogen atoms in Methane(CH 4)?

Hybridization l Carbon’s New Electron Configuration 1 s 2 s 2 p l Moves one of the 2 s electrons into the empty 2 p orbital. l Creating four bonding sites. So we would expect 3 bonds to be identical and one to be different!.
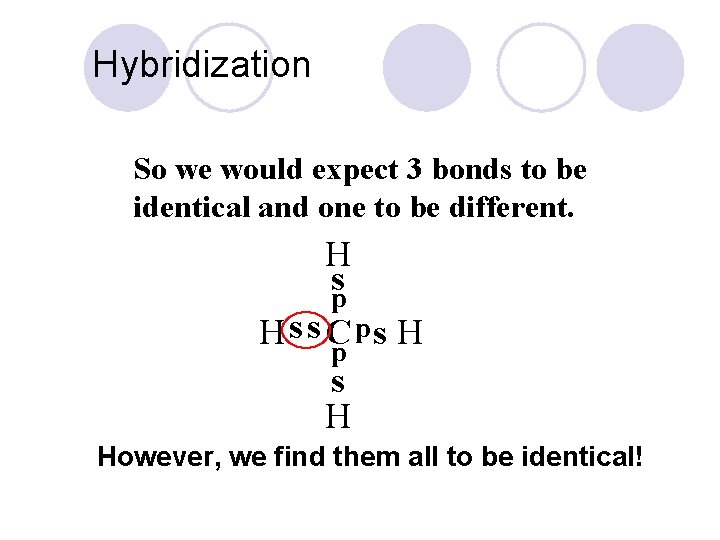
Hybridization So we would expect 3 bonds to be identical and one to be different. H sp ps H Hss. C p s H However, we find them all to be identical!
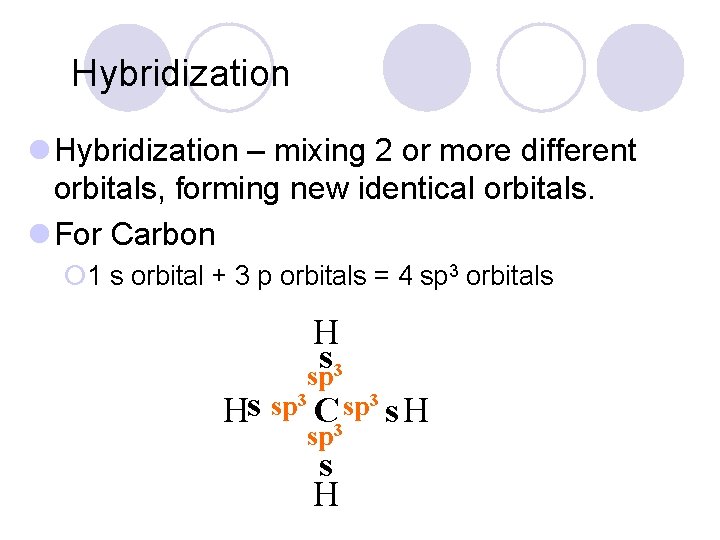
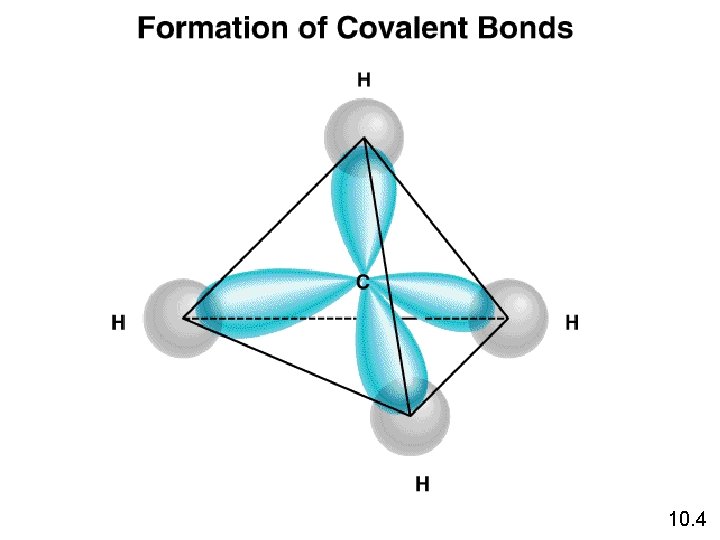
Hybridization l Hybridization – mixing 2 or more different orbitals, forming new identical orbitals. l For Carbon ¡ 1 s orbital + 3 p orbitals = 4 sp 3 orbitals H s sp 3 3 3 sp sp s H C 3 s H sp s H
10. 4
10. 4

