5 8 Preparation of Alkenes Elimination Reactions bElimination

5. 8 Preparation of Alkenes: Elimination Reactions

b-Elimination Reactions Overview dehydrogenation of alkanes: X=Y=H dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C b Ca Y C C + X Y
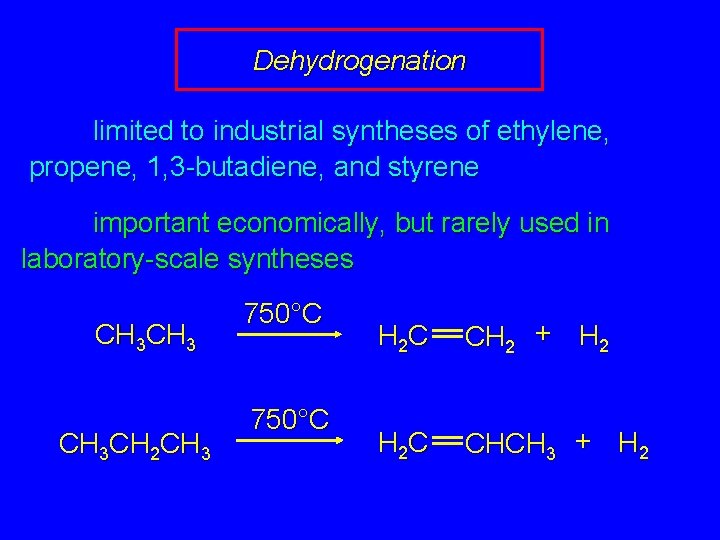
Dehydrogenation limited to industrial syntheses of ethylene, propene, 1, 3 -butadiene, and styrene important economically, but rarely used in laboratory-scale syntheses CH 3 CH 2 CH 3 750°C H 2 C CH 2 + H 2 C CHCH 3 + H 2

5. 9 Dehydration of Alcohols
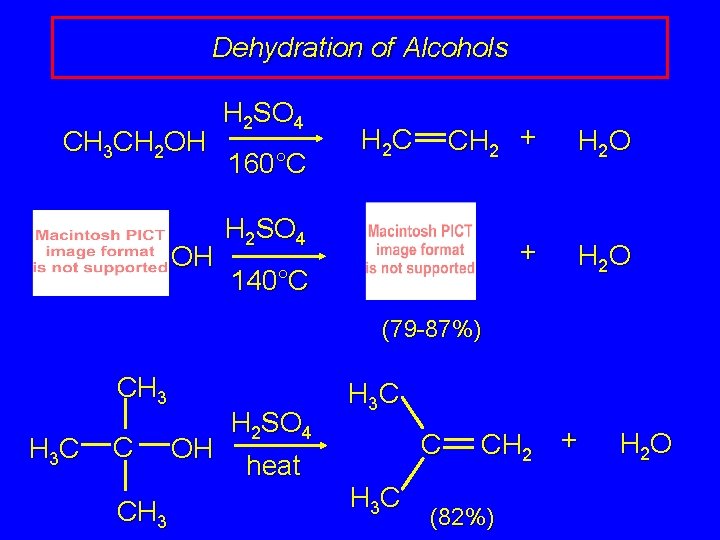
Dehydration of Alcohols CH 3 CH 2 OH OH H 2 SO 4 160°C H 2 C CH 2 + H 2 O H 2 SO 4 140°C (79 -87%) CH 3 H 3 C C CH 3 OH H 2 SO 4 heat H 3 C CH 2 (82%) + H 2 O
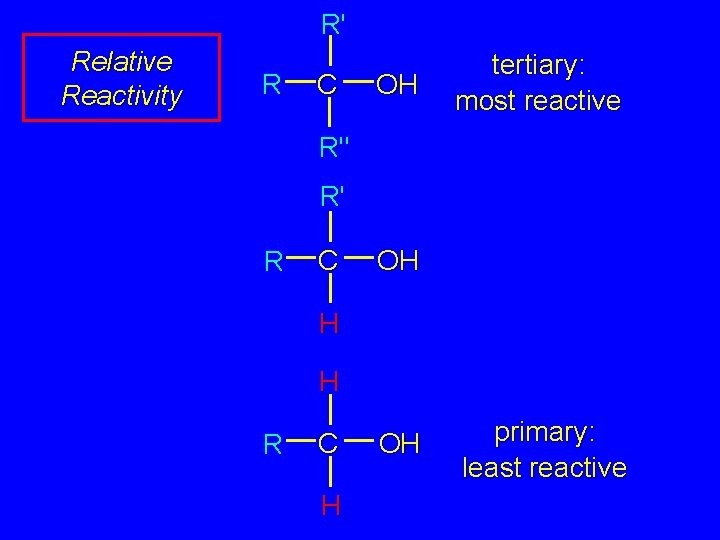
R' Relative Reactivity R C OH tertiary: most reactive R" R' R C OH H H R C H OH primary: least reactive

5. 10 Regioselectivity in Alcohol Dehydration: The Zaitsev Rule

Regioselectivity HO H 2 SO 4 + 80°C 10 % 90 % A reaction that can proceed in more than one direction, but in which one direction predominates, is said to be regioselective.

