5 5 Covalent Bonding and Molecules When compounds

5. 5 Covalent Bonding and Molecules When compounds form between elements with similar properties, electrons are not transferred from one element to another but instead are shared to give each atom a noble gas configuration. This approach is known as the Lewis theory of bonding, named for it’s proponent, Gilbert Lewis theory depicts bond formation in H 2 as H∙ + ∙H → H: H This type of arrangement, where two atoms share a pair of electrons, is known as covalent bonding, and the shared pair of electrons constitutes a covalent bond. • https: //phet. colorado. edu/en/simulation/atomic-interactions • http: //mw. concord. org/modeler/showcase/simulation. html? s=http: //mw 2. concord. o rg/public/part 1/electroncloud/index. html
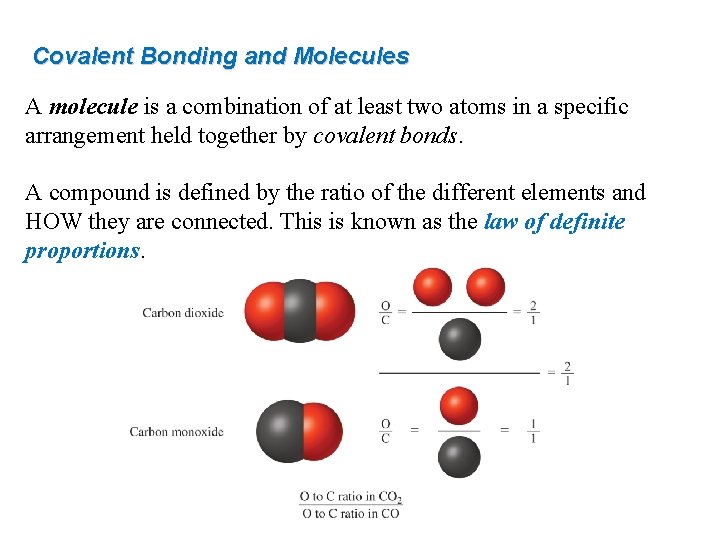
Covalent Bonding and Molecules A molecule is a combination of at least two atoms in a specific arrangement held together by covalent bonds. A compound is defined by the ratio of the different elements and HOW they are connected. This is known as the law of definite proportions.

Covalent Bonding and Molecules Diatomic molecules contain two atoms and may be either heteronuclear or homonuclear. Polyatomic molecules contain more than two atoms.

Covalent Bonding and Molecules
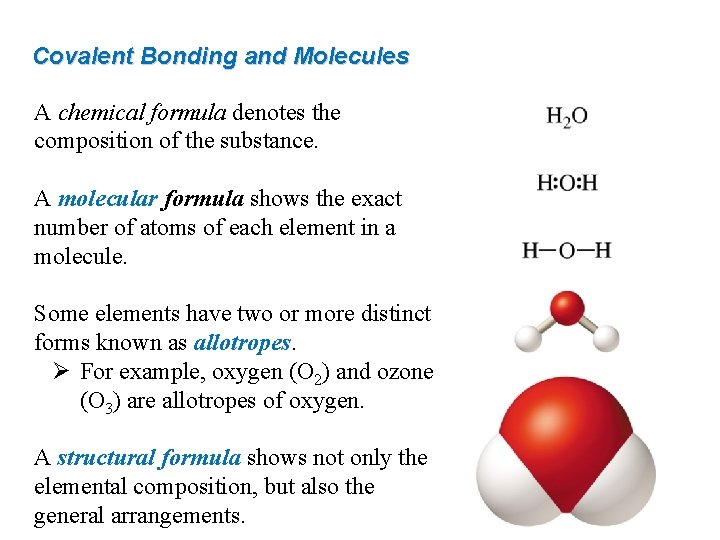
Covalent Bonding and Molecules A chemical formula denotes the composition of the substance. A molecular formula shows the exact number of atoms of each element in a molecule. Some elements have two or more distinct forms known as allotropes. Ø For example, oxygen (O 2) and ozone (O 3) are allotropes of oxygen. A structural formula shows not only the elemental composition, but also the general arrangements.

