5 2 Electron Arrangement in Atoms Orbitals Electron

5. 2 Electron Arrangement in Atoms > Orbitals Electron Configurations 1 Orbitals
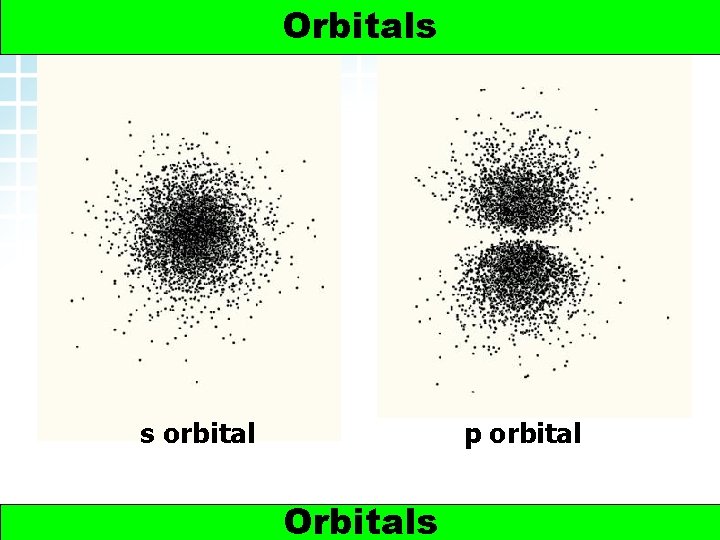
5. 2 Electron Arrangement in Atoms > Orbitals s orbital 2 p orbital Orbitals www. chemsheets. co. uk AS 1009 3 -Jun-2015
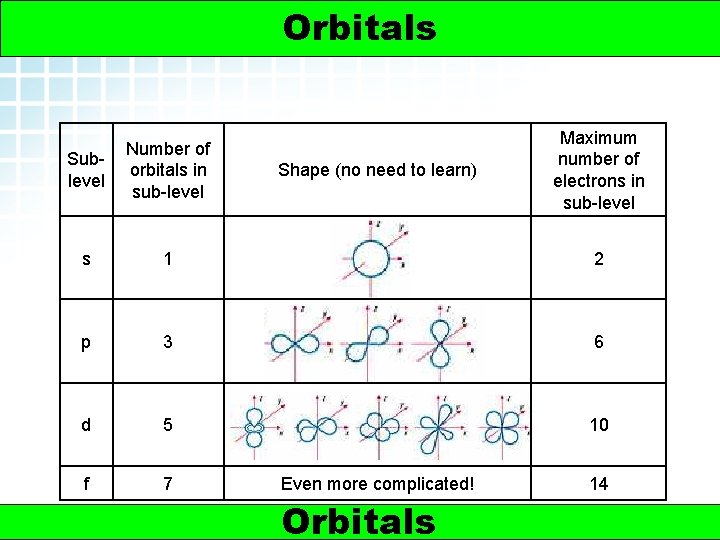
5. 2 Electron Arrangement in Atoms > Orbitals 3 Maximum number of electrons in sub-level Sublevel Number of orbitals in sub-level s 1 2 p 3 6 d 5 10 f 7 Shape (no need to learn) Even more complicated! Orbitals © www. chemsheets. co. uk 14 AS 1009 Electrons 3 -Jun-2015

