5 THREE STATES OF MATTER 2006 BrooksCole Thomson

5 THREE STATES OF MATTER © 2006 Brooks/Cole - Thomson

General Properties of Gases • There is a lot of “free” space in a gas. • Gases can be expanded infinitely. • Gases occupy containers uniformly and completely. • Gases diffuse and mix rapidly. © 2006 Brooks/Cole - Thomson 6

7 Properties of Gases Gas properties can be modeled using math. Model depends on— • • © 2006 Brooks/Cole - Thomson V = volume of the gas (L) T = temperature (K) n = amount (moles) P = pressure (atmospheres)

Pressure Hg rises in tube until force of Hg (down) balances the force of atmosphere (pushing up). P of Hg pushing down related to • Hg density • column height © 2006 Brooks/Cole - Thomson 9

Pressure Column height measures P of atmosphere • 1 standard atm = 760 mm Hg = 29. 9 inches Hg = about 34 feet of water SI unit is PASCAL, Pa, where 1 atm = 101. 325 k. Pa © 2006 Brooks/Cole - Thomson 10

IDEAL GAS LAW P V = n R T Brings together gas properties. Can be derived from experiment and theory. © 2006 Brooks/Cole - Thomson 11

12 Boyle’s Law If n and T are constant, then PV = (n. RT) = k This means, for example, that P goes up as V goes down. © 2006 Brooks/Cole - Thomson Robert Boyle (1627 -1691). Son of Earl of Cork, Ireland.

Boyle’s Law A bicycle pump is a good example of Boyle’s law. As the volume of the air trapped in the pump is reduced, its pressure goes up, and air is forced into the tire. © 2006 Brooks/Cole - Thomson 13

14 Charles’s Law If n and P are constant, then V = (n. R/P)T = k. T V and T are directly related. © 2006 Brooks/Cole - Thomson Jacques Charles (17461823). Isolated boron and studied gases. Balloonist.

16 Charles’s Law Balloons immersed in liquid N 2 (at -196 ˚C) will shrink as the air cools (and is liquefied). © 2006 Brooks/Cole - Thomson
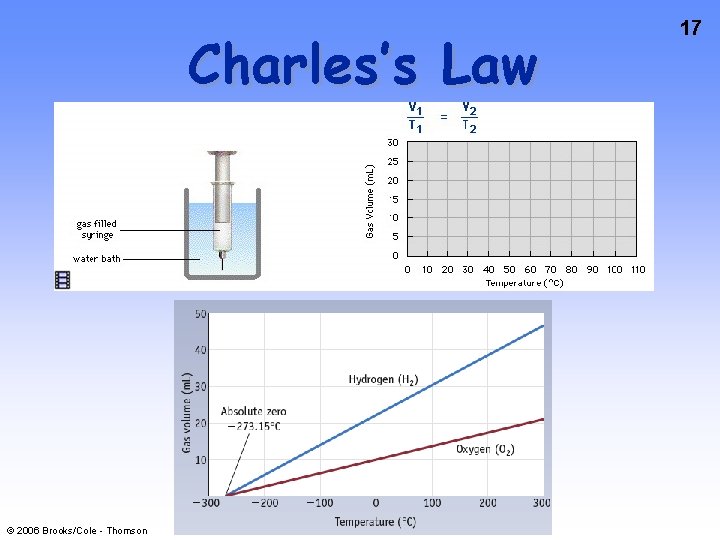
Charles’s Law © 2006 Brooks/Cole - Thomson 17

Avogadro’s Hypothesis Equal volumes of gases at the same T and P have the same number of molecules. V = n (RT/P) = kn V and n are directly related. twice as many molecules © 2006 Brooks/Cole - Thomson 18

21 © 2006 Brooks/Cole - Thomson

GAS DENSITY Screen 12. 5 Higher Density air © 2006 Brooks/Cole - Thomson 22 Low density helium

23 What are the units of density? Grams/volume Molar mass / molar volume = Molar mass / 22. 4 L = Density of a gas at STP D(gas) = M / 22. 4 L © 2006 Brooks/Cole - Thomson

24 Given: d(gas) = M / 22. 4 L d Is directly related to …. 1. M 2. 3. 4. 5. 22. 4 Gas Molar mass Both 1 and 4 © 2006 Brooks/Cole - Thomson

25 The gas with greatest density at STP is …. . . 1. 2. 3. 4. NH 3 H 2 O CO 2 SO 2 © 2006 Brooks/Cole - Thomson

26 USING GAS DENSITY The density of air at 15 o. C and 1. 00 atm is 1. 23 g/L. What is the molar mass of air? 1. Calc. moles of air. V = 1. 00 L P = 1. 00 atm T = 288 K n = PV/RT = 0. 0423 mol 2. Calc. molar mass/mol = 1. 23 g/0. 0423 mol = 29. 1 g/mol © 2006 Brooks/Cole - Thomson