Worked Example 5. 5 Write the molecular formula of ethanol based on its ball-and-stick model, shown here. Strategy Refer to the labels on the atoms (or see Table 5. 3). There are two carbon atoms, six hydrogen atoms, and one oxygen atom, so the subscript on C will be 2 and the subscript on H will be 6, and there will be no subscript on O. Test Practice: 5. 40 5. 42 Activity: Which elements on the periodic table are most likely to form covalent bonds versus ionic bonds? Molecules are usually thought of as too small to see with the naked eye. Give some exceptions to this idea.

Chemistry: Atoms First Second Edition Julia Burdge & Jason Overby Chapter 6 Representing Molecules M. Stacey Thomson Pasco-Hernando State College Copyright (c) The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.
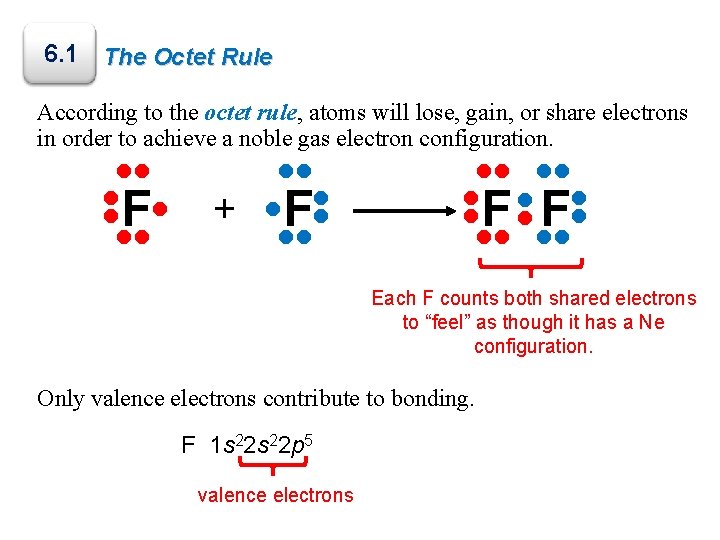
6. 1 The Octet Rule According to the octet rule, atoms will lose, gain, or share electrons in order to achieve a noble gas electron configuration. • • F • • • • + • • • • F • Each F counts both shared electrons to “feel” as though it has a Ne configuration. Only valence electrons contribute to bonding. F 1 s 22 p 5 valence electrons
