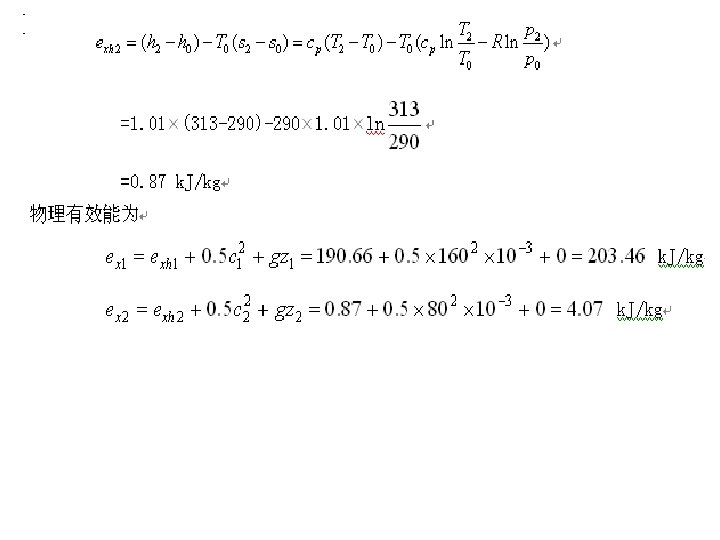5 Exergy 5 1 Introduction Evaluation of heat






