5 Types of Chemical Reactions 1 Combustion Reactions

5 Types of Chemical Reactions

1. Combustion Reactions n Occurs when oxygen reacts with a hydrocarbon to produce water and carbon dioxide. n A hydrocarbon is a compound containing only Carbon, Hydrogen and sometimes oxygen. n Example: C 10 H 8(s) + 12 O 2 (g) 10 CO 2 (g) + 4 H 2 O(g)

2. Synthesis (or Combination) n the combination of 2 or more substances to form a compound n only one product A + B AB n. Example: 2 Cu + O 2 2 Cu. O
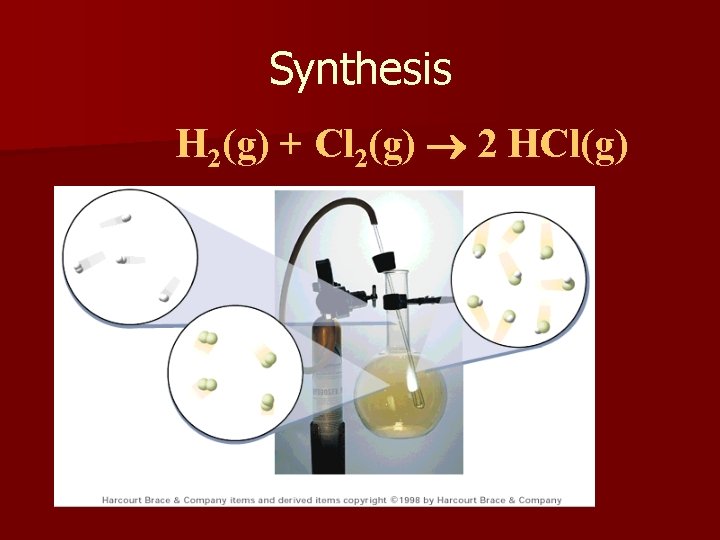