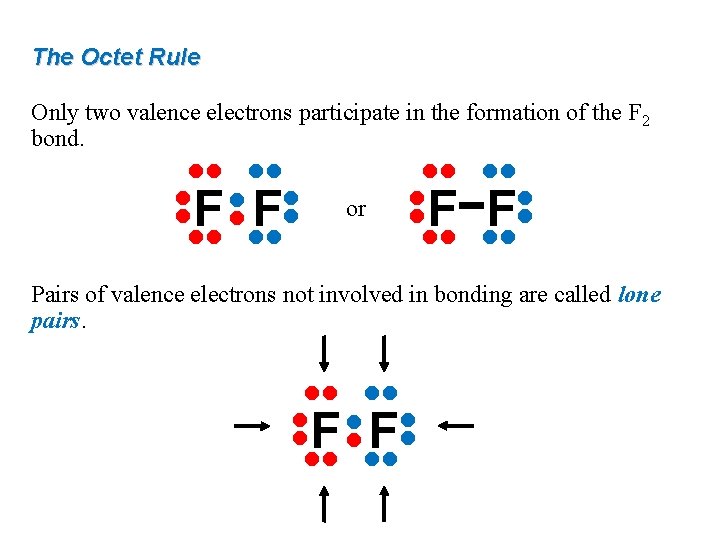
The Octet Rule Only two valence electrons participate in the formation of the F 2 bond. • • or • • F F • • • • F • • Pairs of valence electrons not involved in bonding are called lone pairs. • • • • F • •
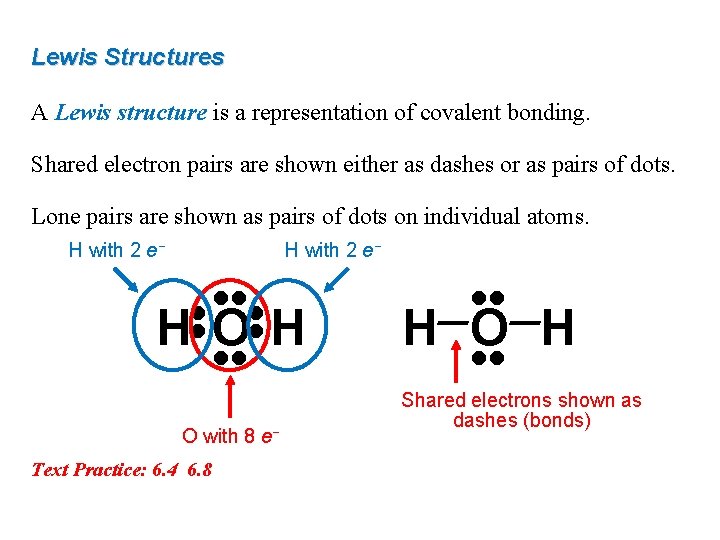
Lewis Structures A Lewis structure is a representation of covalent bonding. Shared electron pairs are shown either as dashes or as pairs of dots. Lone pairs are shown as pairs of dots on individual atoms. H with 2 e− • • HOH • • O with 8 e− Text Practice: 6. 4 6. 8 • • H O H • • Shared electrons shown as dashes (bonds)
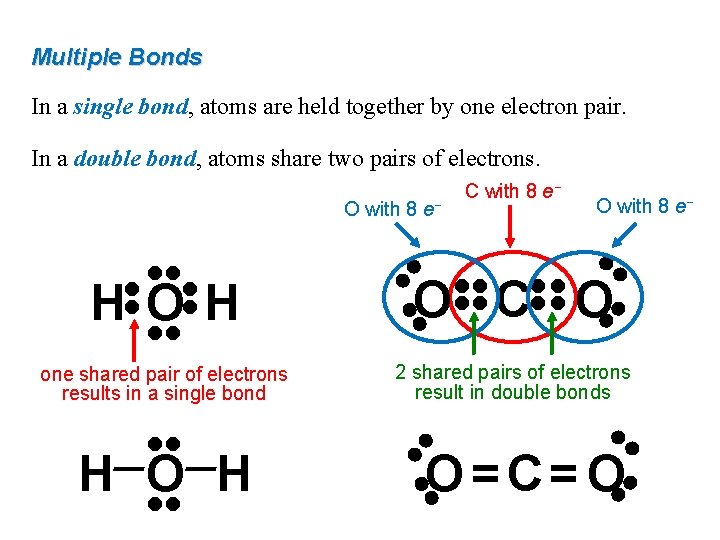
Multiple Bonds In a single bond, atoms are held together by one electron pair. In a double bond, atoms share two pairs of electrons. • • 2 shared pairs of electrons result in double bonds • • O=C=O • • H O H • • O C O • • one shared pair of electrons results in a single bond • • • • HOH • • O with 8 e− C with 8 e−
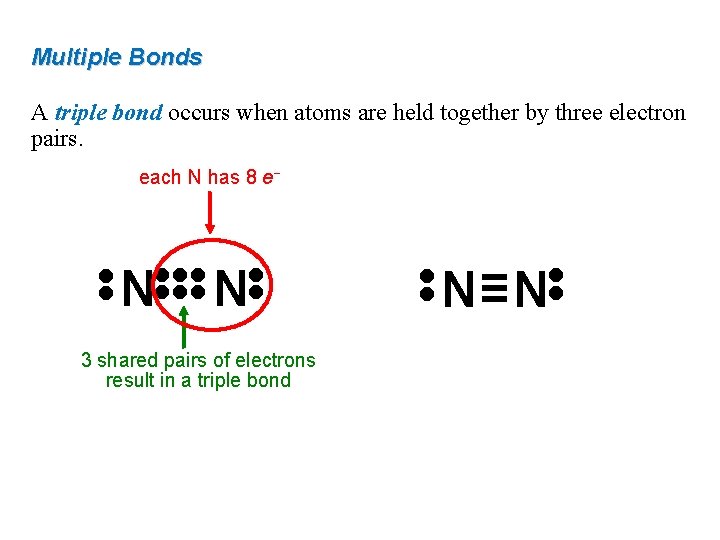
Multiple Bonds A triple bond occurs when atoms are held together by three electron pairs. each N has 8 e− • • 3 shared pairs of electrons result in a triple bond N≡N • • • • N