5. 2 Electron Arrangement in Atoms > Electron Configurations Orbitals The electrons are found orbitals or electron configurations. They follow 3 rules: • The Aufbau Principle • The Paulie Exclusion Principle • Hunds Rule 4 Orbitals Electrons
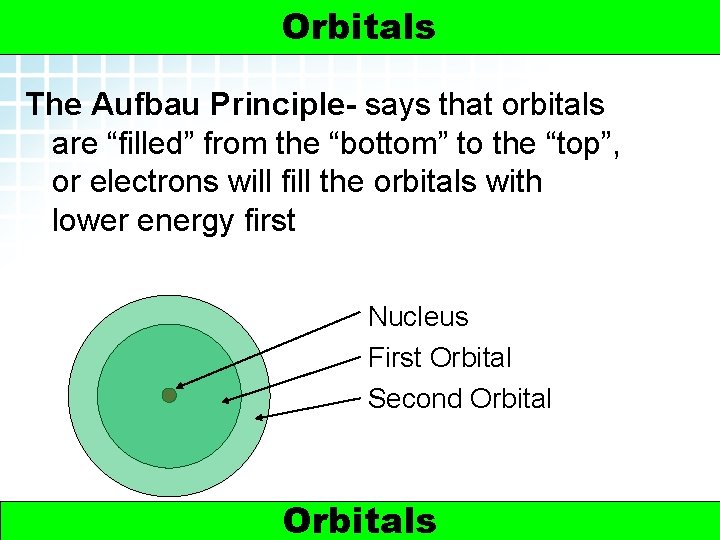
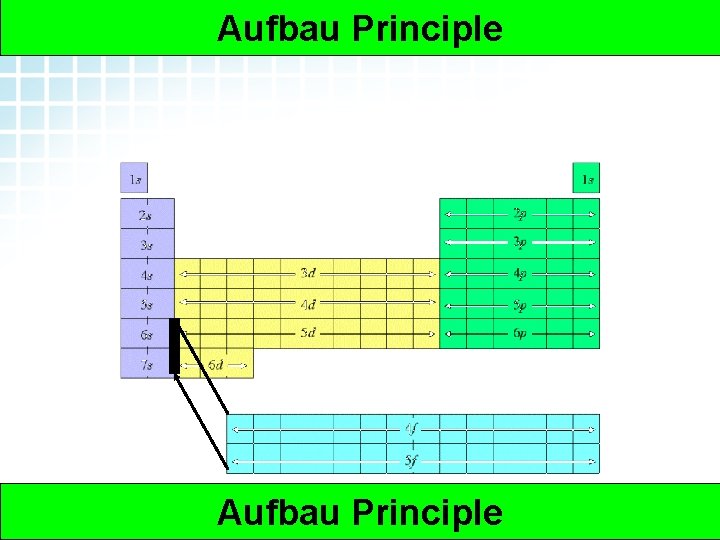
5. 2 Electron Arrangement in Atoms > Orbitals The Aufbau Principle- says that orbitals are “filled” from the “bottom” to the “top”, or electrons will fill the orbitals with lower energy first Nucleus First Orbital Second Orbital 5 Orbitals Electrons
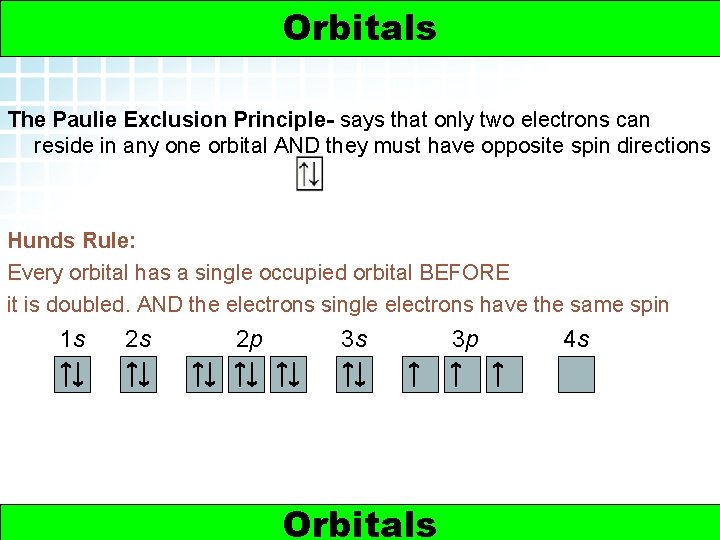
5. 2 Electron Arrangement in Atoms > Orbitals The Paulie Exclusion Principle- says that only two electrons can reside in any one orbital AND they must have opposite spin directions Hunds Rule: Every orbital has a single occupied orbital BEFORE it is doubled. AND the electrons single electrons have the same spin 1 s 6 2 s 2 p 3 s Orbitals 3 p 4 s Electrons