Synthesis H 2(g) + Cl 2(g) 2 HCl(g)
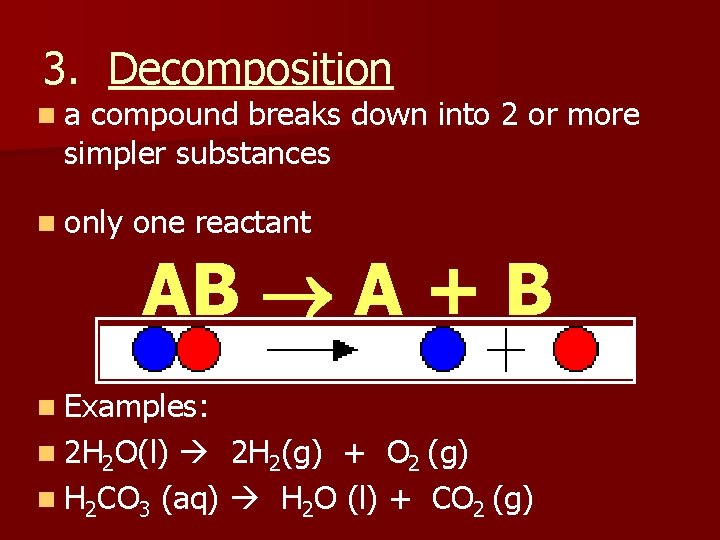
3. Decomposition na compound breaks down into 2 or more simpler substances n only one reactant AB A + B n Examples: n 2 H 2 O(l) 2 H 2(g) + O 2 (g) n H 2 CO 3 (aq) H 2 O (l) + CO 2 (g)
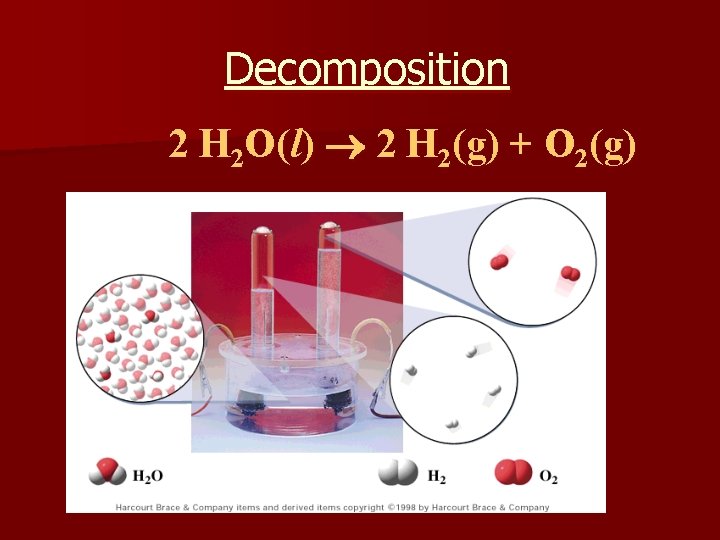
Decomposition 2 H 2 O(l) 2 H 2(g) + O 2(g)
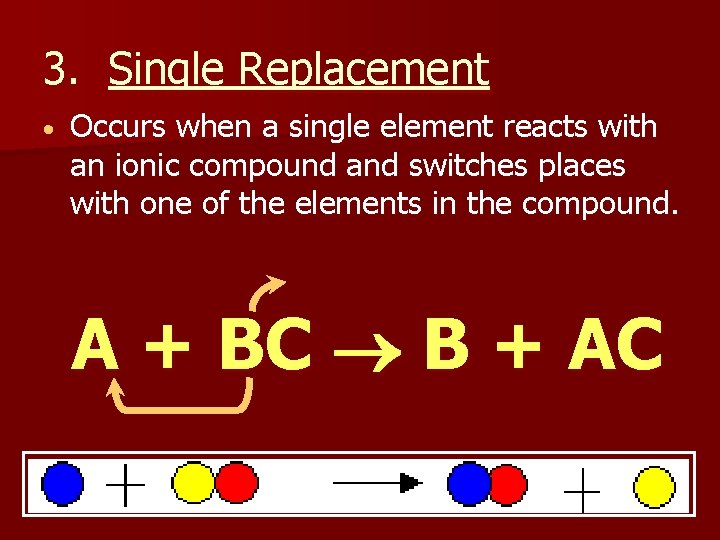
3. Single Replacement Occurs when a single element reacts with an ionic compound and switches places with one of the elements in the compound. A + BC B + AC

