5 5 Phase diagrams of threecomponent systems ternary



- Slides: 30

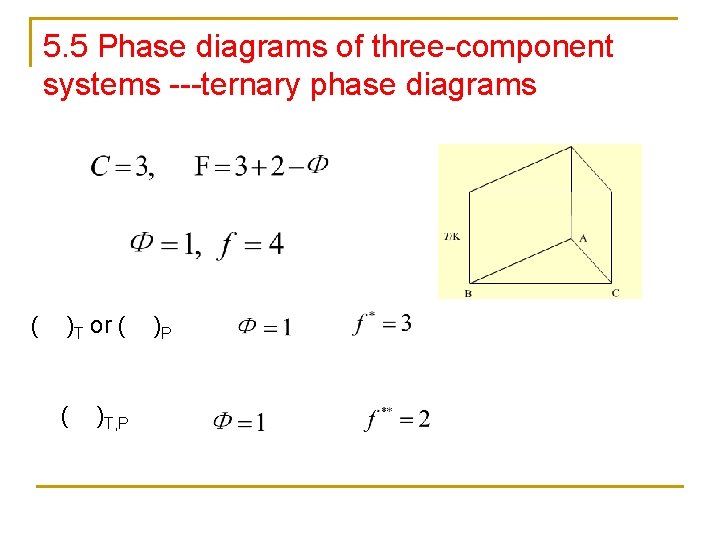
5. 5 Phase diagrams of three-component systems ---ternary phase diagrams ( )T or ( ( )T, P )P

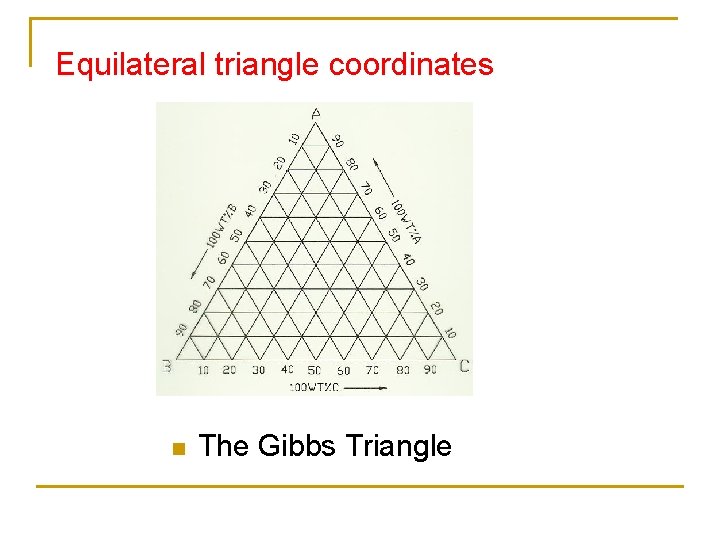
Equilateral triangle coordinates n The Gibbs Triangle

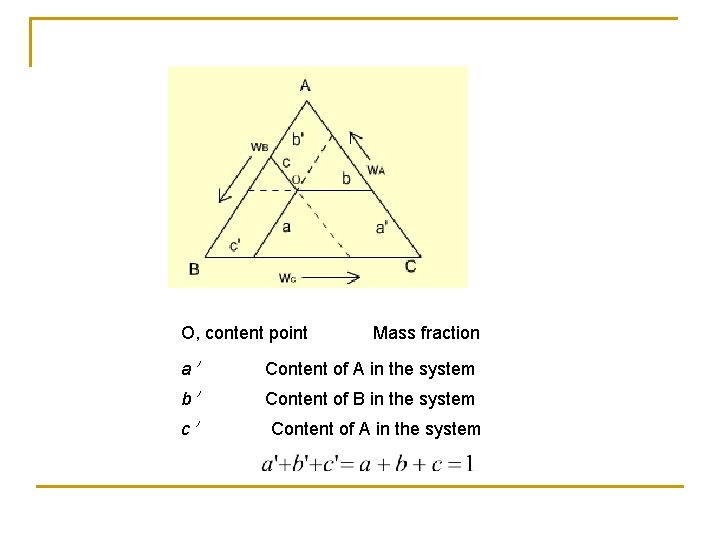
O, content point Mass fraction a' Content of A in the system b' Content of B in the system c' Content of A in the system

