5 2 Electron Arrangement in Atoms Chapter 5



























- Slides: 29

5. 2 Electron Arrangement in Atoms > Chapter 5 Electrons In Atoms 5. 1 Revising the Atomic Model 5. 2 Electron Arrangement in Atoms 5. 3 Atomic Emission Spectra and the Quantum Mechanical Model 1 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > CHEMISTRY & YOU What gives gas-filled lights their colors? An electric current passing through the gas in each glass tube makes the gas glow with its own characteristic color. 2 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Light and Atomic Emission Spectra What causes atomic emission spectra? 3 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Light and Atomic Emission Spectra When atoms absorb energy, their electrons move to higher energy levels. These electrons lose energy, by emitting light, when they return to their original (ground) energy levels. 4 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

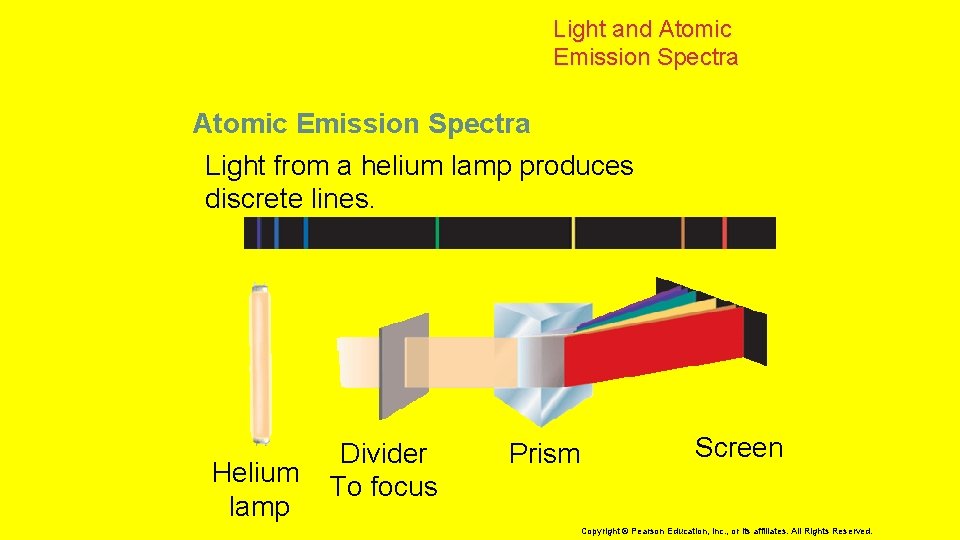
5. 2 Electron Arrangement in Atoms > Light and Atomic Emission Spectra A prism separates light into the colors it contains. White light produces a rainbow of colors. Light source 5 Divider To focus Prism Screen Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

Light and Atomic Emission Spectra Light from a helium lamp produces discrete lines. Helium lamp Divider To focus Prism Screen Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > An Explanation of Atomic Spectra How is the color of light emitted by an atom related to changes of electron energies? 7 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

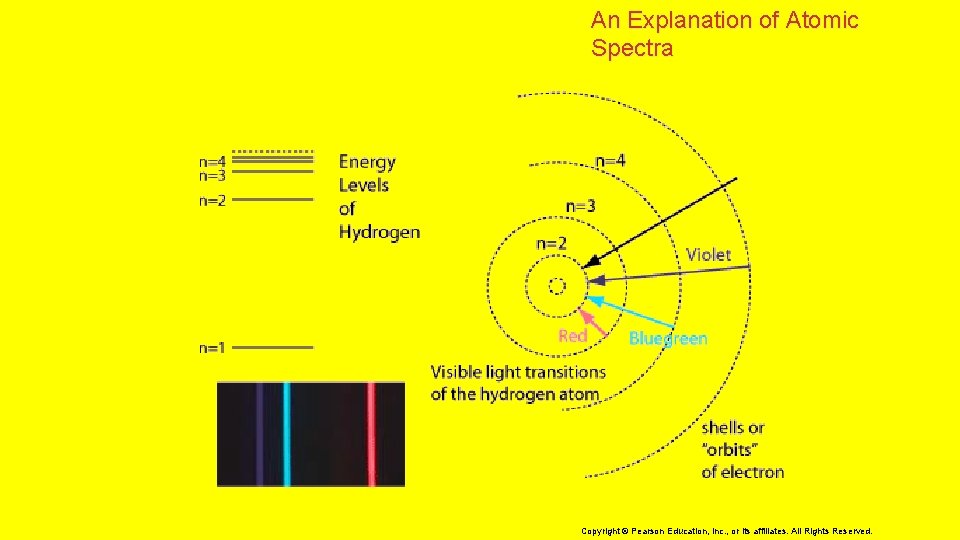
5. 2 Electron Arrangement in Atoms > An Explanation of Atomic Spectra The light emitted by an electron moving from a higher to a lower energy level has a color directly proportional to the energy change of the electron. 8 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > An Explanation of Atomic Spectra When an electron has its lowest possible energy, the atom is in its ground state. 9 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > An Explanation of Atomic Spectra When an electron has its lowest possible energy, the atom is in its ground state. • In the ground state, the principal quantum number (n) is equal to it’s energy level. 10 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > An Explanation of Atomic Spectra When an electron has its lowest possible energy, the atom is in its ground state. • In the ground state, the principal quantum number (n) is equal to it’s energy level. • Excitation of the electron by absorbing energy raises the atom to an excited state. 11 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > An Explanation of Atomic Spectra When an electron has its lowest possible energy, the atom is in its ground state. • In the ground state, the principal quantum number (n) is equal to it’s energy level. • Excitation of the electron by absorbing energy raises the atom to an excited state. • A quantum of energy in the form of light is emitted when the electron drops back its ground state. 12 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