4. Single Displacement n General Form: AB + C AC + B n Cation Displacement Al + Cu. Cl 2 (aq) Cu + Al. Cl 3(aq) n Anion Displacement Cl 2 + KBr KCl + Br 2
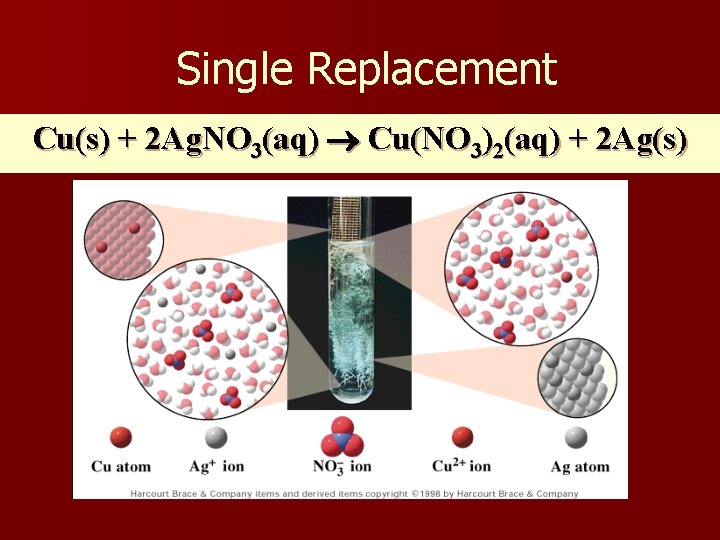
Single Replacement Cu(s) + 2 Ag. NO 3(aq) Cu(NO 3)2(aq) + 2 Ag(s)
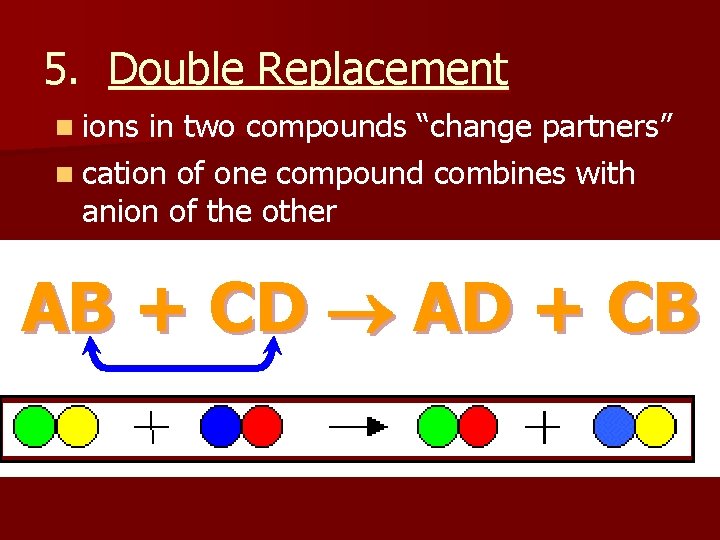
5. Double Replacement n ions in two compounds “change partners” n cation of one compound combines with anion of the other AB + CD AD + CB
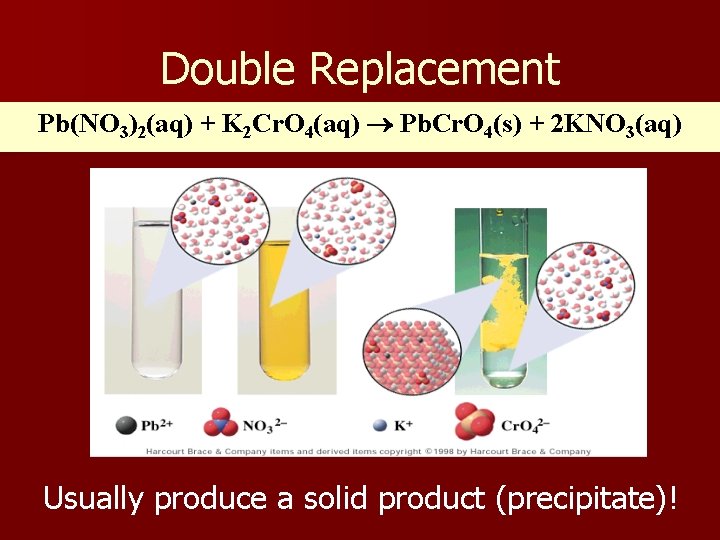
Double Replacement Pb(NO 3)2(aq) + K 2 Cr. O 4(aq) Pb. Cr. O 4(s) + 2 KNO 3(aq) Usually produce a solid product (precipitate)!

Steps to Classifying Reactions 1. Does the reaction contain oxygen, carbon dioxide and water? If Yes, then it is combustion 2. Do simple molecules form one more complicated product? If yes, then it is synthesis/combination. 3. Does a single reactant break apart to 2 or more simpler products? Then it is decomposition

Steps to Classifying Reactions Continued. . 4. Is there an ionic compound a single element on the reactant side and product side? Then it is single displacement. 5. Are all compounds ionic? Then it is double displacment/Replacement.

Examples C 3 H 7 OH + O 2 CO 2 + H 2 O n Combustion Mg + O 2 Mg. O n Synthesis H 2 O 2 H 2 O + O 2 n Decomposition n Al + Cu. Cl 2 Cu + Al. Cl 3 n n Pb(NO 3)2 + KI Pb. I 2 + KNO 3 n n Mg. O n Single Displacement n Double Displacement
- Slides: 14