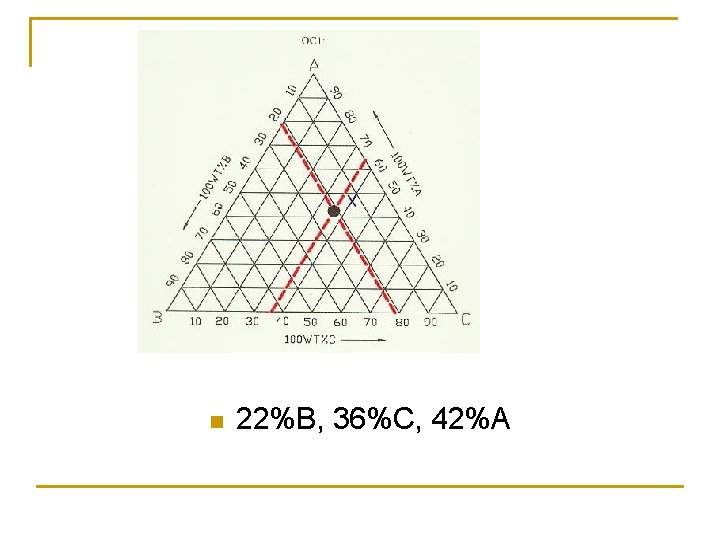
n 22%B, 36%C, 42%A

5. 5. 1 Composition characters for equilateral triangle coordinates d, e, f the mass fractions of A are equal Points on AD line, On line AD, D’ is closer to A than D, the mass fraction of A in D’ is more than that in D

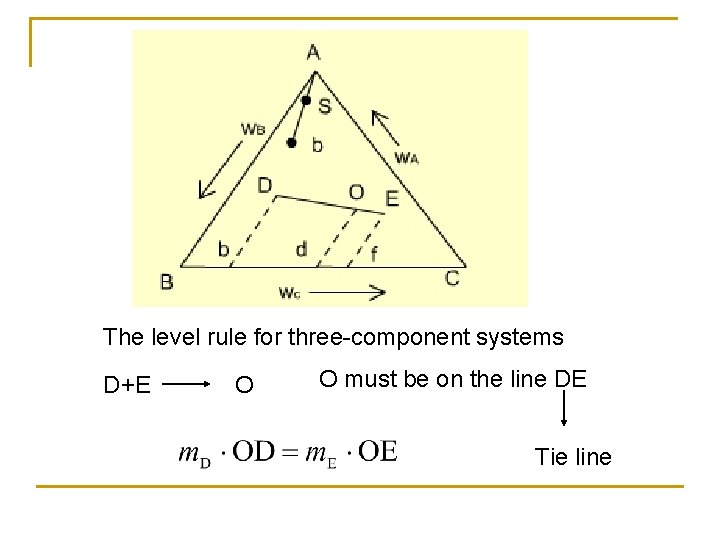
The level rule for three-component systems D+E O O must be on the line DE Tie line

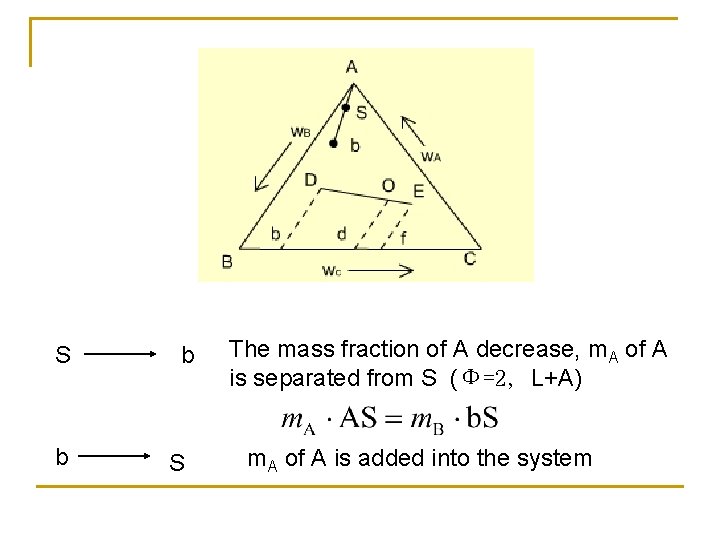
S b b S The mass fraction of A decrease, m. A of A is separated from S (Φ=2, L+A) m. A of A is added into the system

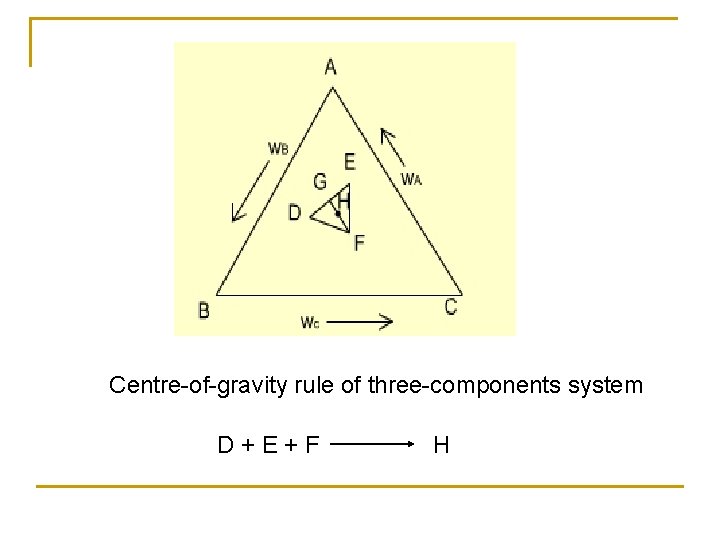
Centre-of-gravity rule of three-components system D+E+F H