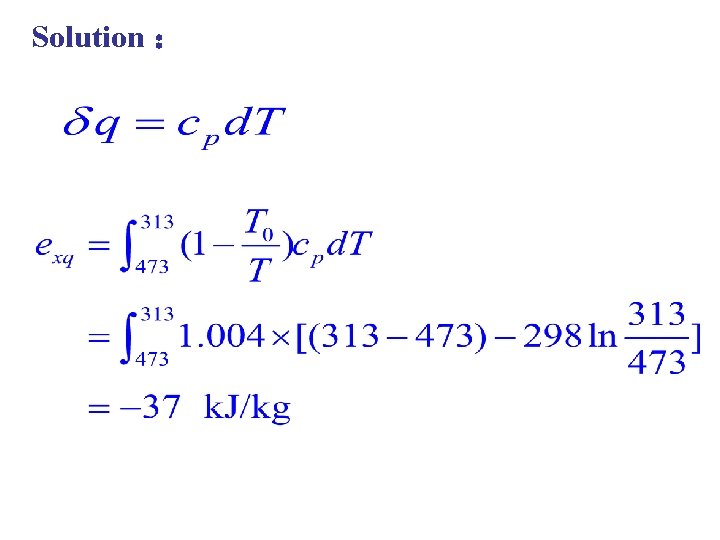





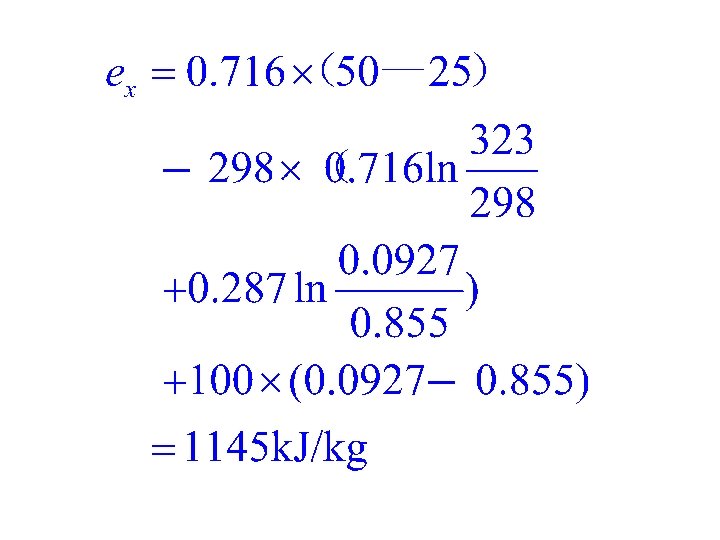





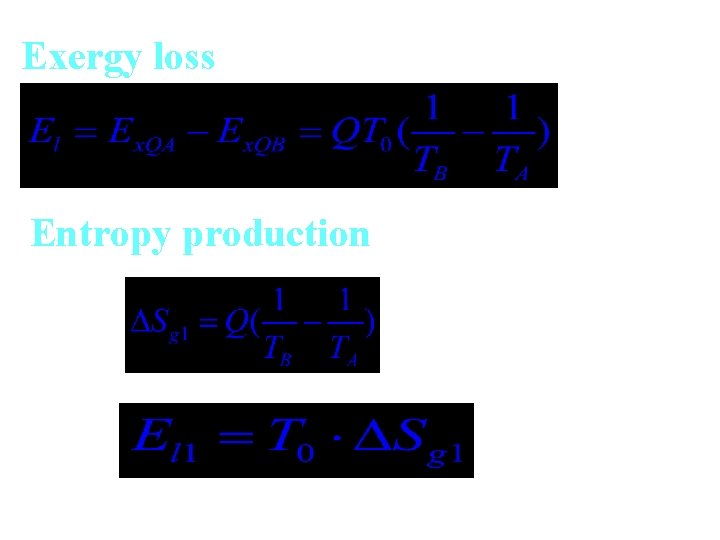



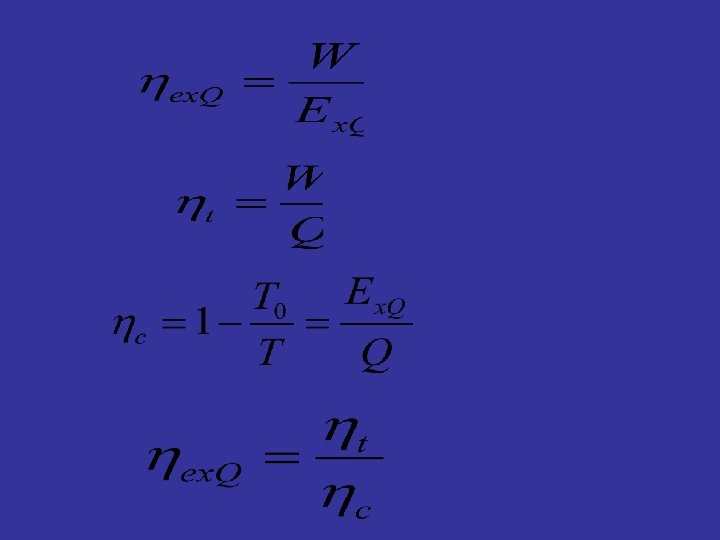


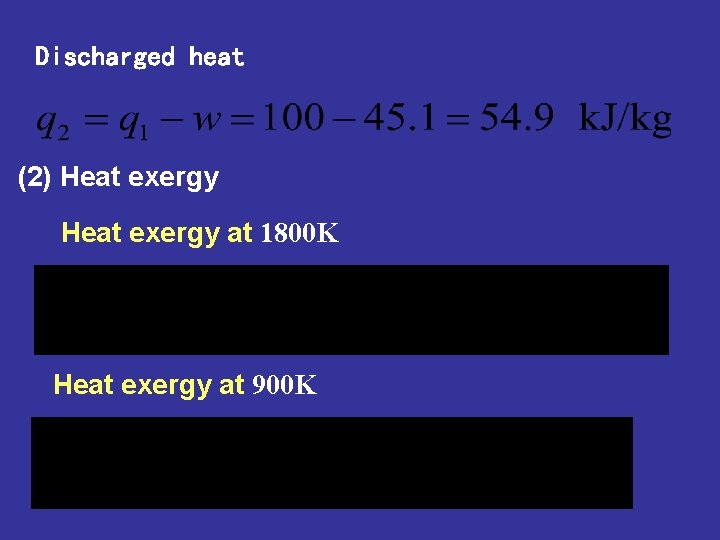












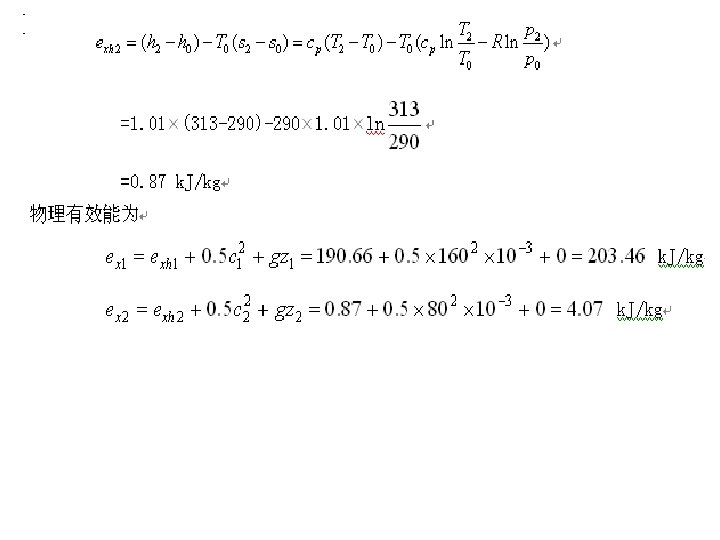









- Slides: 67

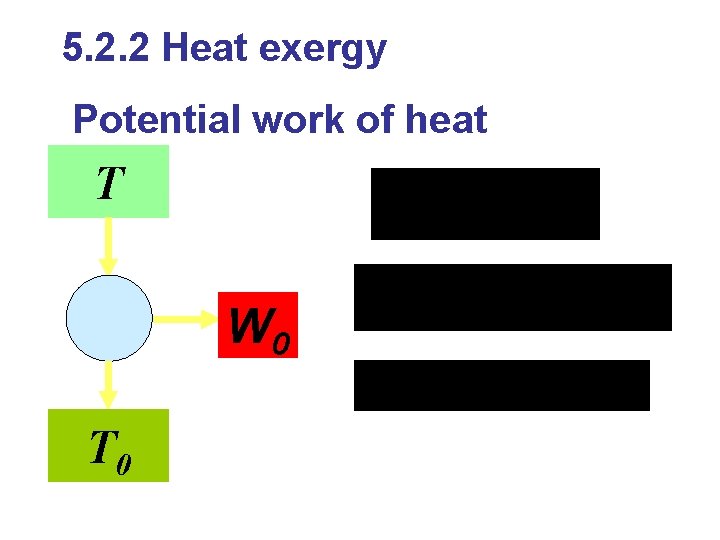
5 Exergy 5. 1 Introduction Evaluation of heat Quantity Quality Exergy and Anergy Full convertible energy: mechanical Partial convertible energy: heat Unconvertible energy: environmental

Exergy: useful work potential; available energy; The maximum useful work a system can delivered from a specified state to the state of its environment in theory. Anergy: unavailable energy, unconvertible energy Conditions for definition of exergy (1)based on environment; exergy of environmental energy is zero (2)reversible process (3)there is no other heat resource in the process.