Multiple Bonds Bond length is defined as the distance between the nuclei of two covalently bonded atoms.
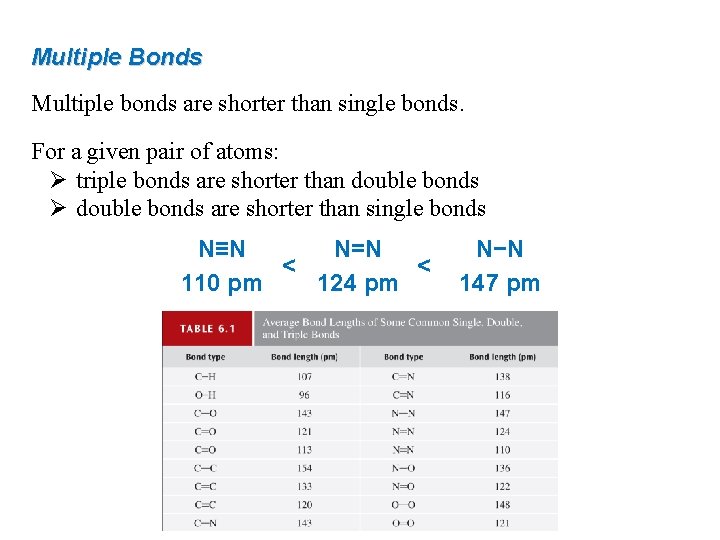
Multiple Bonds Multiple bonds are shorter than single bonds. For a given pair of atoms: Ø triple bonds are shorter than double bonds Ø double bonds are shorter than single bonds N≡N N=N < < 110 pm 124 pm N−N 147 pm

Multiple Bonds The shorter multiple bonds are also stronger than single bonds. We quantify bond strength by measuring the quantity of energy required to break it. H 2(g) → H(g) + H(g) Bond energy = 436. 4 k. J/mol We will examine this in more detail in Chapter 10.