Regioselectivity CH 3 OH H 3 PO 4 CH 3 + heat 16 % 84 % A reaction that can proceed in more than one direction, but in which one direction predominates, is said to be regioselective.

The Zaitsev Rule When elimination can occur in more than one direction, the principal alkene is the one formed by loss of H from the b carbon having the fewest hydrogens. R R OH C C H CH 3 CH 2 R three protons on this b carbon
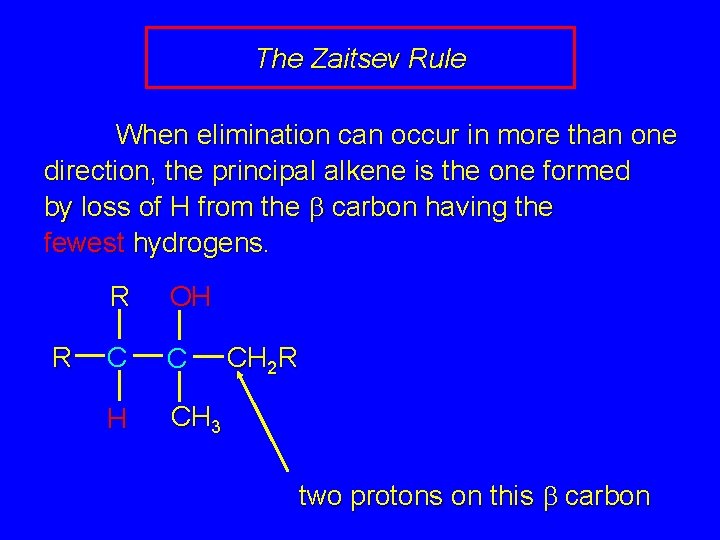
The Zaitsev Rule When elimination can occur in more than one direction, the principal alkene is the one formed by loss of H from the b carbon having the fewest hydrogens. R R OH C C H CH 3 CH 2 R two protons on this b carbon
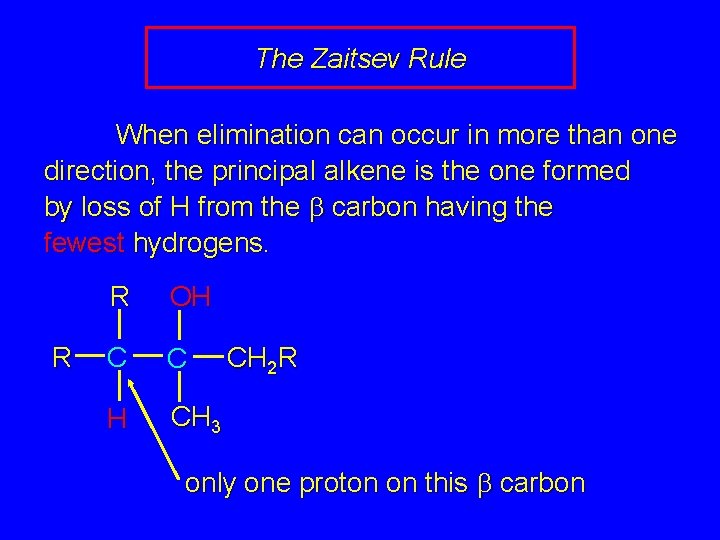
The Zaitsev Rule When elimination can occur in more than one direction, the principal alkene is the one formed by loss of H from the b carbon having the fewest hydrogens. R R OH C C H CH 3 CH 2 R only one proton on this b carbon
