5Sulfur and oxygen react in a combination reaction

5)Sulfur and oxygen react in a combination reaction to produce sulfur trioxide, an environmental 5) pollutant: 2 S (s) + 3 O 2 (g) 2 SO 3 (g) In a particular experiment, the reaction of 1. 0 g S with 1. 0 g O 2 produced 0. 80 g of SO 3. The % yield in this experiment is _____. AND calculate the moles of unreacted excess reagent. Calculate the moles of the excess reagent that is unconsumed at the end ofreaction. MOLES = MASS/GFM S MOLES = 1. 0 g s/ 32. 04 g/mol MOLES = 0. 031191 mol S MOLES = MASS/GFM O 2 MOLES = 1. 0 g O 2 / 32. 00 g/mol MOLES = 0. 03125 mol O 2 S O 2 = 0. 031191 , 3 0. 03125. 666 < 1 The actual ratio is larger than theoretical, therefore the actual ratio fraction is too large, the denominator is to small, O 2 limiting.
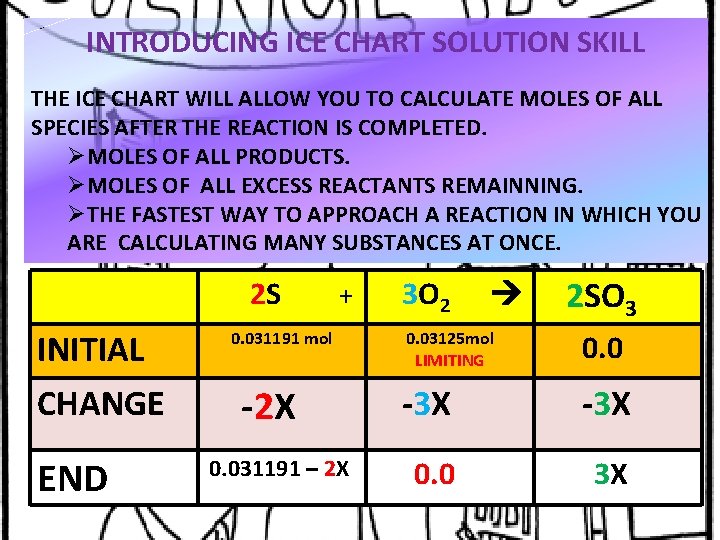
INTRODUCING ICE CHART SOLUTION SKILL THE ICE CHART WILL ALLOW YOU TO CALCULATE MOLES OF ALL SPECIES AFTER THE REACTION IS COMPLETED. ØMOLES OF ALL PRODUCTS. ØMOLES OF ALL EXCESS REACTANTS REMAINNING. ØTHE FASTEST WAY TO APPROACH A REACTION IN WHICH YOU ARE CALCULATING MANY SUBSTANCES AT ONCE. 2 S INITIAL CHANGE END + 0. 031191 mol -2 X 0. 031191 – 2 X 3 O 2 2 SO 3 0. 03125 mol LIMITING 0. 0 -3 X 0. 0 3 X

DETERMINING THE VALUE OF “X” 1. X IS DETERMINED BY THE LIMITING REAGENT. 2. IN THIS PROBLEM 0. 03125 mol = 3 X FROM THE LIMITING REAGENT OXYGEN. X = 0. 0104111 MOL. 3. THE EXCESS REAGENT IS SULFER, WHICH IS 0. 031191 – 2 X 0. 031191 – 2(0. 0104111) 0. 01036 MOLE OF S REMAINS 4. THE PRODUCT SO 3 = 2 X = 2(0. 0104111) = 0. 0208222 MOL ( 1. 667 GRAMS)
- Slides: 3