6. 2 Electronegativity is the tendency of an atom to attract electrons it is SHARING with another atom. Electronegative – electron greedy Will the more electronegative atom be more or less likely to be found as a central atom in a molecule (covalent compound involving shared electrons)?
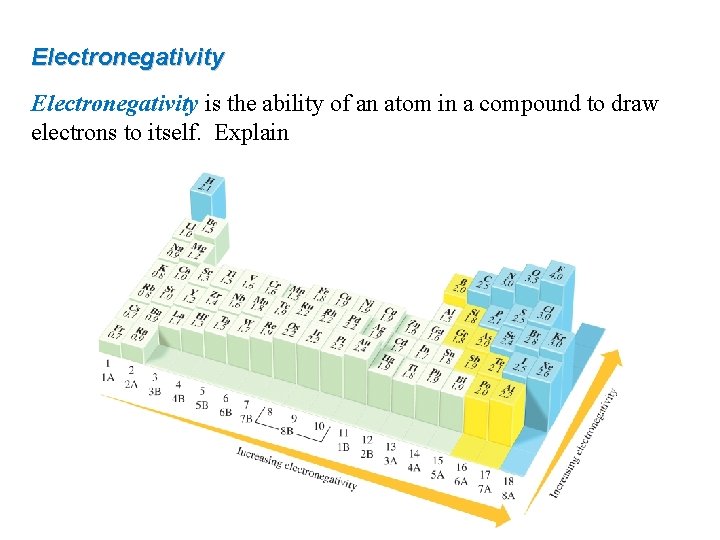
Electronegativity is the ability of an atom in a compound to draw electrons to itself. Explain
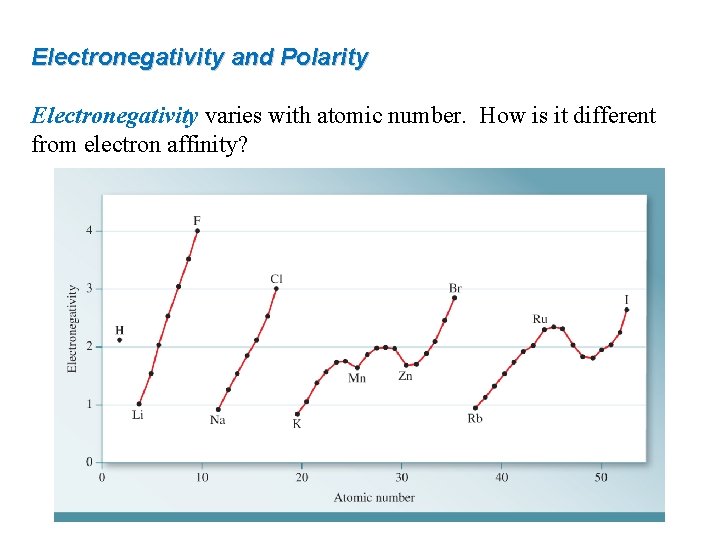
Electronegativity and Polarity Electronegativity varies with atomic number. How is it different from electron affinity?

6. 3 Drawing Lewis Structures Follow these steps when drawing Lewis structure for molecules and polyatomic ions. 1) Draw the skeletal structure of the compound. The least electronegative atom is usually the central atom. Draw a single covalent bond between the central atom and each of the surrounding atoms. 2) Count the total number of valence electrons present; add electrons for negative charges and subtract electrons for positive charges. 3) For each bond in the skeletal structure, subtract two electrons from the total valence electrons. 4) Use the remaining electrons to complete octets of the terminal atoms by placing pairs of electrons on each atom. Complete the octets of the most electronegative atom first. 5) Place any remaining electrons in pairs on the central atom. 6) If the central atom has fewer than eight electrons, move one or more pairs from the terminal atoms to form multiple bonds between the central atom and terminal atoms.
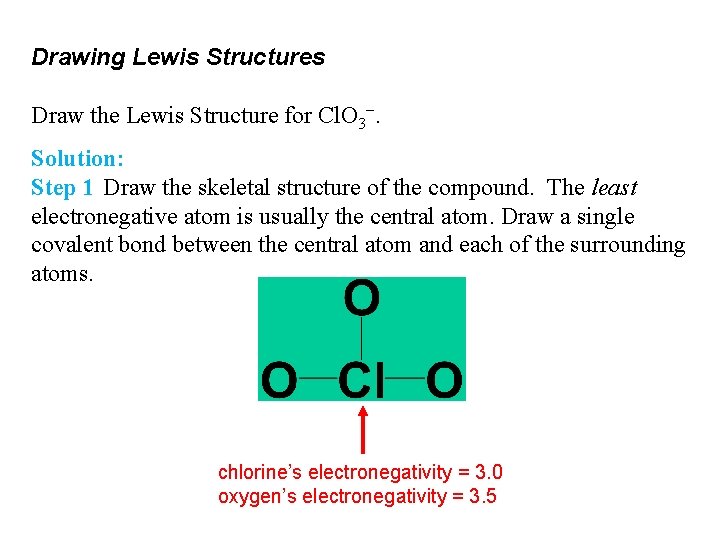
Drawing Lewis Structures Draw the Lewis Structure for Cl. O 3−. Solution: Step 1 Draw the skeletal structure of the compound. The least electronegative atom is usually the central atom. Draw a single covalent bond between the central atom and each of the surrounding atoms. chlorine’s electronegativity = 3. 0 oxygen’s electronegativity = 3. 5