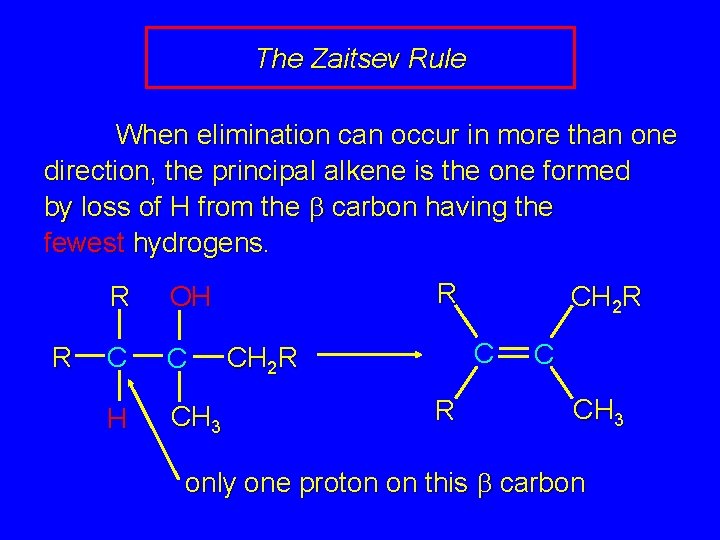
The Zaitsev Rule When elimination can occur in more than one direction, the principal alkene is the one formed by loss of H from the b carbon having the fewest hydrogens. R R OH C C H CH 3 R CH 2 R C CH 2 R R C CH 3 only one proton on this b carbon

5. 11 Stereoselectivity in Alcohol Dehydration

Stereoselectivity H 2 SO 4 + heat OH (25%) (75%) A stereoselective reaction is one in which a single starting material can yield two or more stereoisomeric products, but gives one of them in greater amounts than any other.

5. 12 The Mechanism of Acid-Catalyzed Dehydration of Alcohols

A connecting point. . . The dehydration of alcohols and the reaction of alcohols with hydrogen halides share the following common features: 1) Both reactions are promoted by acids 2) The relative reactivity decreases in the order tertiary > secondary > primary These similarities suggest that carbocations are intermediates in the acid-catalyzed dehydration of alcohols, just as they are in the reaction of alcohols with hydrogen halides.