5. 2 Calculation of exergy Type Chemical Physical � unbalance Chemical potential Temperature and pressure Kinetic � Velocity Position � Position Distribution Concentration

Unbalance Source� Electricity� voltage Hydraulic� Water level Pneumatic Wind pressure Wave� � Difference between interior and surface

5. 2. 1 Work resource Electricity, mechanical energy, pneumatic energy, hydraulic energy, can be converted to work entirely. Exergy of work resource = its total energy

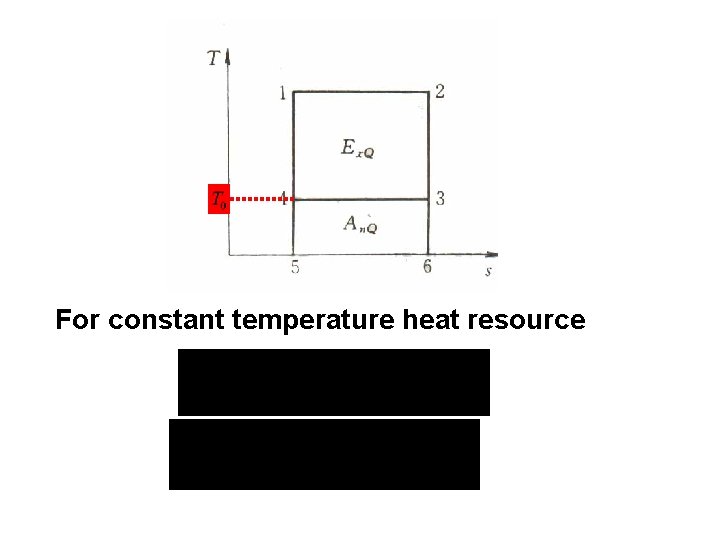
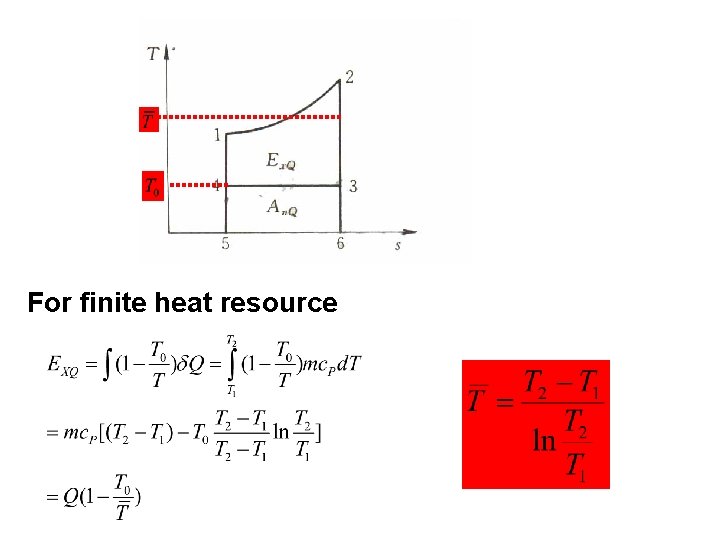
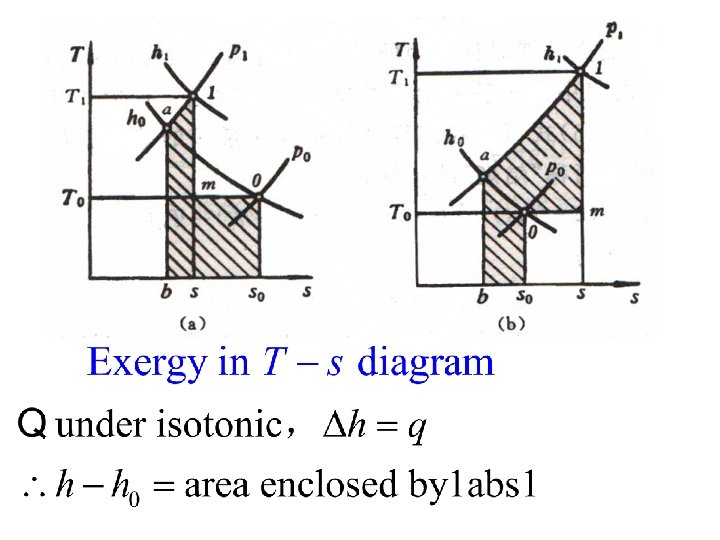
5. 2. 2 Heat exergy Potential work of heat T W 0 T 0