Drawing Lewis Structures Draw the Lewis Structure for Cl. O 3−. Solution: Step 2 Count the total number of valence electrons present; add electrons for negative charges and subtract electrons for positive charges. Cl: 1 x 7 = 7 e− O: 3 x 6 = 18 e− Charge: 1 e− Total: 26 valence e−
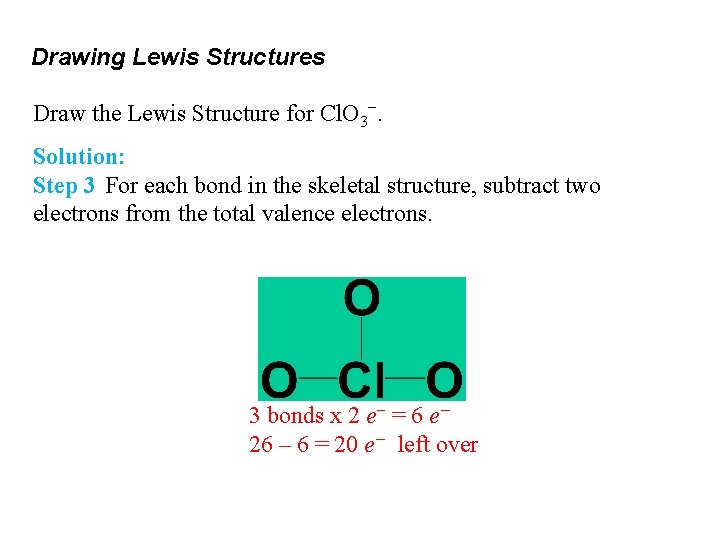
Drawing Lewis Structures Draw the Lewis Structure for Cl. O 3−. Solution: Step 3 For each bond in the skeletal structure, subtract two electrons from the total valence electrons. 3 bonds x 2 e− = 6 e− 26 – 6 = 20 e− left over

Drawing Lewis Structures Draw the Lewis Structure for Cl. O 3−. Solution: Step 4 Use the remaining electrons to complete octets of the terminal atoms by placing pairs of electrons on each atom. Complete the octets of the most electronegative atom first. 6 electrons used in single bonds 20 electrons must be placed in the structure starting with terminal atoms The structure now has a total of 24 valence electrons of the available 26.
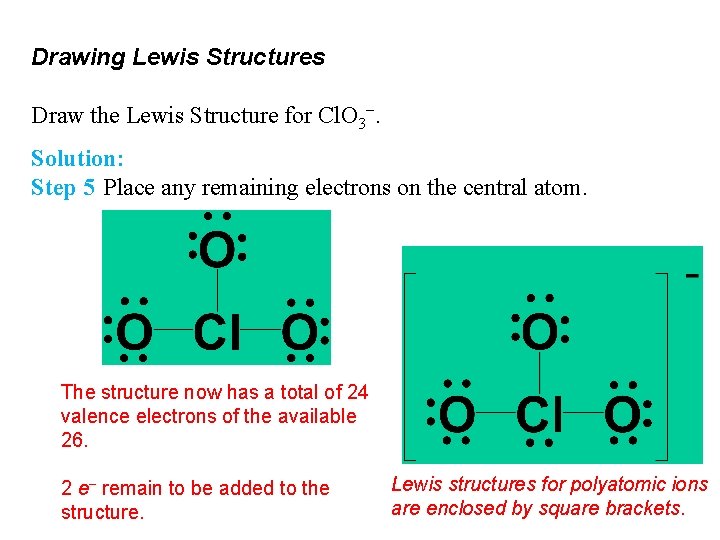
Drawing Lewis Structures Draw the Lewis Structure for Cl. O 3−. Solution: Step 5 Place any remaining electrons on the central atom. The structure now has a total of 24 valence electrons of the available 26. 2 e− remain to be added to the structure. Lewis structures for polyatomic ions are enclosed by square brackets.
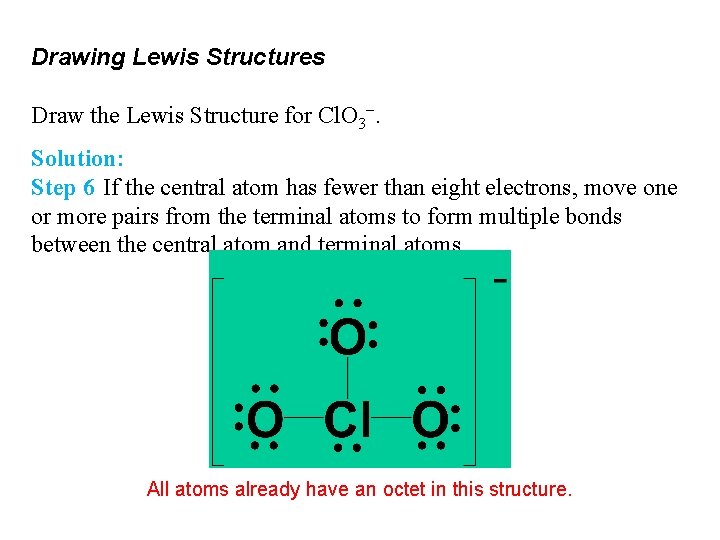
Drawing Lewis Structures Draw the Lewis Structure for Cl. O 3−. Solution: Step 6 If the central atom has fewer than eight electrons, move one or more pairs from the terminal atoms to form multiple bonds between the central atom and terminal atoms. All atoms already have an octet in this structure.
Drawing Lewis Structures