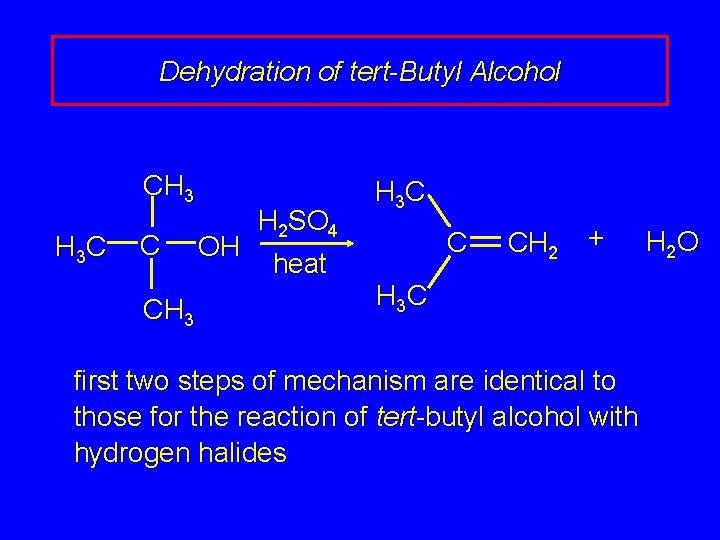
Dehydration of tert-Butyl Alcohol CH 3 H 3 C C CH 3 OH H 2 SO 4 heat H 3 C C CH 2 + H 3 C first two steps of mechanism are identical to those for the reaction of tert-butyl alcohol with hydrogen halides H 2 O
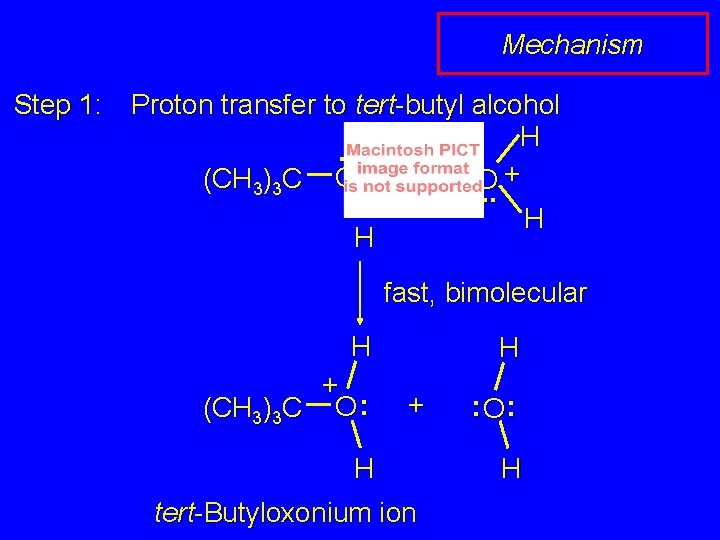
Mechanism Step 1: Proton transfer to tert-butyl alcohol H. . + (CH 3)3 C O : + H O. . H H fast, bimolecular H + (CH 3)3 C O : H + H tert-Butyloxonium ion : O: H
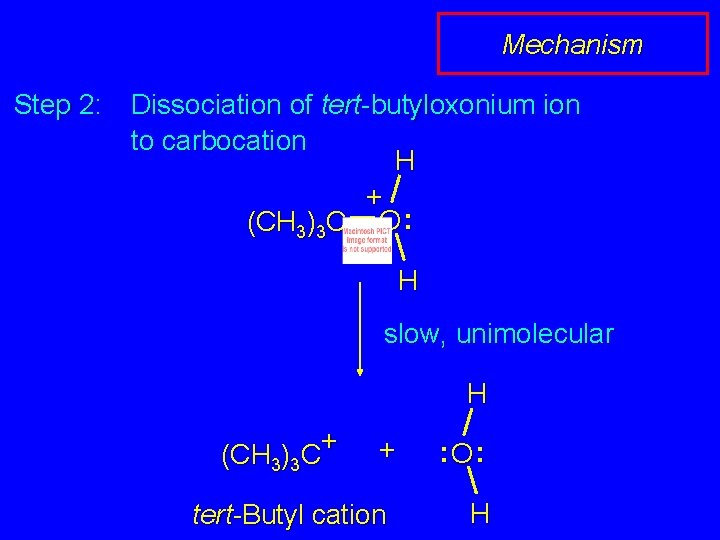
Mechanism Step 2: Dissociation of tert-butyloxonium ion to carbocation H + (CH 3)3 C O : H slow, unimolecular H + (CH 3)3 C + tert-Butyl cation : O: H
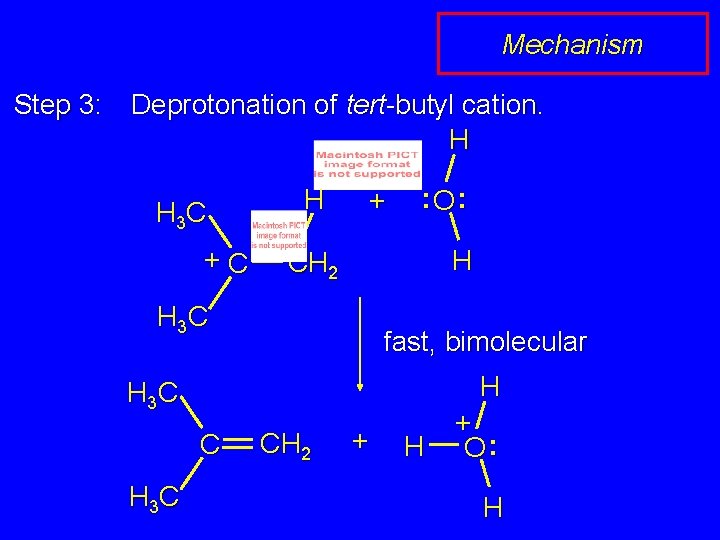
Mechanism Step 3: Deprotonation of tert-butyl cation. H H 3 C +C H + : O: H CH 2 H 3 C fast, bimolecular H H 3 C CH 2 + H + O: H