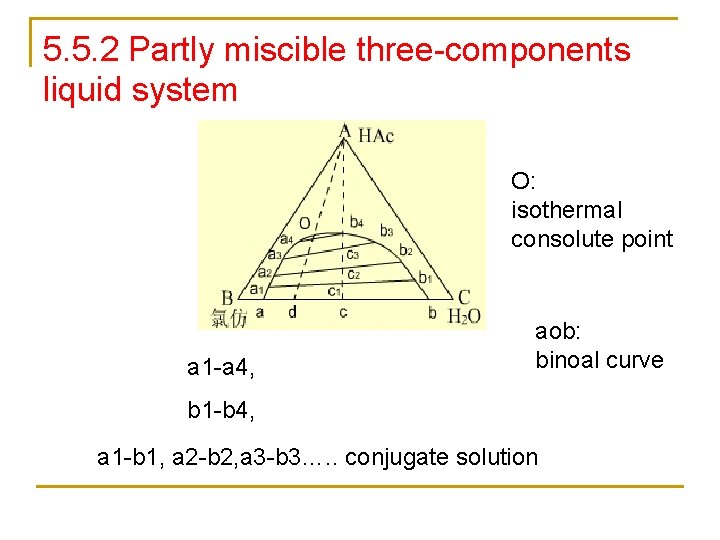
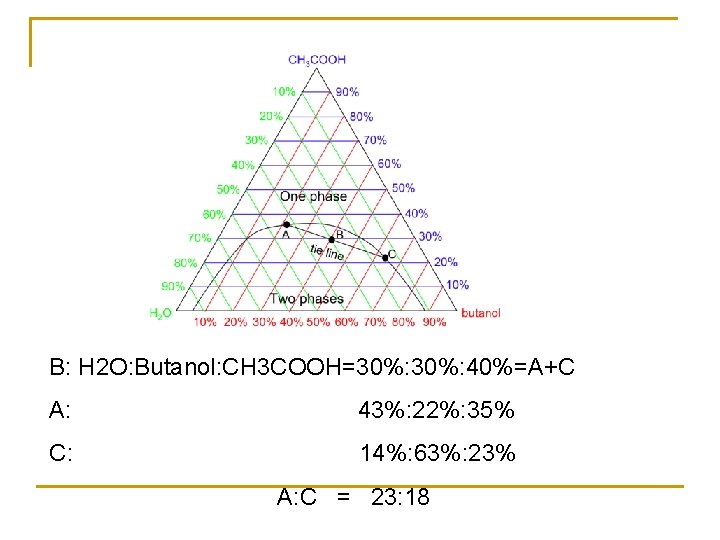
5. 5. 2 Partly miscible three-components liquid system O: isothermal consolute point a 1 -a 4, aob: binoal curve b 1 -b 4, a 1 -b 1, a 2 -b 2, a 3 -b 3…. . conjugate solution

B: H 2 O: Butanol: CH 3 COOH=30%: 40%=A+C A: 43%: 22%: 35% C: 14%: 63%: 23% A: C = 23: 18

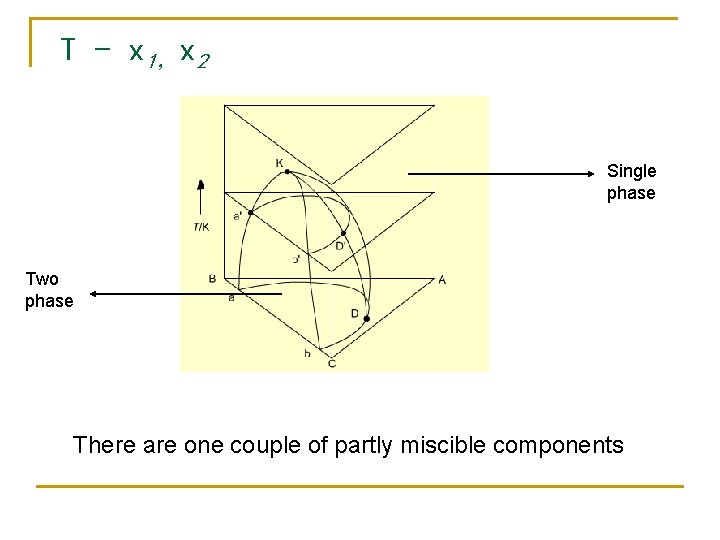
T - x 1,x 2 Single phase Two phase There are one couple of partly miscible components