Worked Example 6. 4 Draw the Lewis structure for carbon disulfide (CS 2).

Study Guide for Sections 5. 5, 6. 1 -6. 3 DAY 11, Terms to know: Sections 5. 5, 6. 1 -6. 3: covalent bonding, molecule, law of definite proportions, diatomic molecules, heteronuclear, homonuclear, polyatomic molecules, molecular formula, allotropes, structural formula, electronegativity, octet rule, lone pairs, Lewis structure, single bond, double bond, triple bond, bond length, bond energy DAY 11, Specific outcomes and skills that may be tested on exam 2: Sections 5. 5, 6. 1 -6. 3 • Given a structural formula, be able to give a corresponding molecular formula • Given a molecular or structural formula, be able to determine the formula • Given a formula, be able to predict whether a substance is likely bonded by ionic bonds, covalent bonds, or both • Given a formula, be able to predict which atoms present are bonded through ionic bonds and which atoms are bonded through covalent bonds • Be able to explain why some bonds are shorter and stronger than others • Be able to rank relative electronegativities and explain WHY some atoms are more electronegative than others • Given a molecular formula, be able to draw a reasonable Lewis structure obeying octets and minimizing formal charge where possible

Extra Practice Problems for Sections 5. 5, 6. 1 -6. 3 Complete these problems outside of class until you are confident you have learned the SKILLS in this section outlined on the study guide and we will review some of them next class period. 5. 41 5. 43 5. 49 5. 101 5. 103 5. 107 5. 109 5. 115 5. 121 6. 23 6. 25 6. 67 6. 81 6. 83

Prep for Day 12 Must Watch videos: https: //www. youtube. com/watch? v=7 g. IBj. ZWdh 4 E (formal charge) https: //www. youtube. com/watch? v=MWDL 5 WCZBz. E (resonance) http: //echem 1 a. cchem. berkeley. edu/modules/module 4 -lesson 11. php (UC-Berkeley lesson 11) Other helpful videos: http: //ps. uci. edu/content/chem-1 a-general-chemistry (UC-Irvine lecture 9) http: //ocw. mit. edu/courses/chemistry/5 -111 -principles-of-chemical-science-fall-2008/video-lectures/ (MIT lecture 12) Read sections 6. 4 -6. 6
- Slides: 30