Arrangement of the 5. 2 Electron Arrangement in Atoms > Aufbau Principle Orbitals Some other needed knowledge • Each Orbital belongs to a “Principle Energy Level” • There are 4 main subtypes of orbitals in an atom -s, p, d, and f. And the energy goes s>p>d>f • There is 1 s orbital, 3 p orbitals, 5 d orbitals and 7 F’s • There arrangement follows the periodic table • 7 Or does the periodic table follow their arrangement? ? ? Aufbau Principle Electrons
5. 2 Electron Arrangement in Atoms > Aufbau Principle 8 Aufbau Principle Electrons
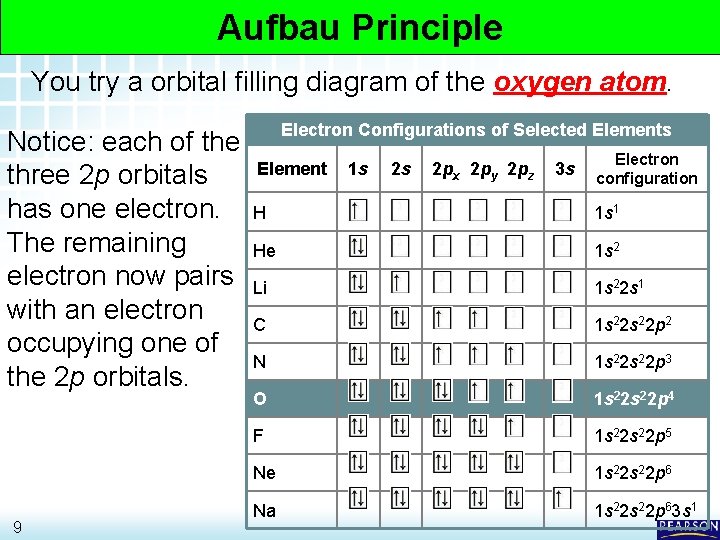
5. 2 Electron Arrangement in Atoms > Electron Configurations Aufbau Principle You try a orbital filling diagram of the oxygen atom. Notice: each of the three 2 p orbitals has one electron. The remaining electron now pairs with an electron occupying one of the 2 p orbitals. 9 Electron Configurations of Selected Elements Element 1 s 2 s 2 px 2 py 2 pz 3 s Electron configuration H 1 s 1 He 1 s 2 Li 1 s 22 s 1 C 1 s 22 p 2 N 1 s 22 p 3 O 1 s 22 p 4 F 1 s 22 p 5 Ne 1 s 22 p 6 Na 1 s 22 p 63 s 1

5. 2 Electron Arrangement in Atoms > CHEMISTRY & YOU Aufbau Principle Explain why the correct electron configuration of oxygen is 1 s 22 p 4 and not 1 s 22 p 33 s 1. Answer: it does not obey the Aufbau Principle 10 Orbitals Electrons

5. 2 Electron Arrangement in Atoms > Sample Problem 5. 1 Aufbau Principle of Phosphorous The atomic number of phosphorus is 15. Write the electron configuration of a phosphorus atom. 1 s 2 s 2 p 3 s 3 p 4 s • The electron configuration of phosphorus is 1 s 22 p 63 s 23 p 3. • The superscripts add up to the number of electrons. 11 Electrons

5. 2 Electron Arrangement in Atoms. Configurations. > Electron Configurations Exceptional Electron This is what a usual one looks like Cr 1 s 22 p 63 s 23 p 63 d 44 s 2 Cu 1 s 22 p 63 s 23 p 63 d 94 s 2 The correct electron configurations are as follows: Cr 1 s 22 p 63 s 23 p 63 d 54 s 1 Cu 1 s 22 p 63 s 23 p 63 d 104 s 1 These arrangements give chromium a half-filled d sublevel and copper a filled d sublevel- This is because filled and half filled orbitals are more stable 12 Electrons

5. 2 Electron Arrangement in Atoms > Orbitals What is the correct electron configuration of a sulfur atom? A. 1 s 22 p 43 s 23 p 6 B. 1 s 22 p 63 s 23 p 3 C. 1 s 22 p 63 s 23 p 4 D. 1 s 22 p 63 s 63 p 2 13 Orbitals Electrons

5. 2 Electron Arrangement in Atoms > Orbitals What is the correct electron configuration of a sulfur atom? A. 1 s 22 p 43 s 23 p 6 B. 1 s 22 p 63 s 23 p 3 C. 1 s 22 p 63 s 23 p 4 D. 1 s 22 p 63 s 63 p 2 14 Orbitals Electrons

5. 2 Electron Arrangement in Atoms > Ionization Energy What is ionisation energy? Definitions Film Clip https: //www. youtube. com/watch? v=ks. Zo. Ode-7 z. Y 15 Ionization Energy Electrons

5. 2 Electron Arrangement in Atoms > Ionization Energy WHAT IS IONISATION ENERGY? Ionisation Energy is a measure of the amount of energy needed to remove electrons from atoms. - The greater the pull of the nucleus, the harder it will be to pull an electron away from an atom. Attraction between the nucleus and an electron FIRST IONISATION ENERGY - Definition The energy required to remove ONE MOLE of electrons from each atom in ONE MOLE of gaseous atoms to form ONE MOLE of gaseous positive ions. e. g. 16 Na(g) Na+(g) + e- Al(g) Al+(g) + e- Make sure you write in the (g) Ionization Energy Electrons