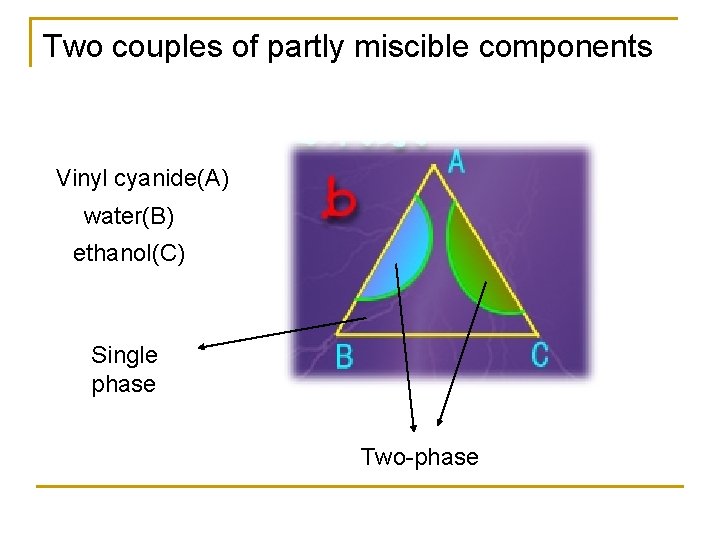
Two couples of partly miscible components Vinyl cyanide(A) water(B) ethanol(C) Single phase Two-phase

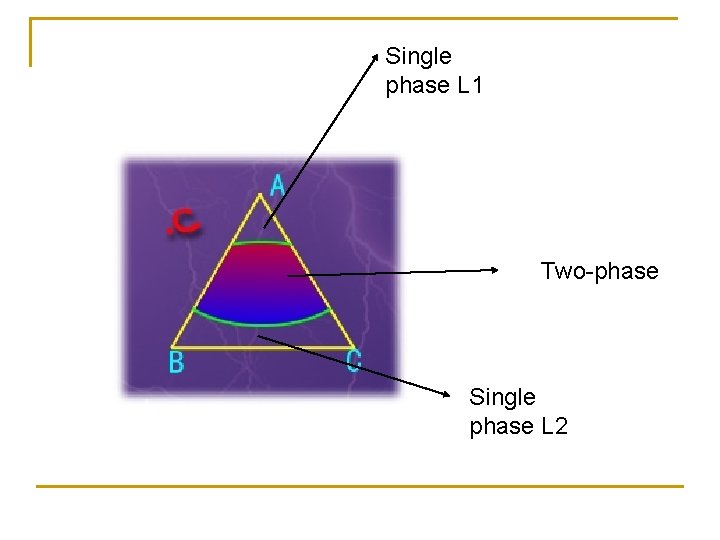
Single phase L 1 Two-phase Single phase L 2

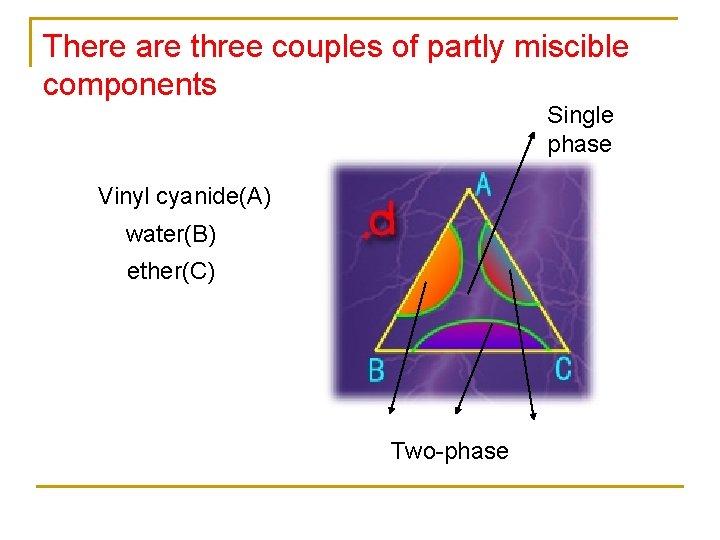
There are three couples of partly miscible components Single phase Vinyl cyanide(A) water(B) ether(C) Two-phase