For constant temperature heat resource

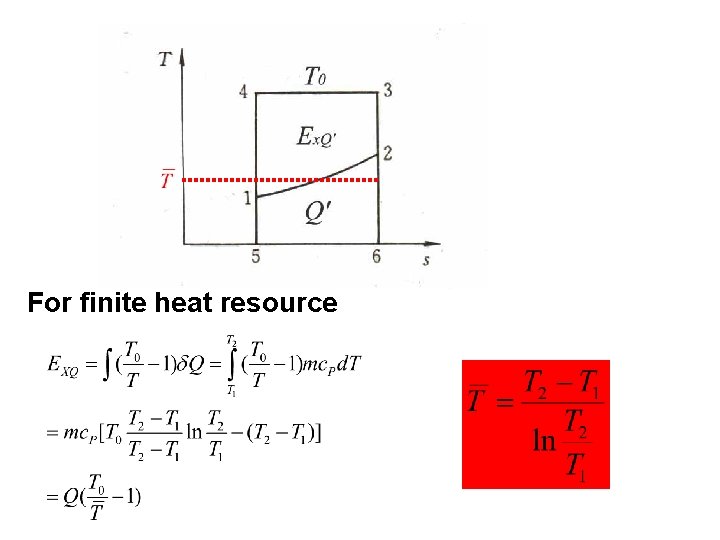
T 0 For finite heat resource

Influential factors: l. Heat quantity l. Heat resource temperature l. Environmental temperature T 0 If the system absorbs heat, it absorbs exergy; If the system discharges heat, it discharges exergy;

Example 1 kg air with temperature of 200℃ was cooled to 40℃. Please calculate the heat exergy. The specific cp=1. 004 k. J/(kg. K). the temperature is T 0=25℃。 heat of air is environmental
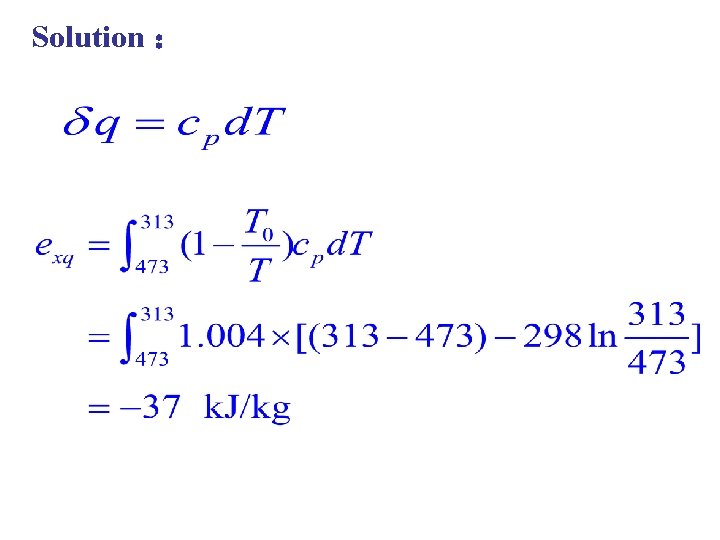
Solution :

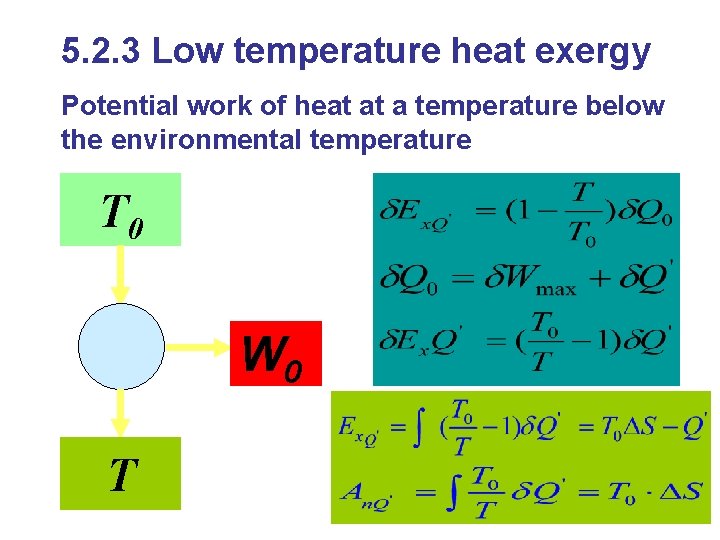
5. 2. 3 Low temperature heat exergy Potential work of heat at a temperature below the environmental temperature T 0 W 0 T

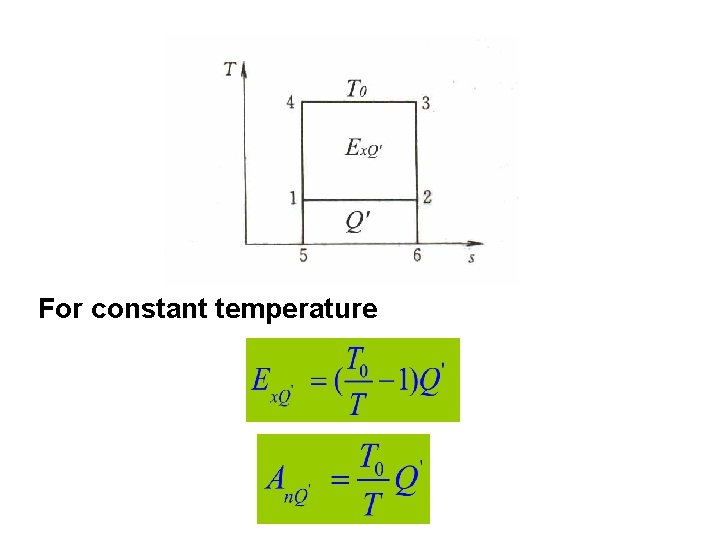
For constant temperature

For finite heat resource

Influential factors: l. Heat quantity l. Heat resource temperature l. Environmental temperature T 0 If the system absorbs heat, it discharges exergy; If the system discharges heat, it absorbs exergy;