27 © 2006 Brooks/Cole - Thomson

28 The Ideal Gas Law • PV = n. RT • n is moles of gas • The ideal gas equation is used whenever moles (or grams) of a substance is either given or needed. • Watch out for the units! • Units must match the units of R. • R = 0. 082057 L atm /mol K © 2006 Brooks/Cole - Thomson

Using PV = n. RT How much N 2 is required to fill a small room with a volume of 960 cubic feet (27, 000 L) to P = 745 mm Hg at 25 o. C? R = 0. 082057 L • atm/K • mol © 2006 Brooks/Cole - Thomson 29

30 Work on this problem while the class registers their clickers. © 2006 Brooks/Cole - Thomson

Using PV = n. RT How much N 2 is req’d to fill a small room with a volume of 960 cubic feet (27, 000 L) to P = 745 mm Hg at 25 o. C? R = 0. 082057 L • atm/K • mol Solution 1. Get all data into proper units V = 27, 000 L T = 25 o. C + 273 = 298 K P = 745 mm Hg (1 atm/760 mm Hg) = 0. 98 atm © 2006 Brooks/Cole - Thomson 31

Using PV = n. RT How much N 2 is req’d to fill a small room with a volume of 960 cubic feet (27, 000 L) to P = 745 mm Hg at 25 o. C? R = 0. 082057 L • atm/K • mol Solution 2. Now calc. n = PV / RT n = 1. 1 x 103 mol (or about 30 kg of gas) © 2006 Brooks/Cole - Thomson 32

Given: Ab=XYZ Variables that are directly related are …and… 1. 2. 3. 4. A, Z Y, b X, b All of above © 2006 Brooks/Cole - Thomson 33

Given: a. B = DF Inversely related are… and … 1. 2. 3. 4. D, F B, D B, a Both 1 and 3 © 2006 Brooks/Cole - Thomson 34

36 Gases and Stoichiometry 2 H 2 O 2(liq) ---> 2 H 2 O(g) + O 2(g) Decompose 1. 1 g of H 2 O 2 in a flask with a volume of 2. 50 L. What is the pressure of O 2 at 25 o. C? Of H 2 O? Strategy: Can you get moles of anything? What? Can you use moles of a liquid in the ideal gas equation? © 2006 Brooks/Cole - Thomson

37 NO! It is the ideal GAS law! Gases only © 2006 Brooks/Cole - Thomson

38 Gases and Stoichiometry 2 H 2 O 2(liq) ---> 2 H 2 O(g) + O 2(g) Decompose 1. 1 g of H 2 O 2 in a flask with a volume of 2. 50 L. What is the pressure of O 2 at 25 o. C? Of H 2 O? Strategy: What moles do you need? How will you get this? © 2006 Brooks/Cole - Thomson

39 Gases and Stoichiometry 2 H 2 O 2(liq) ---> 2 H 2 O(g) + O 2(g) Decompose 1. 1 g of H 2 O 2 in a flask with a volume of 2. 50 L. What is the pressure of O 2 at 25 o. C? Of H 2 O? Solution Now what will you do? © 2006 Brooks/Cole - Thomson

Gases and Stoichiometry 2 H 2 O 2(liq) ---> 2 H 2 O(g) + O 2(g) Decompose 1. 1 g of H 2 O 2 in a flask with a volume of 2. 50 L. What is the pressure of O 2 at 25 o. C? Of H 2 O? Solution P of O 2 = 0. 16 atm © 2006 Brooks/Cole - Thomson 40

Gases and Stoichiometry 2 H 2 O 2(liq) ---> 2 H 2 O(g) + O 2(g) Decompose 1. 1 g of H 2 O 2 in a flask with a volume of 2. 50 L. What is the pressure of O 2 at 25 o. C? Of H 2 O? Solution How can you find the pressure of water? Do a calculation like we just did but for water? Any other way? © 2006 Brooks/Cole - Thomson 41

Gases and Stoichiometry 2 H 2 O 2(liq) ---> 2 H 2 O(g) + O 2(g) What is P of H 2 O? Could calculate as above. But recall Avogadro’s hypothesis. V n at same T and P P n at same T and V P is proportional to n. There are 2 times as many moles of H 2 O as moles of O 2. Therefore, P of H 2 O is twice that of O 2. P of H 2 O = 0. 32 atm © 2006 Brooks/Cole - Thomson 42

43 Dalton’s Law of Partial Pressures 2 H 2 O 2(liq) ---> 2 H 2 O(g) + O 2(g) 0. 32 atm 0. 16 atm What is the total pressure in the flask? Ptotal in gas mixture = PA + PB +. . . Therefore, Ptotal = P(H 2 O) + P(O 2) = 0. 48 atm Dalton’s Law: total P is sum of PARTIAL pressures. © 2006 Brooks/Cole - Thomson

