5 2 QUANTUM THEORY ATOM BOHR MODEL 1913

5. 2 QUANTUM THEORY & ATOM
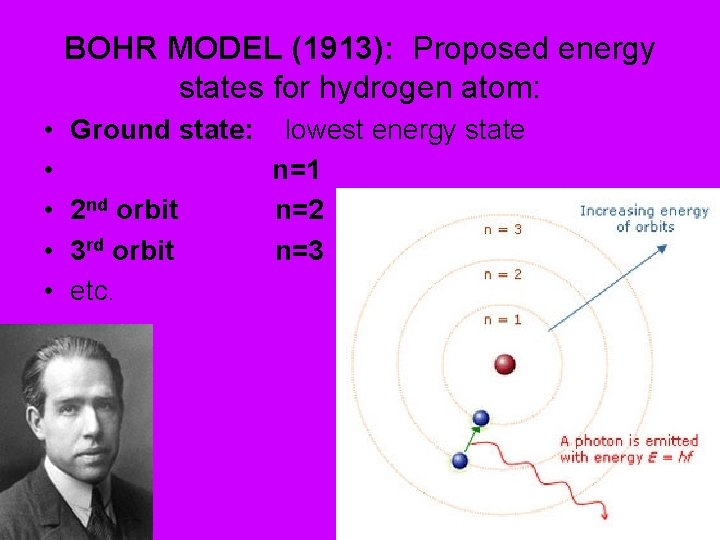
BOHR MODEL (1913): Proposed energy states for hydrogen atom: • • • Ground state: 2 nd orbit 3 rd orbit etc. lowest energy state n=1 n=2 n=3

Louis de Broglie (1924): Accounted for the fixed energy levels in Bohr’s model. • All moving particles have wave characteristics. • May be too small to be detected. • Ex. car • Equation: = h • • m=mass • v=velocity mv • Wave/particle duality of matter.

EXCITED ELECTRON: Energy added from outside source causes electron to move to a higher level. • • Ex. Move from n=1 to n=3 When electron returns to lower state, energy is released: • • E = Ehigher orbit – Elower orbit = Ephoton = h • Energy released gives off a photon corresponding to its frequency. • • Shown as spectral lines (Flame Test)

ELECTRON – Goes to higher energy level when absorb energy. Releases energy when returns to ground state.


HEISENBURG UNCERTAINTY PRINCIPLE: Impossible to know precisely both velocity & position of a particle at the same time. • When take a measurement, the object is disturbed. • This effect is significant if the object is very small (ex. electron). • To find position of an electron, need photon of light. • Photon “bumps” into electron, changing its position.
SCHRODINGER WAVE EQUATION: Very complex • Further developed de Broglie’s wave-particle theory.

QUIZ • 1) The ___ Model proposed energy states for the hydrogen atom. • 2) De Broglie stated that all moving particles have ____ characteristics. • 3) When energy is added, the atom becomes ___. Energy is ___ when the atom returns to the lower state. • 4) Heisenburg’s ____ Principle states that it is impossible to know both the ___ and ___ of a small particle at the same time. • 5) Schrodinger’s ____ equation further developed de Broglie’s wave particle theory.