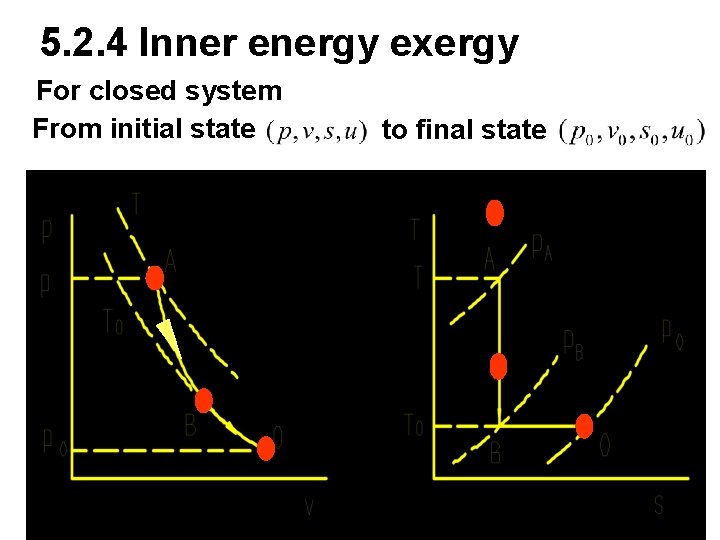
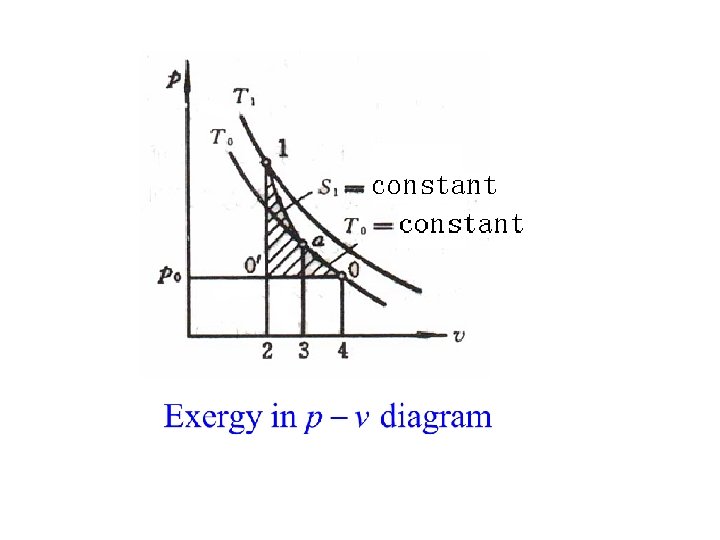
5. 2. 4 Inner energy exergy For closed system From initial state to final state

For A→B→O The dead work to resist environment The maximum work

For m kg substance The maximum available work


Example Please calculate the inner energy exergy of air in the state of 1 MPa and 50℃. The environmental pressure is p 0=0. 1 MPa,the temperature is T 0=25℃ and the specific heat is cv=0. 716 k. J/(kg·K)。

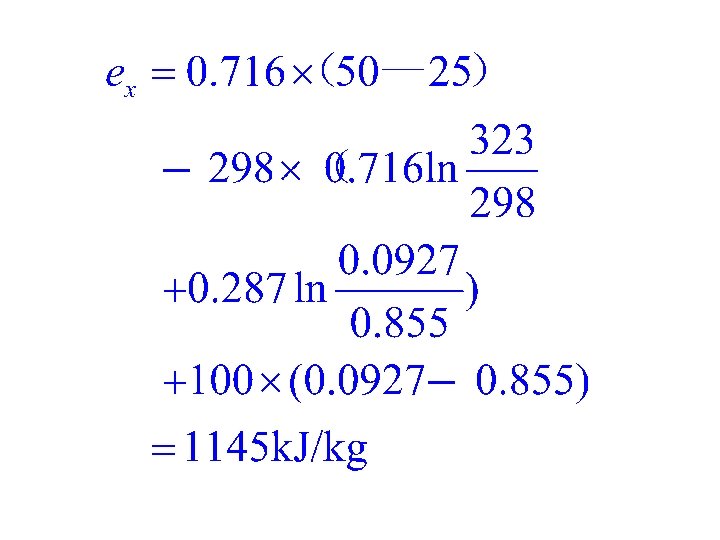

5. 2. 5 Enthalpy exergy For steady flow system From initial state to final state

For A→B→O For m kg substance The maximum available work


Example Please compare the enthalpy exergy value of saturated steam of 0. 5 MPa with 5 MPa. The environmental state is p 0=0. 1 MPa and T 0=20℃。

Solution : Look up properties in the table of water or steam h 0=84 k. J/kg s 0=0. 2963 k. J/(kg·K) h 1=2747. 5 k. J/kg s 1=6. 8192 k. J/(kg·K) h 2=2794. 2 k. J/kg s 2=5. 9735 k. J/(kg·K)

ex 1 =(h 1-h 0)-T 0(s 1-s 0) =(2747. 5-84)-293×(6. 8192-0. 2963) =752. 3 k. J/kg ex 2=(h 2-h 0)-T 0(s 2-s 0) =(2791. 2-84)-293×(5. 9735-0. 2963) =1046. 8 k. J/kg�

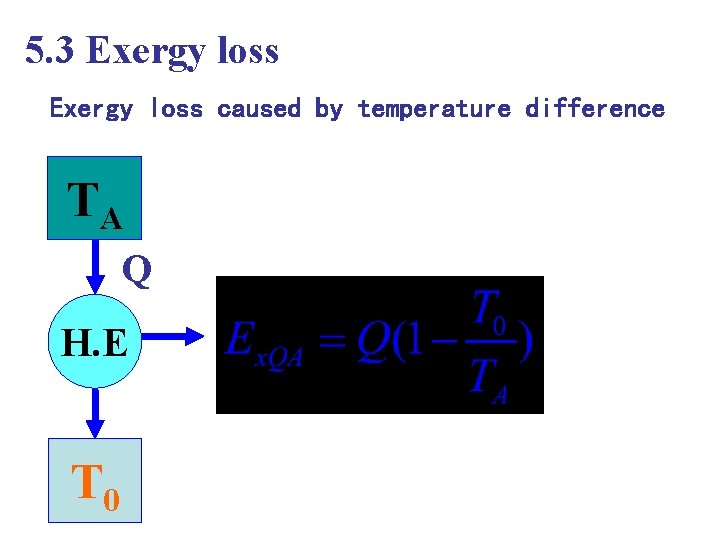
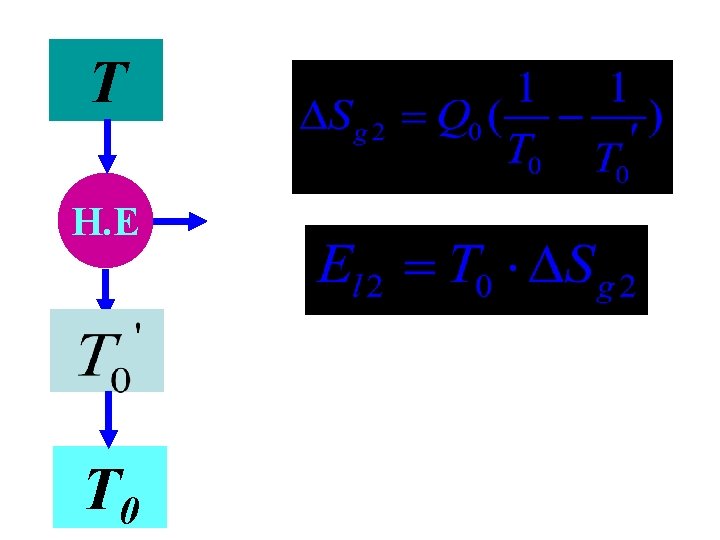
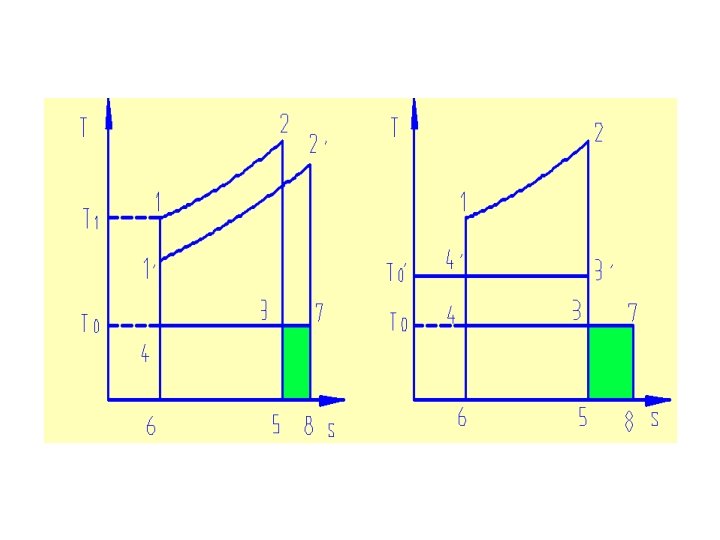
5. 3 Exergy loss caused by temperature difference TA Q H. E T 0