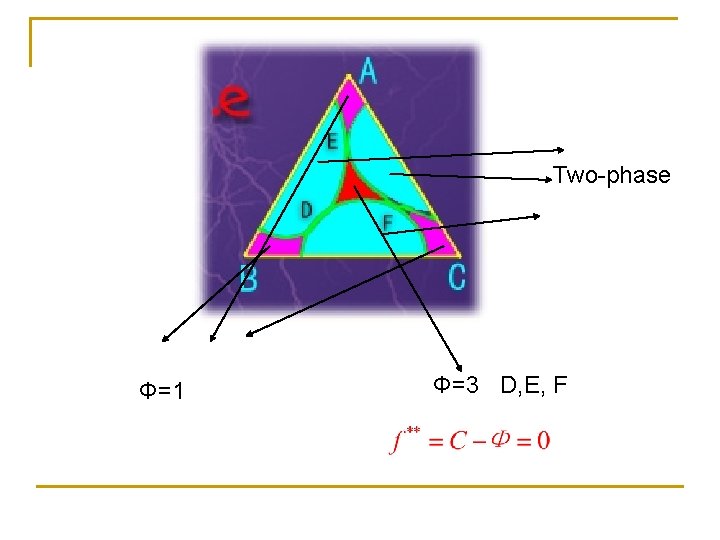
Two-phase Φ=1 Φ=3 D, E, F

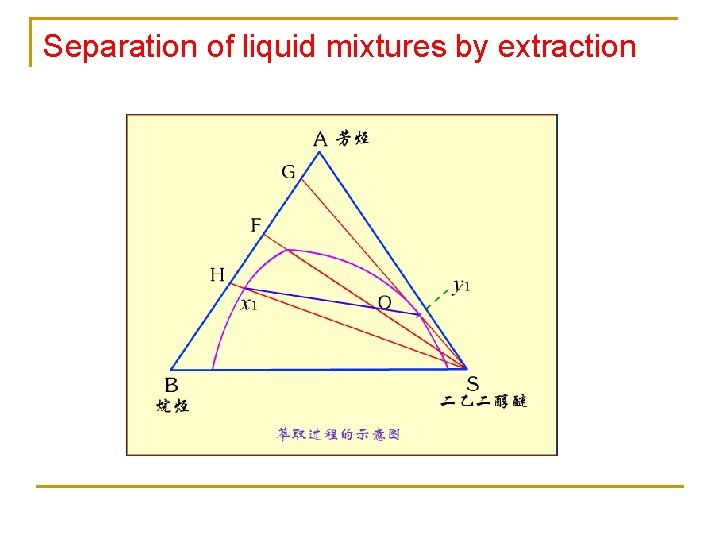
Separation of liquid mixtures by extraction

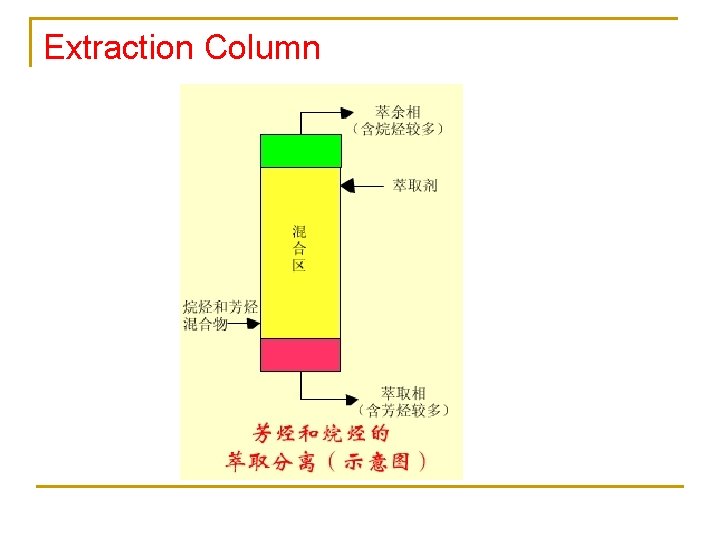
Extraction Column

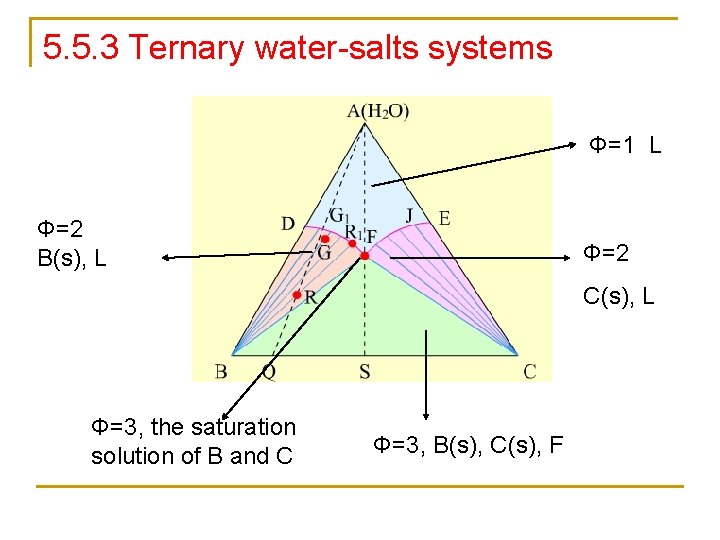
5. 5. 3 Ternary water-salts systems Φ=1 L Φ=2 B(s), L Φ=2 C(s), L Φ=3, the saturation solution of B and C Φ=3, B(s), C(s), F