Carbocations are intermediates in the acid-catalyzed dehydration of tertiary and secondary alcohols carbocations can: react with nucleophiles lose a b-proton to form an alkene
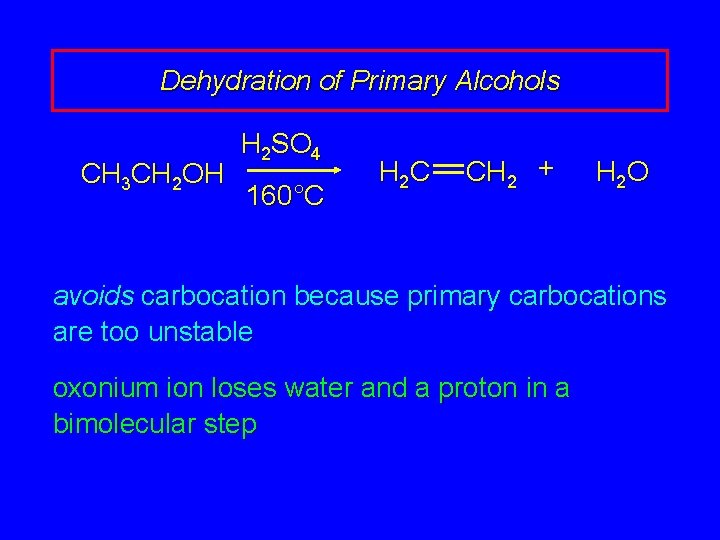
Dehydration of Primary Alcohols CH 3 CH 2 OH H 2 SO 4 160°C H 2 C CH 2 + H 2 O avoids carbocation because primary carbocations are too unstable oxonium ion loses water and a proton in a bimolecular step
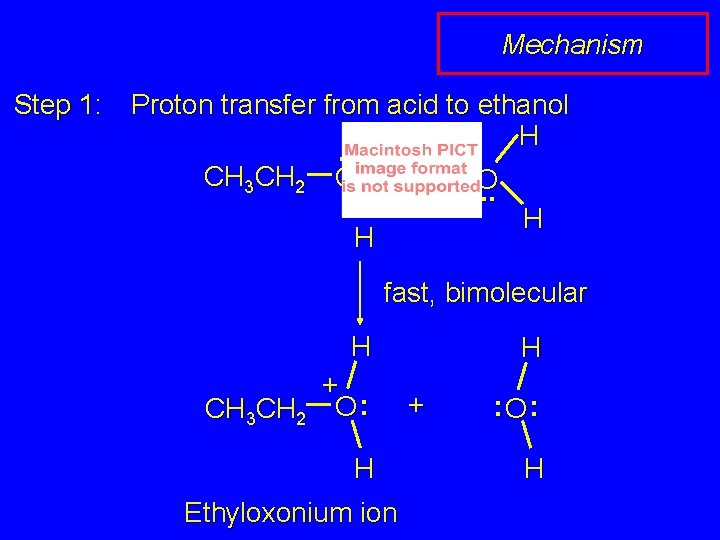
Mechanism Step 1: Proton transfer from acid to ethanol H. . CH 3 CH 2 O : + H O. . H H fast, bimolecular H + CH 3 CH 2 O : H Ethyloxonium ion H + : O: H
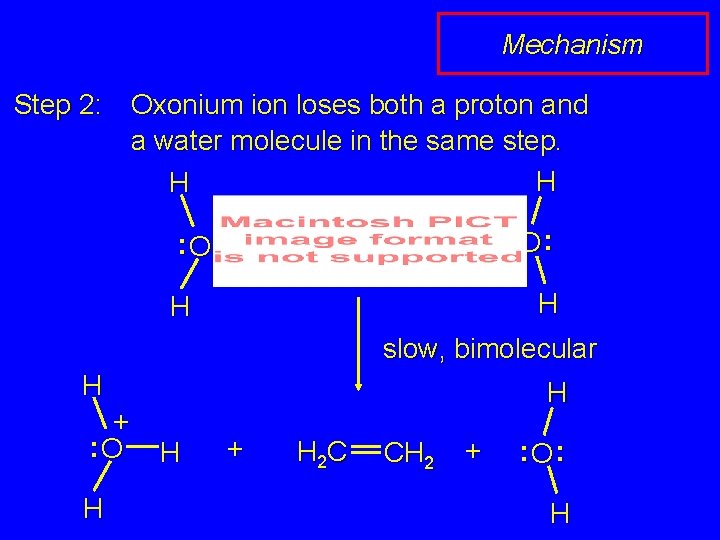
Mechanism Step 2: Oxonium ion loses both a proton and a water molecule in the same step. H H + : O : + H CH 2 O : H H slow, bimolecular H + : O H H H + H 2 C CH 2 + : O: H

5. 13 Rearrangements in Alcohol Dehydration Sometimes the alkene product does not have the same carbon skeleton as the starting alcohol.