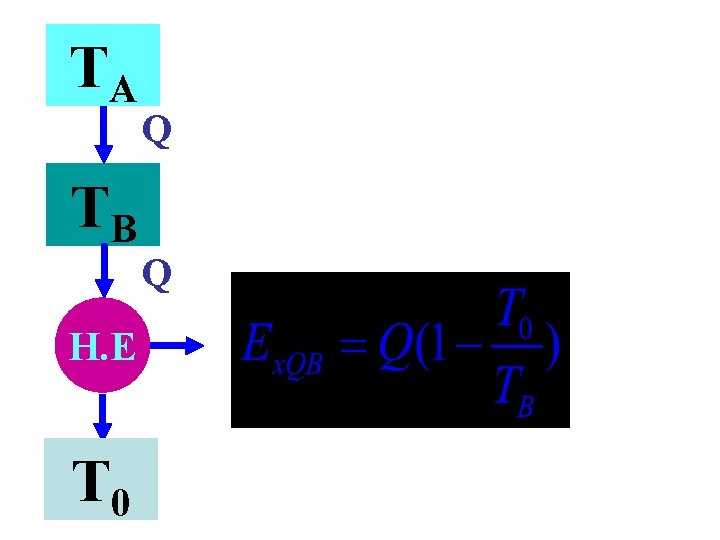
TA Q TB Q H. E T 0
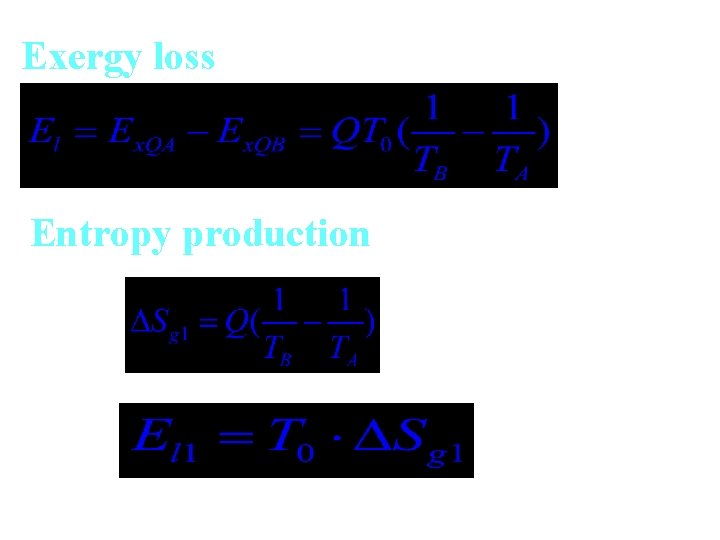
Exergy loss Entropy production

T H. E T 0


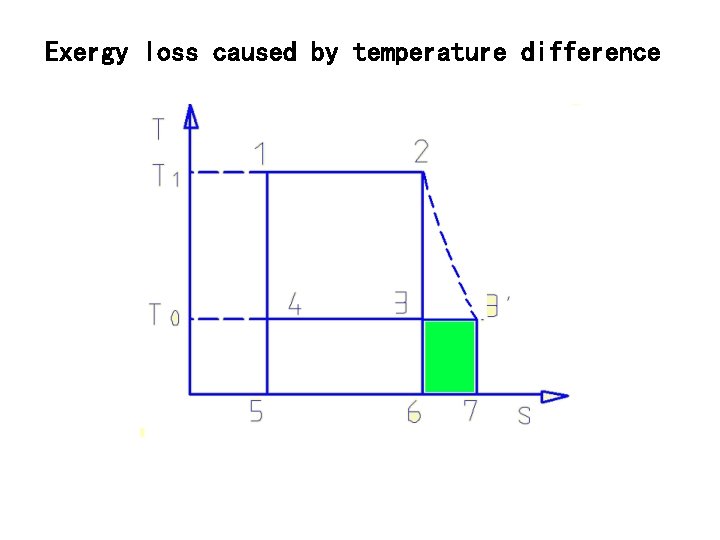
Exergy loss caused by temperature difference

