5 4 Coefficients and Subscripts And you A

5 -4 Coefficients and Subscripts And you

• A coefficient is the number in front of chemical formula and represents the number of non-bonded, separate species. In 3 H 2 O, the coefficient 3 indicates there are 3 water molecules.
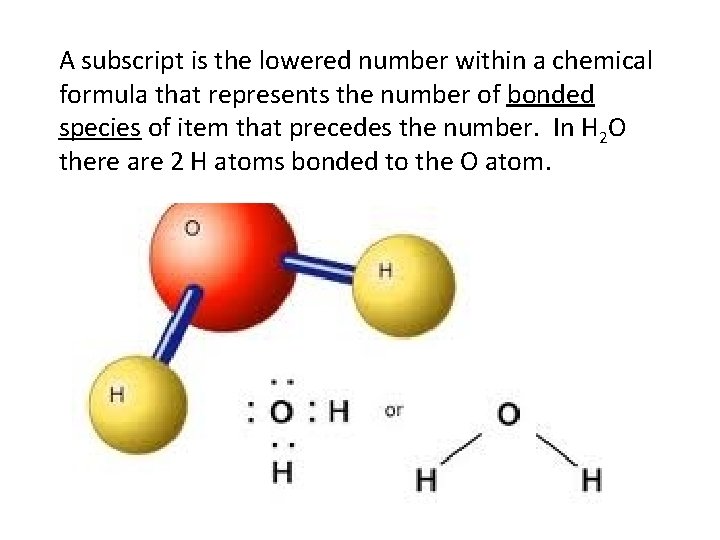
A subscript is the lowered number within a chemical formula that represents the number of bonded species of item that precedes the number. In H 2 O there are 2 H atoms bonded to the O atom.

Building the Concept of Particle Ratios: • • 1 H 2 O molecule 2 H 2 O molecules 1000 H 2 O molec. = = 2 H atoms 1 O atom 4 H atoms 2 O atoms 20 H atoms 10 O atoms 2000 H atoms 1000 O atoms • Do you see the pattern here? ? ?

• 106 H 2 O molec. = 2 x 106 H atoms , 1 x 106 O atoms • 6. 022 x 1023 H 2 O molecules 2(6. 022 x 1023) = H atoms 1(6. 022 x 1023) O atoms • 1 mole H 2 O molec = 2 moles H atoms 1 mole O atoms • 5 moles H 2 O molec = 10 moles H atoms 5 moles O atoms • Great JOB!!!!

• As you can see, the coeffients and subscripts can represent atoms, molecules, or moles. In other words, coefficients and subscripts are merely ratios, representing small things like atoms and molecules, or a large number of these small things, like a mole. Note: coefficients and subscript DO NOT give mass ratios, so we’ll have to convert mass to moles formulas. • Be careful with diatomics, which can be confusing:

• • 6. 022 x 1023 atoms of hydrogen = 1 mole H = 1. 01 g 6. 022 x 1023 molecules of hydrogen = 1 mole H 2 = 2. 02 g = 2 moles of H atoms

Mole Bridge Extension • We can now extend the mole bridge to link molecules to atoms: • Example: • How many H atoms are present in 0. 200 g H 2 O? • • Grams → Molecules → Atoms

Grams → Molecules → Atoms • 0. 200 g H 2 O x 1 mole H 2 O x 6. 022 x 1023 molecules H 2 O x 2 atoms H 18. 02 g 1 mole H 2 O 1 H 2 O molecule See. . . I told you this would be fun!!!

5 -5 Percent Composition (Section 8. 5) • An elemental analysis is the first step in determining a chemical’s formula and gives results called percent composition. Percent composition is typically reported by mass and is determined by dividing the mass of an element in a formula by the total mass of the compound.

Example 1: A compound contains 7. 00 g of nitrogen out of a total sample mass of 23. 0 g. The rest of the mass is oxygen. Calculate the % composition by mass of nitrogen and oxygen. • Since the total mass is 23. 0 g and 7. 00 g of this are nitrogen, 16. 0 g are oxygen. • % N = 7. 00/23. 0 x 100 = 30. 4 % • % O = 16. 0/23. 0 x 100 = 69. 6 % • Note how the % values should add to 100%

Example 2: Determine the % by mass of each element in Ba(OH)2 • 8 H 2 O • Ba(OH)2 • 8 H 2 O Molar mass = 315. 46 g/mol • • Ba: 137. 33/315. 46 x 100 = 43. 53 % O: 2(16. 00)/315. 46 x 100 = 10. 14 % H: 1(1. 01)/315. 4 x 100 = 0. 64 % H 2 O 8(18. 02)/315. 46 x 100 = 45. 69 % • Note how the % values should add to 100%.
- Slides: 12