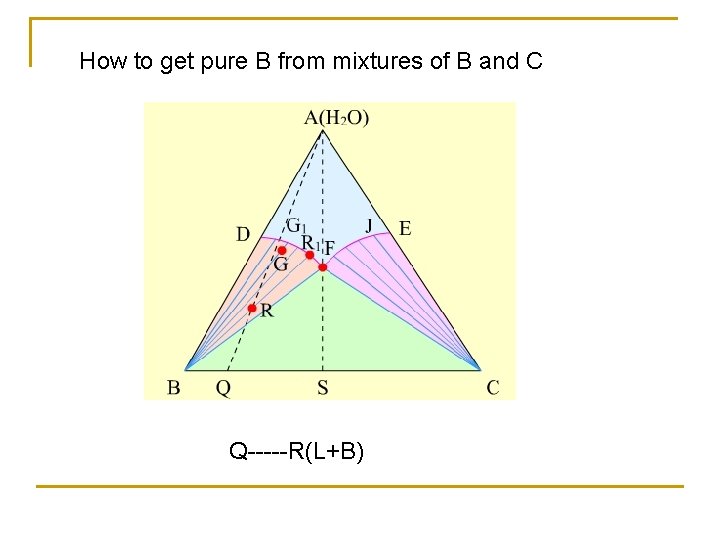
How to get pure B from mixtures of B and C Q-----R(L+B)

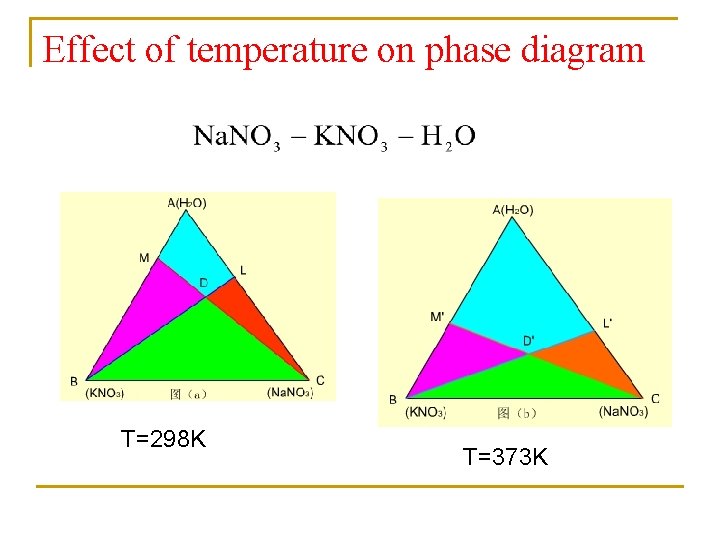
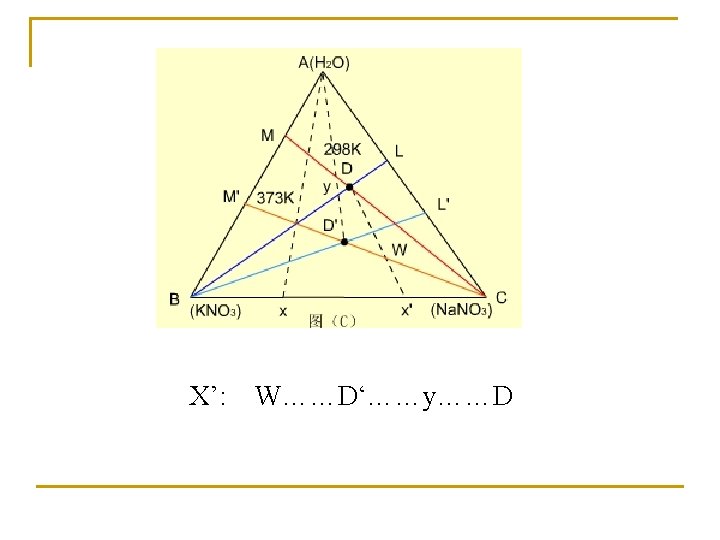
Effect of temperature on phase diagram T=298 K T=373 K