Possible Combinations l 1 s +1 p = 2 sp orbitals l 1 s +2 p’s = 3 sp 2 orbitals l 1 s +3 p’s = 4 sp 3 orbitals l 1 s +3 p’s +1 d = 5 sp 3 d orbitals l 1 s +3 p’s +2 d’s = 6 sp 3 d 2 orbitals

Simplify l To determine hybrid orbitals for a molecule. ¡ 1 st count the number of bonded atoms & unshared pairs ¡ 2 nd use the following chart, where the number is the count from step 1. 123456 spppdd ¡ 3 rd use a superscript for a letter that appears more than once. Example H 2 O = 4 sppp sp 3 ¡ Example BF 3= 3 spp sp 2

Practice l Determine the hybridization and Polarity of the central atom for the following molecules: 1) CO 2 2) SO 2 3) SF 6 4) Si. O 2

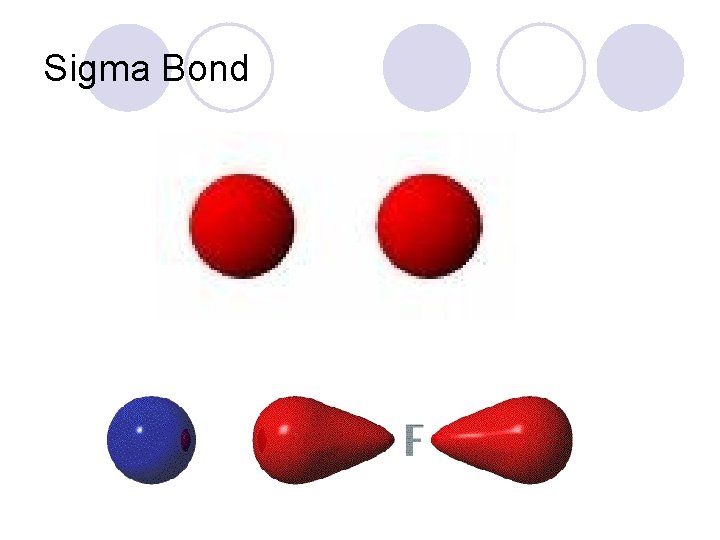
Molecular Bonding l Molecular Orbitals ¡Resulting orbital formed by the overlap of 2 atomic orbitals. l 2 Types of Molecular Bonds ¡Sigma Bond l. Molecular orbital that is formed along the bonding axis. ¡Pi Bond l. Molecular orbital that is formed above or below the bonding axis.
Sigma Bond