5. 2 Electron Arrangement in Atoms > Ionization Energy Successive Ionisation Energies What does this mean to you? Mg(g) Mg+(g) + e- 1 st I. E. =+738 k. J. mol-1 Mg+(g) Mg 2+(g) + e. Mg 2+(g) Mg 3+(g) + e- 17 2 nd I. E. = + 1451 k. J. mol-1 3 rd I. E. = + 7733 k. J. mol-1 Ionization Energy Electrons
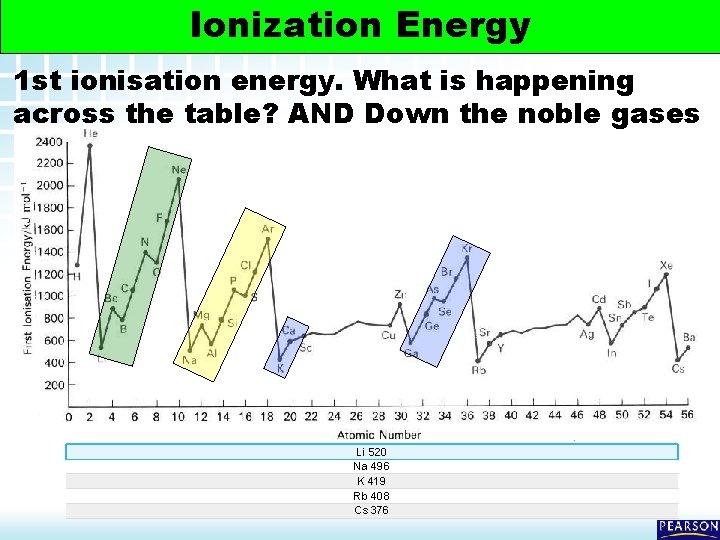
5. 2 Electron Arrangement in Atoms > Ionization Energy 1 st ionisation energy. What is happening across the table? AND Down the noble gases Li 520 Na 496 K 419 Rb 408 Cs 376

Ionization Energy 5. 2 Electron Arrangement in Atoms > Ionization Energy Groups Ionization energies decrease going down a Group, Why? 1) Moving down a group the principle energy level increases distance of electron from the pull of the positive nucleus 2) AND here are more electrons providing inner shell shielding of th nucleus. Therefore the valence electrons are less strongly held Na 2 -8 -1 (IE = 496 KJ/Mol) Rb 2 -8 -18 -8 -1 (IE = 403 KJ/mol) 19 Ionization Energy

5. 2 Electron Arrangement in Atoms > Orbitals 1 st ionisation energy (across period) Atomic Radius DECREASES, why? 1) Increased nuclear charge (i. e. more protons) 2) Thus a stronger attraction from nucleus to electron in outer shell 3) Atoms get smaller 20 Orbitals © www. chemsheets. co. uk AS 1009 Electrons 3 -Jun-2015

5. 2 Electron Arrangement in Atoms > Ionization Energy Orbitals Trends General Trends 1) Ionization energy increases as each successive electron is removed 2) Ionization energy DECREASES going DOWN a GROUP 3) Ionization energy INCREASES as you go ACROSS a PERIOD 4) The noble gases have the highest Ionization energies 5) The alkali metals have the lowest Ionization energies 21 Orbitals 21

5. 2 Electron Arrangement in Atoms > Ions https: //www. youtube. com/watch? v=vp 9 mf. W 7 dq. E 0 IONS FORM by: ALWAYS removing the highest level of n. Or the highest energy electrons are lost when an ion is formed. AND the energy is as follows: least s<p<d<f (most) 22 Orbitals © www. chemsheets. co. uk AS 1009 Electrons 3 -Jun-2015

5. 2 Electron Arrangement in Atoms > Ions ELECTRONIC CONFIGURATION OF IONS 3 Things: Positive ions (cations) are formed by removing electrons from atoms Negative ions (anions) are formed by adding electrons to atoms Electrons are removed first from the highest occupied orbitals SODIUM CHLORINE Na 1 s 2 2 p 6 3 s 1 Na+ 1 s 2 2 p 6 Cl 1 s 2 2 p 6 3 s 2 3 p 5 Cl¯ 1 s 2 2 p 6 3 s 2 3 p 6 1 electron removed from the 3 s orbital 1 electron added to the 3 p orbital • Anions are always larger than their parent atom. • Cations are always smaller than their parent atom. 23 Ions

5. 2 Electron Arrangement in Atoms > Ions ELECTRONIC CONFIGURATION OF IONS FIRST ROW TRANSITION METALS Despite being of lower energy and being filled first, electrons in the 4 s orbital are removed before any electrons in the 3 d orbitals. TITANIUM 24 Ti Ti+ Ti 2+ Ti 3+ Ti 4+ 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 2 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 2 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 3 d 1 1 s 2 2 p 6 3 s 2 3 p 6 Ions
- Slides: 24