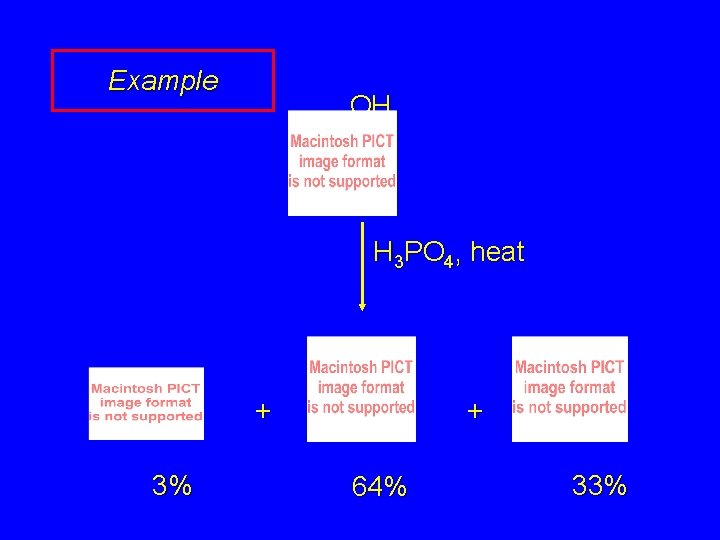
Example OH H 3 PO 4, heat + 3% + 64% 33%
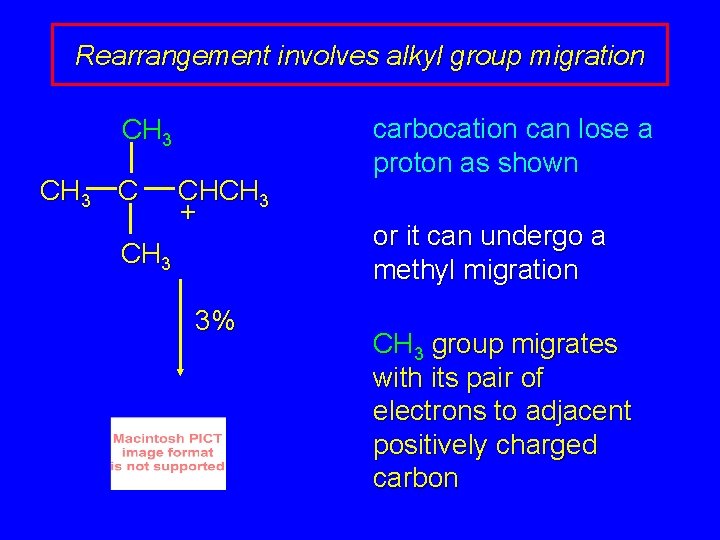
Rearrangement involves alkyl group migration CH 3 C CHCH 3 + CH 3 3% carbocation can lose a proton as shown or it can undergo a methyl migration CH 3 group migrates with its pair of electrons to adjacent positively charged carbon
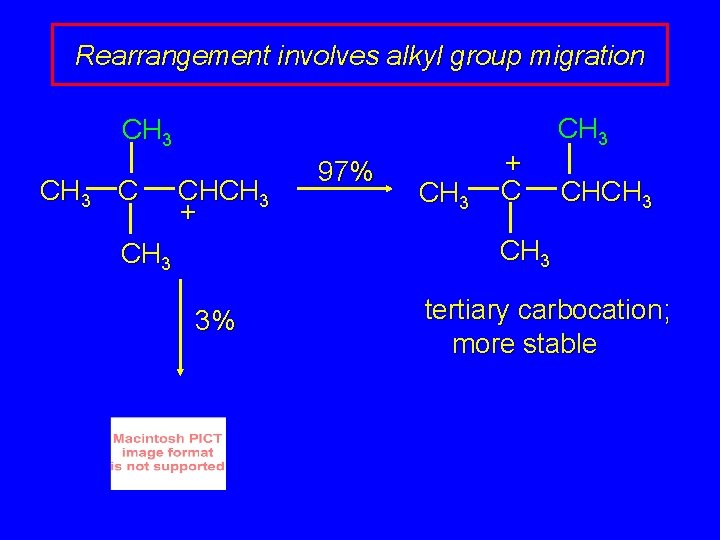
Rearrangement involves alkyl group migration CH 3 C CHCH 3 + 97% CH 3 + C CH 3 CH 3 3% tertiary carbocation; more stable
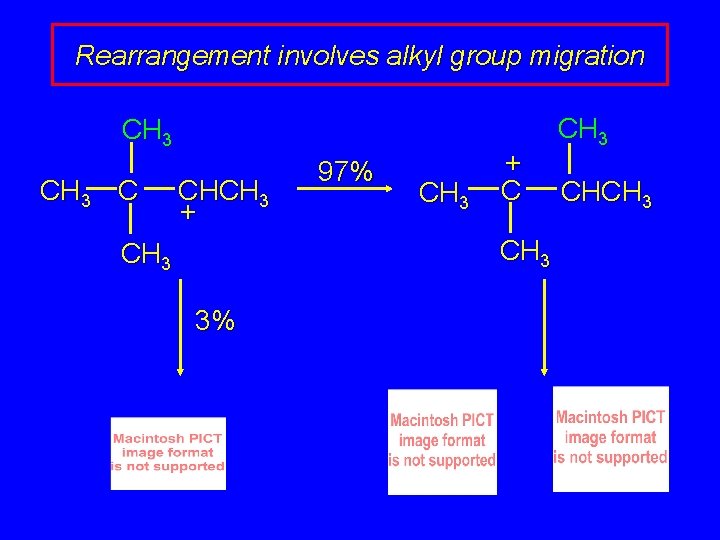
Rearrangement involves alkyl group migration CH 3 C CHCH 3 + 97% CH 3 + C CH 3 3% CH 3 CHCH 3