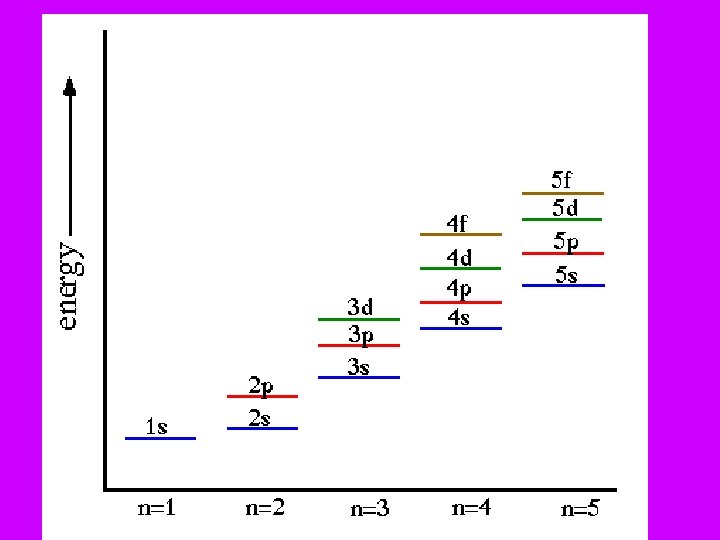
ATOMIC ORBITAL: Describes the electron’s probable location • • 3 -D region around nucleus (“fuzzy cloud”) Quantum #’s – describe each electron in atom (each is unique) • • 1) Principle Quantum Numbers (n): Called Principle Energy Levels • Indicate relative sizes & energies of atomic orbitals. n = 1 (ground state) n values of 1 – 7 (7 energy levels) • •

PRINCIPLE ENERGY LEVELS
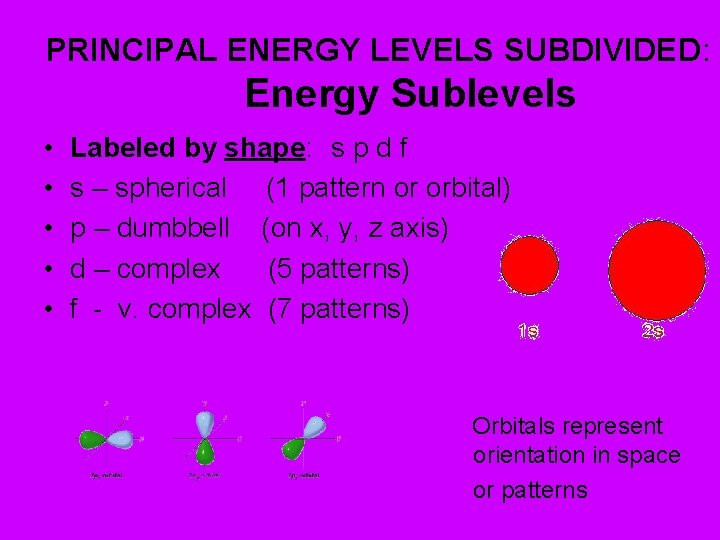
PRINCIPAL ENERGY LEVELS SUBDIVIDED: Energy Sublevels • • • Labeled by shape: s p d f s – spherical (1 pattern or orbital) p – dumbbell (on x, y, z axis) d – complex (5 patterns) f - v. complex (7 patterns) Orbitals represent orientation in space or patterns

s, p, d, f ORBITALS • f orbitals:

ENERGY LEVELS & SUBLEVELS 3 rd • n= 1 • 2 • 3 • 4 • • s s p d f s=1 orbital p=3 orbitals d=5 orbitals f=7 orbitals 1 s 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f f orbital

# ORBITALS = n 2 n = energy level • At: n= 1: • n= 2: • n= 3: 1 orbital (s) 4 orbitals (s, px, py, pz) 9 orbitals (s, px, py, pz, 5 “d”) • Each orbital holds 2 electrons: • (electron can spin clockwise or counterclockwise) • Thus # electrons at an orbital = 2 n 2 • Ex. At n=1, holds 2 electrons • n=2, holds 8 electrons

Orbitals fill from low to high energy:

ENERGY OVERLAP: “d” & “f” have high energies!

QUIZ • 1) The lowest energy state for the electron is called the ____ state. An electron is ___ when it moves to a higher energy level. • 2) The electron’s probable location is called the atomic ____. This is the 3 -D region around the ___. • 3) The 4 types are s, __, and __. • 4) “s” orbitals have a ___ shape. “p” are ___. • 5) Orbitals fill from ___ to ___ energy. Overlap is seen for d and __ orbitals. • 6) How many orbitals in n=4? • How many electrons in n=4?

THE END
- Slides: 20