Sigma with Pi bond

Sigma with 2 Pi bond
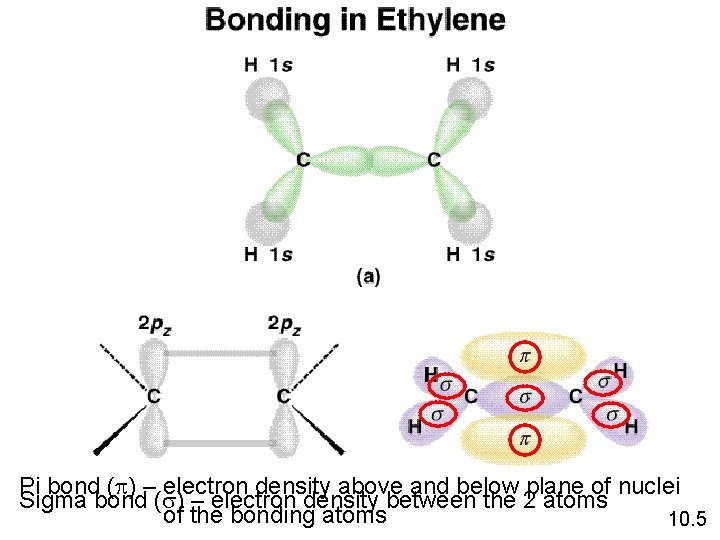
Pi bond (p) – electron density above and below plane of nuclei Sigma bond (s) – electron density between the 2 atoms of the bonding atoms 10. 5
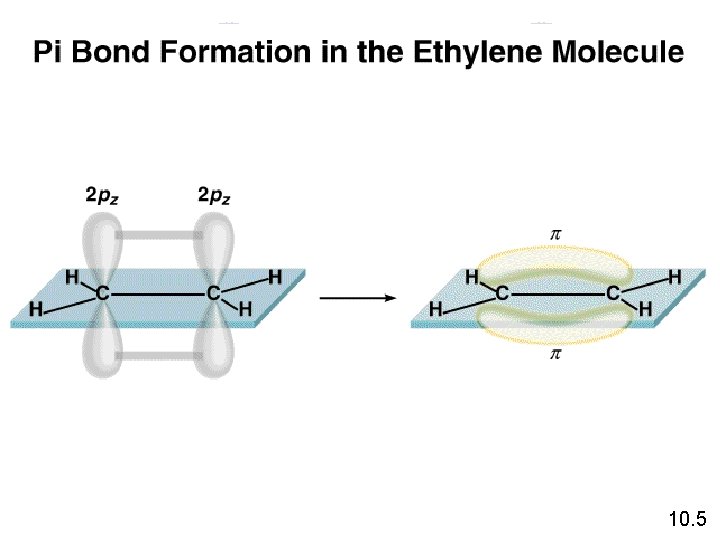
10. 5
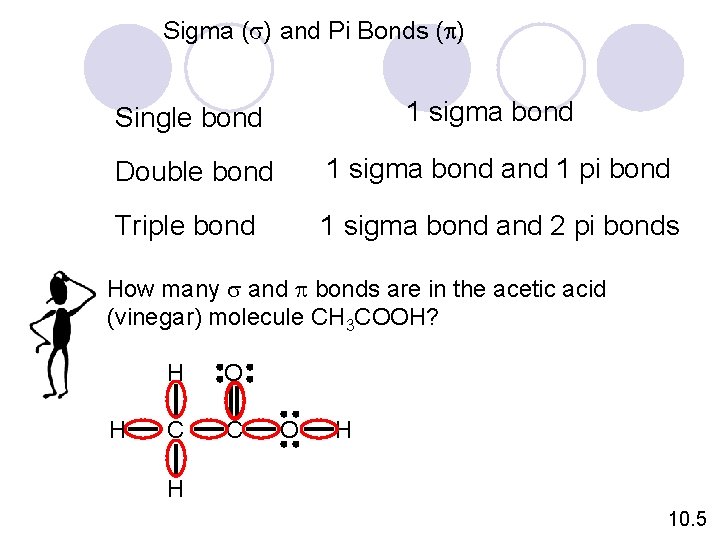
Sigma (s) and Pi Bonds (p) 1 sigma bond Single bond Double bond 1 sigma bond and 1 pi bond Triple bond 1 sigma bond and 2 pi bonds How many s and p bonds are in the acetic acid (vinegar) molecule CH 3 COOH? O H H C C O H H 10. 5