Dalton’s Law John Dalton 1766 -1844 © 2006 Brooks/Cole - Thomson 44

45 © 2006 Brooks/Cole - Thomson

KINETIC MOLECULAR THEORY (KMT) Theory used to explain gas laws. KMT assumptions are • Gases consist of molecules in constant, random motion. • P arises from collisions with container walls. • No attractive or repulsive forces between molecules. Collisions elastic. • Volume of molecules is negligible. © 2006 Brooks/Cole - Thomson 47
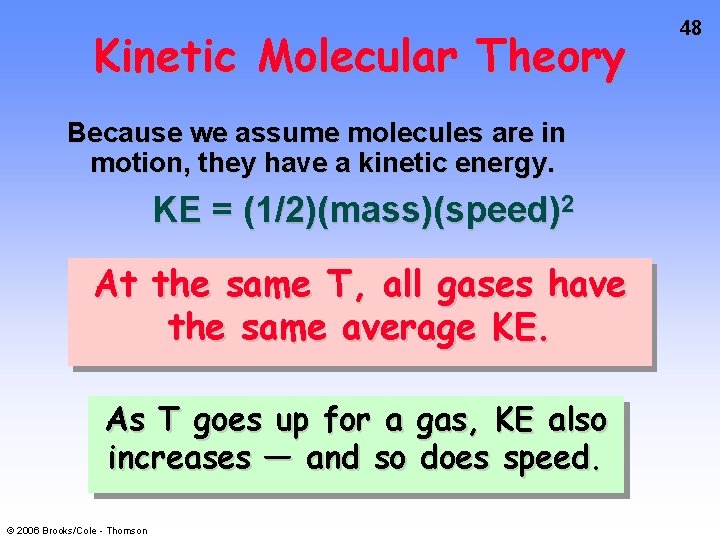
Kinetic Molecular Theory Because we assume molecules are in motion, they have a kinetic energy. KE = (1/2)(mass)(speed)2 At the same T, all gases have the same average KE. As T goes up for a gas, KE also increases — and so does speed. © 2006 Brooks/Cole - Thomson 48

49 © 2006 Brooks/Cole - Thomson
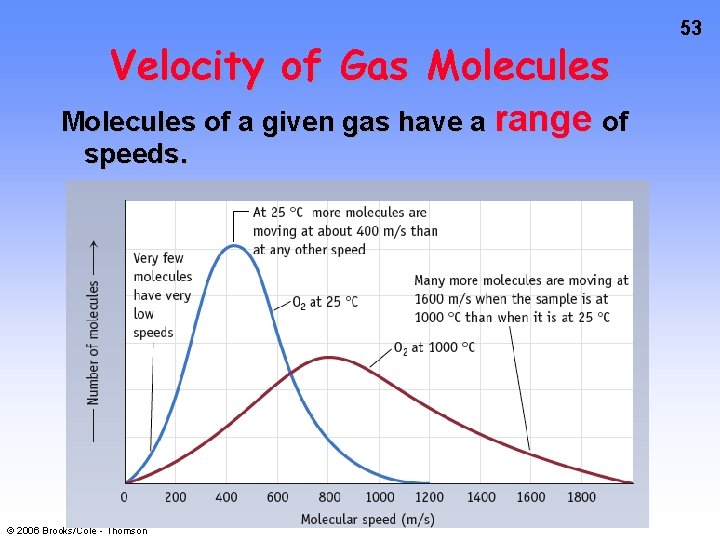
Velocity of Gas Molecules of a given gas have a range of speeds. © 2006 Brooks/Cole - Thomson 53
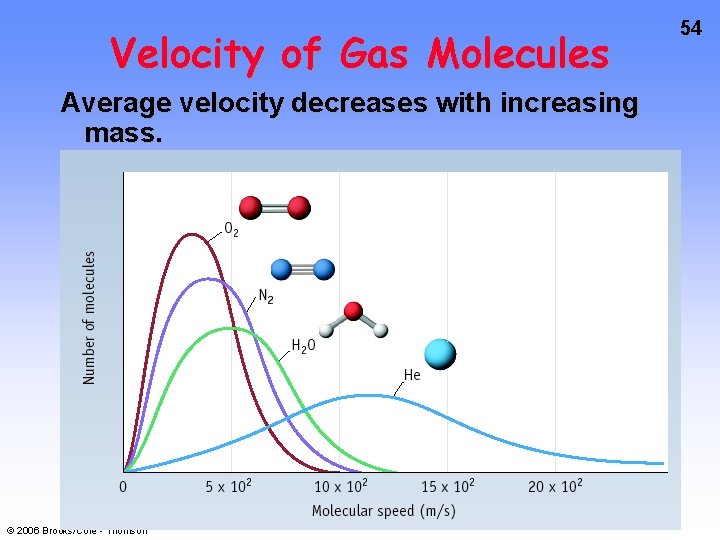
Velocity of Gas Molecules Average velocity decreases with increasing mass. © 2006 Brooks/Cole - Thomson 54