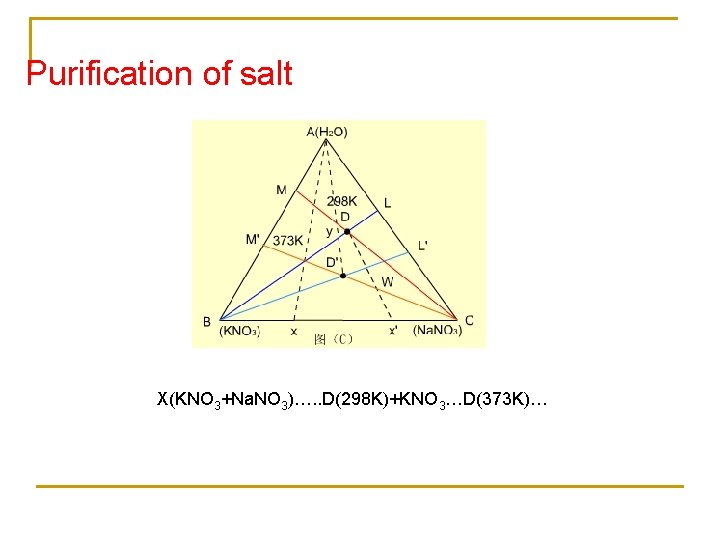
Purification of salt X(KNO 3+Na. NO 3)…. . D(298 K)+KNO 3…D(373 K)…


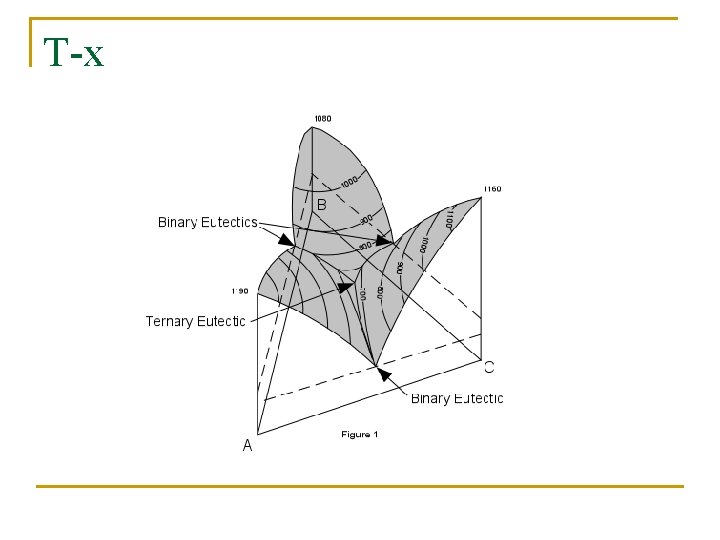
5. 5. 4 Three-component system containing three binary eutectic points

T-x

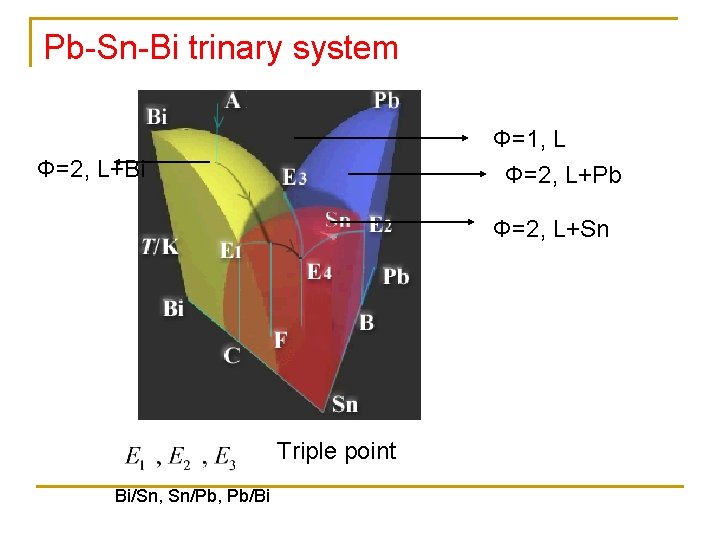
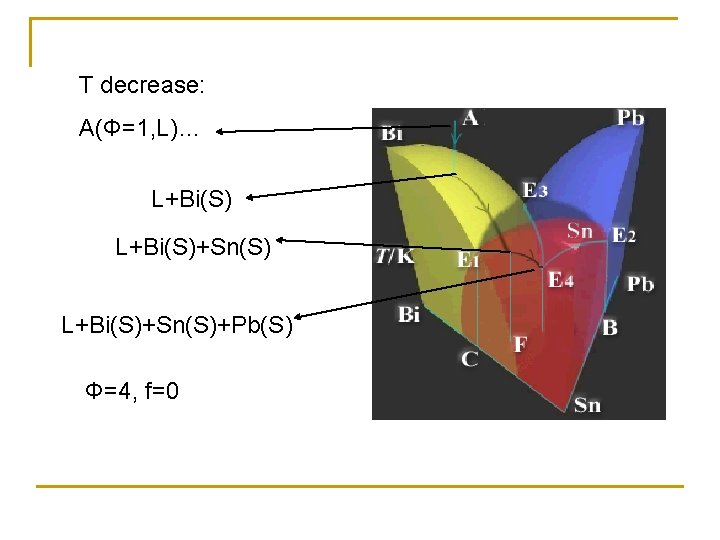
Pb-Sn-Bi trinary system Φ=1, L Φ=2, L+Bi Φ=2, L+Pb Φ=2, L+Sn Triple point Bi/Sn, Sn/Pb, Pb/Bi