Intermolecular Forces l Forces of attraction between molecules. ¡ Vary in strength ¡ Weaker than Intramolecular Forces (Bonds) l Strength of IMF’s is related to the substance’s boiling points ¡ IMF BP l 3 Types of IMF’s ¡ Dipole-Dipole ¡ Hydrogen Bonding ¡ London Dispersion
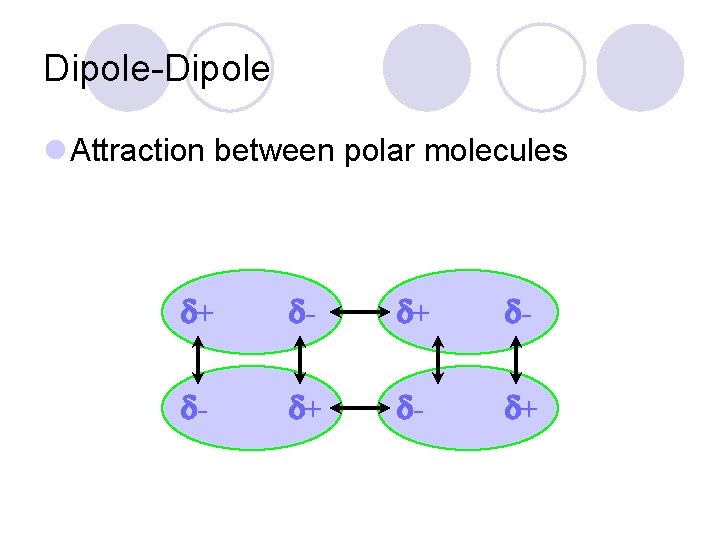
Dipole-Dipole l Attraction between polar molecules + - - +
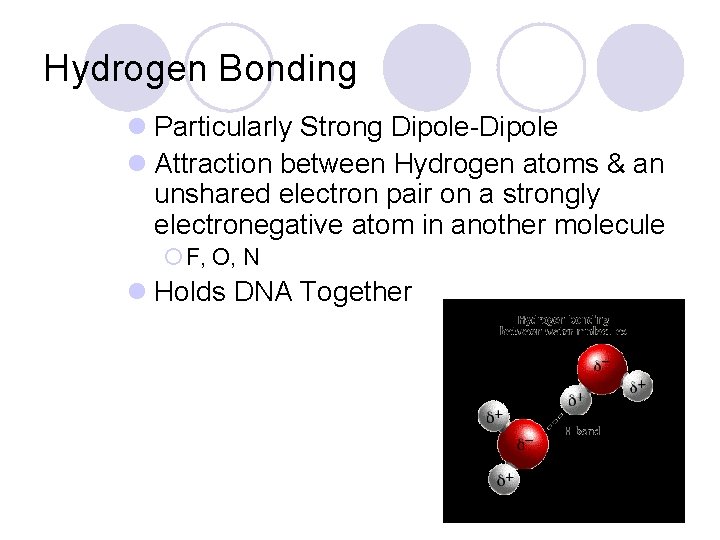
Hydrogen Bonding l Particularly Strong Dipole-Dipole l Attraction between Hydrogen atoms & an unshared electron pair on a strongly electronegative atom in another molecule ¡ F, O, N l Holds DNA Together

London Dispersion Forces l Attractions between molecules that have an induced dipole or a momentary dipole ¡Due to the constant motion of molecules Induced dipole
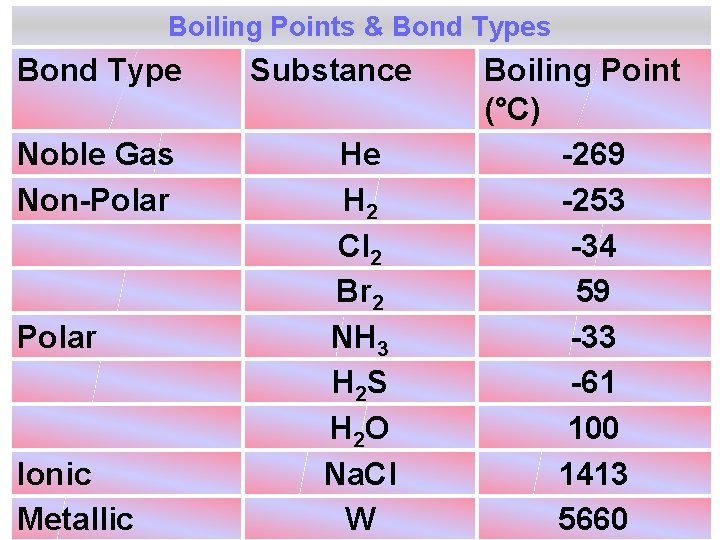
Boiling Points & Bond Types Bond Type Noble Gas Non-Polar Ionic Metallic Substance He H 2 Cl 2 Br 2 NH 3 H 2 S H 2 O Na. Cl W Boiling Point (°C) -269 -253 -34 59 -33 -61 100 1413 5660
- Slides: 64