55 GAS DIFFUSION AND EFFUSION DIFFUSION is the gradual mixing of molecules of different gases. © 2006 Brooks/Cole - Thomson

GAS EFFUSION is the movement of molecules through a small hole into an empty container. Figure 12. 19 © 2006 Brooks/Cole - Thomson 56

57 GAS DIFFUSION AND EFFUSION Molecules effuse thru holes in a rubber balloon, for example, at a rate (= moles/time) that is • proportional to T • inversely proportional to M. Therefore, He effuses more rapidly than O 2 at same T. © 2006 Brooks/Cole - Thomson He

GAS DIFFUSION AND EFFUSION Graham’s law governs effusion and diffusion of gas molecules. Rate of effusion is inversely proportional to its molar mass. © 2006 Brooks/Cole - Thomson Thomas Graham, 1805 -1869. Professor in Glasgow and London. 58

Gas Diffusion relation of mass to rate of diffusion • HCl and NH 3 diffuse from opposite ends of tube. • Gases meet to form NH 4 Cl • HCl heavier than NH 3 • Therefore, NH 4 Cl forms closer to HCl end of tube. Active Figure 12. 18 © 2006 Brooks/Cole - Thomson 59

60 Using KMT to Understand Gas Laws Recall that KMT assumptions are • Gases consist of molecules in constant, random motion. • P arises from collisions with container walls. • No attractive or repulsive forces between molecules. Collisions elastic. • Volume of molecules is negligible. © 2006 Brooks/Cole - Thomson

Avogadro’s Hypothesis and Kinetic Molecular Theory P proportional to n — when V and T are constant © 2006 Brooks/Cole - Thomson 61
62 Gas Pressure, Temperature, and Kinetic Molecular Theory P proportional to T — when n and V are constant © 2006 Brooks/Cole - Thomson
Boyle’s Law and Kinetic Molecular Theory P proportional to 1/V — when n and T are constant © 2006 Brooks/Cole - Thomson 63

Deviations from Ideal Gas Law • Real molecules have volume. • There are intermolecular forces. – Otherwise a gas could not become a liquid. © 2006 Brooks/Cole - Thomson 64

Deviations from Ideal Gas Law Account for volume of molecules and intermolecular forces with VAN DER WAALS’s EQUATION. Measured V = V(ideal) Measured P ( P 2 + n a ----2 V ) V - nb n. RT vol. correction intermol. forces © 2006 Brooks/Cole - Thomson J. van der Waals, 1837 -1923, Professor of Physics, Amsterdam. Nobel Prize 1910. 65
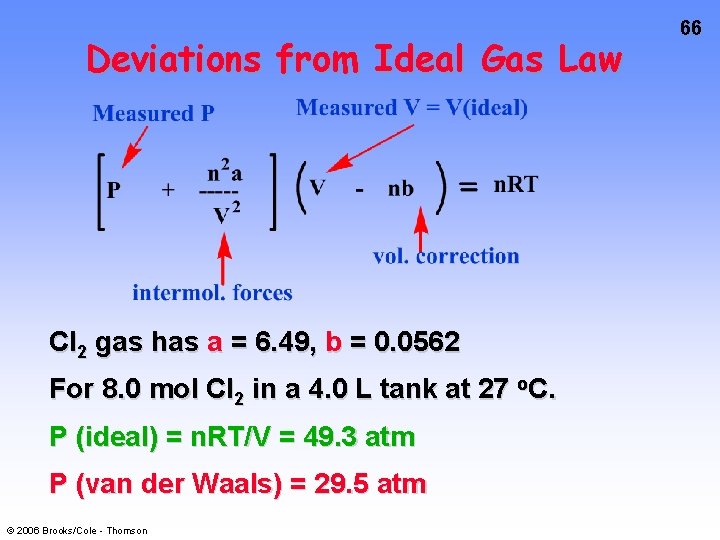
Deviations from Ideal Gas Law Cl 2 gas has a = 6. 49, b = 0. 0562 For 8. 0 mol Cl 2 in a 4. 0 L tank at 27 o. C. P (ideal) = n. RT/V = 49. 3 atm P (van der Waals) = 29. 5 atm © 2006 Brooks/Cole - Thomson 66

67 Please make your selection. . . 1. 2. 3. 4. Choice One Choice Two Choice Three Choice Four 100% 0% 0% © 2006 Brooks/Cole - Thomson Answer Now

68 Please make your selection. . . 1. 2. 3. 4. Choice One Choice Two Choice Three Choice Four 100% 0% 0% © 2006 Brooks/Cole - Thomson Answer Now
- Slides: 55