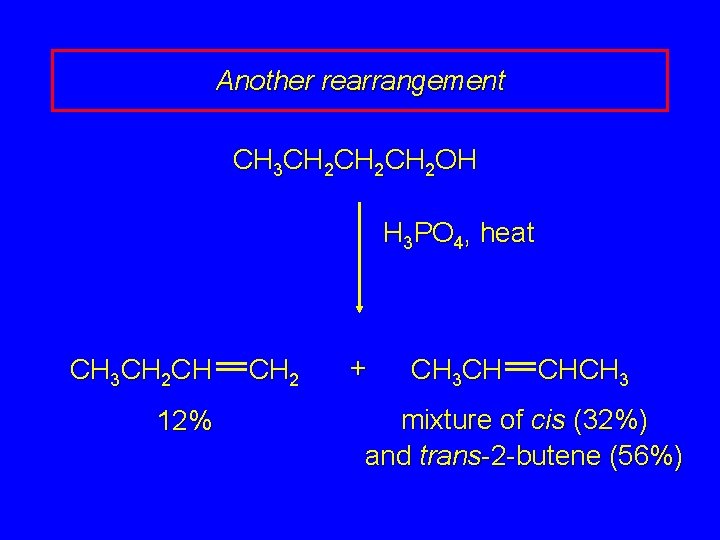
Another rearrangement CH 3 CH 2 CH 2 OH H 3 PO 4, heat CH 3 CH 2 CH 12% CH 2 + CH 3 CH CHCH 3 mixture of cis (32%) and trans-2 -butene (56%)
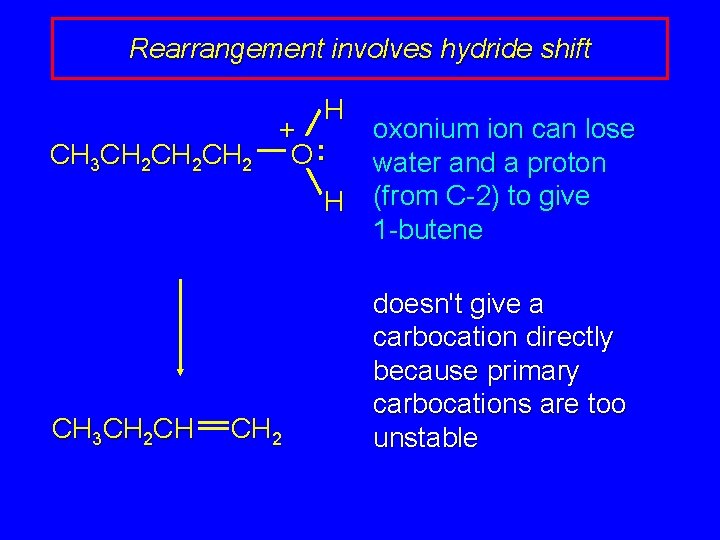
Rearrangement involves hydride shift CH 3 CH 2 CH 2 CH 3 CH 2 CH H oxonium ion can lose + O: water and a proton H (from C-2) to give 1 -butene CH 2 doesn't give a carbocation directly because primary carbocations are too unstable
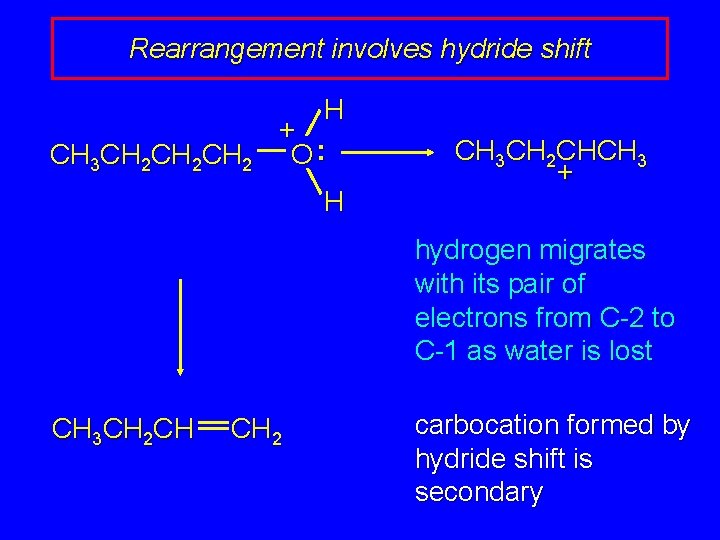