If discharging temperature is The whole exergy loss

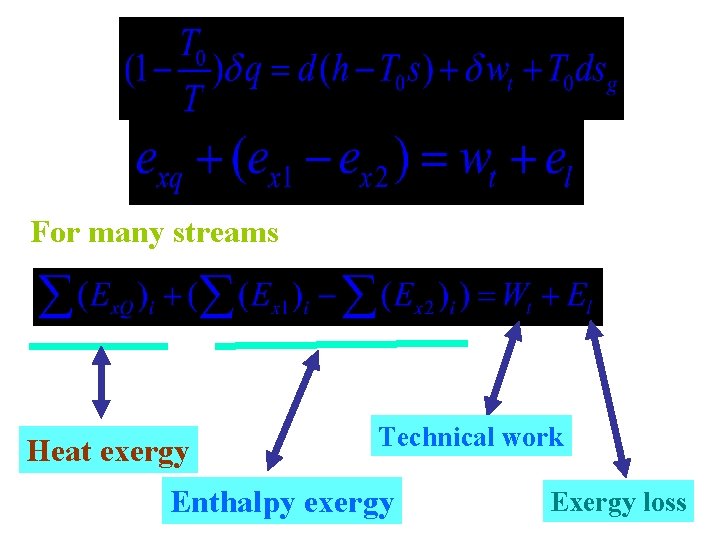
5. 4 exergy equation

For many streams Heat exergy Technical work Enthalpy exergy Exergy loss

5. 5 Exergy efficiency 1 2
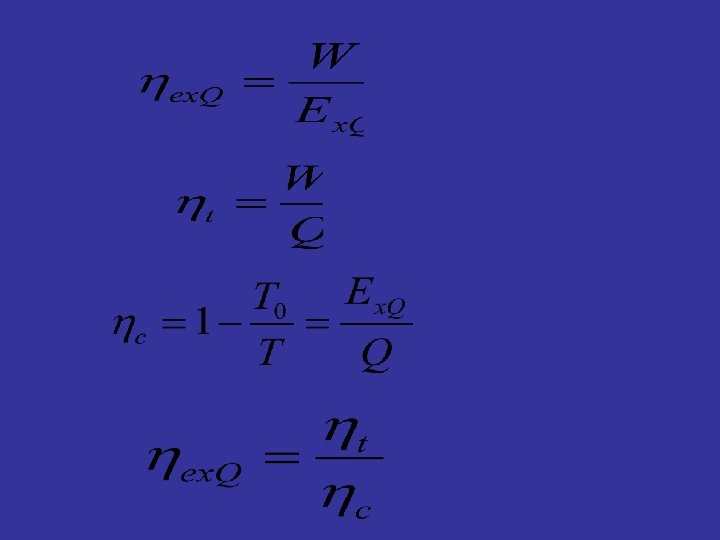

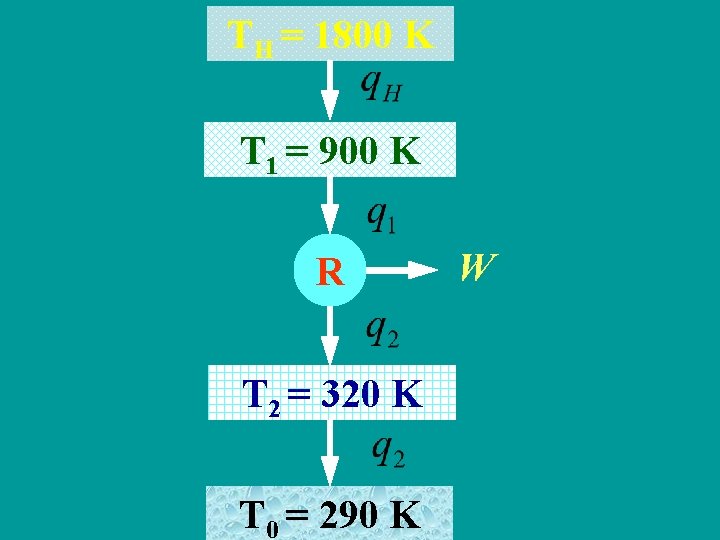
Example The high temperature is TH=1800 K and the low temperature is the environmental temperature that is T 0=290 K. A heat engine absorbs heat at T 1=900 K and discharges heat at T 2=320 K. The engine efficiency is 70% of that of corresponding carnot cycle。If each kilogram substance absorbs heat 100 k. J, please calculate: (1)the practical work of heat engine; (2)the heat exergy at given temperature; (3)the entropy production and exergy loss of each process; (4)the entropy increase of isolated system and the whole exergy loss.

TH = 1800 K T 1 = 900 K R T 2 = 320 K T 0 = 290 K W

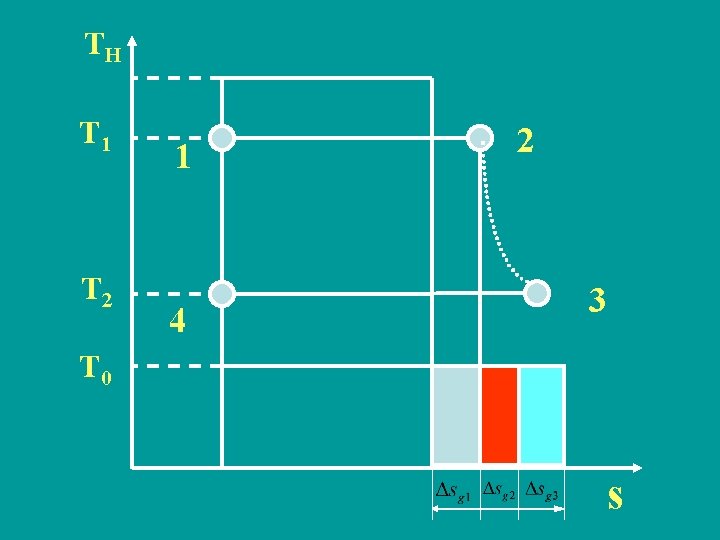
TH T 1 T 2 1 4 2 3 T 0 s

Solution: (1) The carnot cycle works between T 1 and T 2 Practical work of engine
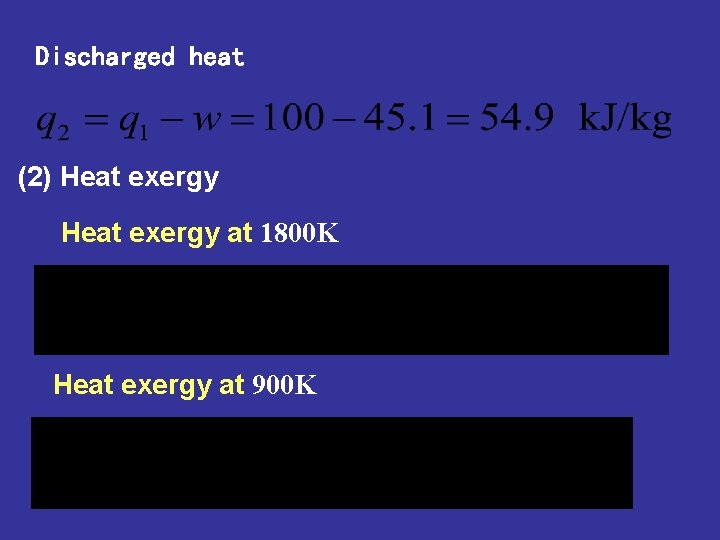
Discharged heat (2) Heat exergy at 1800 K Heat exergy at 900 K

Heat exergy at 320 K (3) Entropy production and exergy loss Entropy production caused by temperature difference in absorbing heat process Exergy loss or

Friction work loss Entropy production Exergy loss 0 r

Entropy production caused by temperature difference in discharging heat process Exergy loss 0 r

(4)Entropy increase of isolated system 0 r

The whole exergy loss 0 r