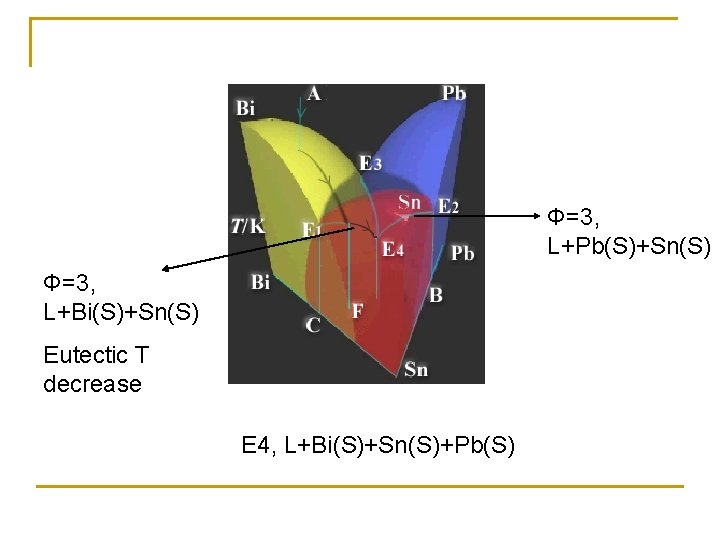
Φ=3, L+Pb(S)+Sn(S) Φ=3, L+Bi(S)+Sn(S) Eutectic T decrease E 4, L+Bi(S)+Sn(S)+Pb(S)

T decrease: A(Φ=1, L)… L+Bi(S)+Sn(S)+Pb(S) Φ=4, f=0

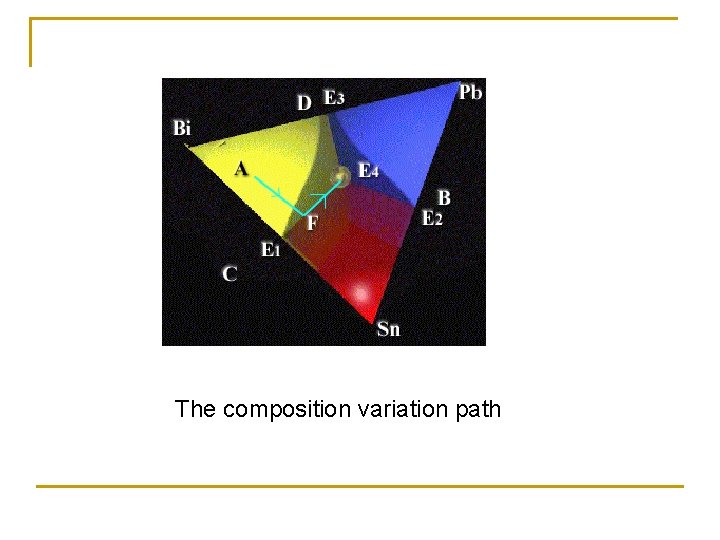
The composition variation path

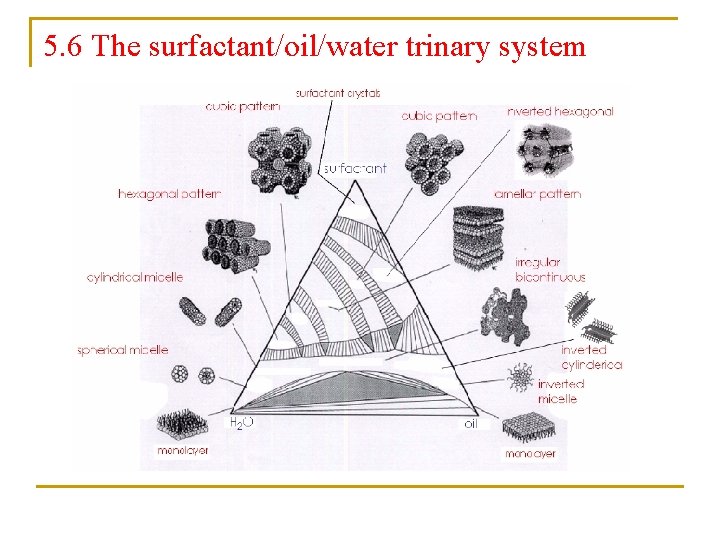
5. 6 The surfactant/oil/water trinary system

Homework n Y: P 188 39 n Review the whole chapter