Rearrangement involves hydride shift CH 3 CH 2 CH 2 H + O: H CH 3 CH 2 CHCH 3 + hydrogen migrates with its pair of electrons from C-2 to C-1 as water is lost CH 3 CH 2 CH CH 2 carbocation formed by hydride shift is secondary
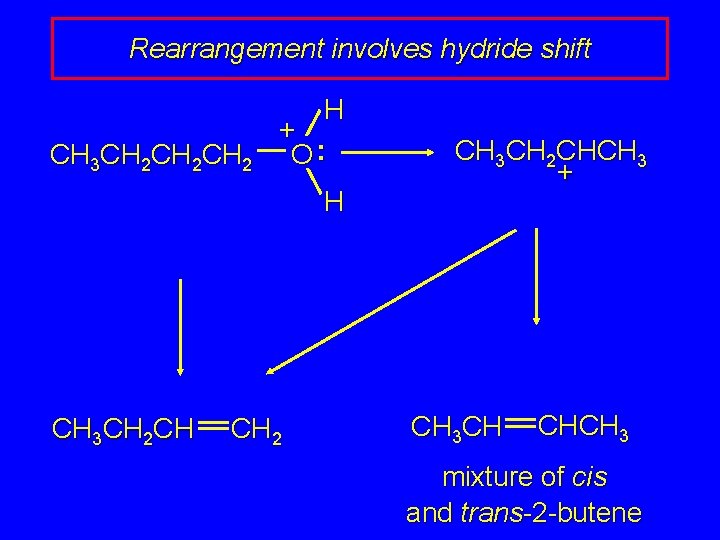
Rearrangement involves hydride shift CH 3 CH 2 CH 2 H + O: H CH 3 CH 2 CH CH 2 CH 3 CH 2 CHCH 3 + CH 3 CH CHCH 3 mixture of cis and trans-2 -butene
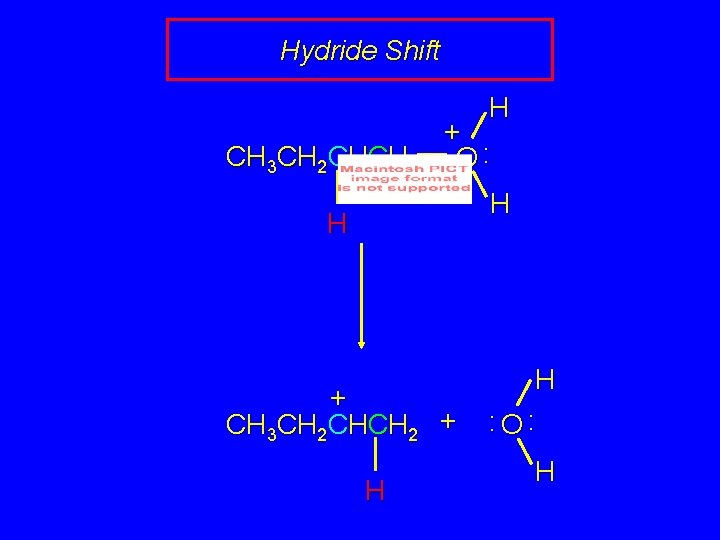
Hydride Shift H CH 3 CH 2 CHCH 2 + O: H H + CH 3 CH 2 CHCH 2 + H H : O: H

Carbocations can. . . • react with nucleophiles • lose a proton from the b-carbon to form an alkene • rearrange (less stable to more stable)
- Slides: 36