An Explanation of Atomic Spectra Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Light and Atomic Emission Spectra • The energy absorbed by an electron is identical to the energy of light emitted by the electron as it drops back to its ground state. • The wavelengths of the spectral lines are characteristic of the element, and they make up the atomic emission spectrum of the element. 14 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Light and Atomic Emission Spectra • The energy absorbed by an electron is identical to the energy of light emitted by the electron as it drops back to its ground state. • The wavelengths of the spectral lines are characteristic of the element, and they make up the atomic emission spectrum of the element. • No two elements have the same emission spectrum. 15 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > CHEMISTRY & YOU What makes the electron configuration of an atom stable? Having a completely filled valence level. Energy and stability play an important role in determining how electrons are configured in an atom. 16 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations What are three rules for writing the electron configurations of elements? 17 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Three rules — the Aufbau Principle, the Pauli Exclusion Principle, and Hund’s Rule — tell you how to find the electron configurations of atoms. 18 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations The ways in which electrons are arranged in various orbitals around the nuclei of atoms are called electron configurations. 19 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Aufbau Principle 6 p 6 s 5 p Increasing energy 5 s 4 p 4 s 3 p 3 s 2 p 2 s 1 s 20 5 d 4 f 4 d 3 d According to the Aufbau Principle, electrons occupy the orbitals of lowest energy first. In the aufbau diagram, each box represents an atomic orbital. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Aufbau Principle 6 p 6 s 5 p Increasing energy 5 s 4 p 4 s 3 p 3 s 2 p 2 s 1 s 21 5 d 4 f 4 d 3 d The aufbau diagram shows the relative energy levels of the various atomic orbitals. Orbitals of greater energy are higher on the diagram. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Aufbau Principle 6 p 6 s 5 p Increasing energy 5 s 4 p 4 s 3 p 3 s 2 p 2 s 1 s 22 5 d 4 f 4 d 3 d The range of energy sublevels within a principal energy level can overlap the energy levels of another principal level. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Pauli Exclusion Principle • According to the Pauli Exclusion Principle, an atomic orbital may describe at most two electrons. 23 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Pauli Exclusion Principle • According to the Pauli Exclusion Principle, an atomic orbital may describe at most two electrons. • To occupy the same orbital, two electrons must have opposite spins. 24 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Pauli Exclusion Principle • According to the Pauli Exclusion Principle, an atomic orbital may describe at most two electrons. • To occupy the same orbital, two electrons must have opposite spins. 25 • The electron spins must be paired to fill the orbital. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Pauli Exclusion Principle • Spin is a quantum mechanical property of electrons and may be thought of as clockwise or counterclockwise. 26 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Pauli Exclusion Principle • Spin is a quantum mechanical property of electrons and may be thought of as clockwise or counterclockwise. • A vertical arrow indicates an electron and its direction of spin ( or ). 27 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > Electron Configurations Pauli Exclusion Principle • Spin is a quantum mechanical property of electrons and may be thought of as clockwise or counterclockwise. • A vertical arrow indicates an electron and its direction of spin ( or ). 28 • An orbital containing paired electrons is written as. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

5. 2 Electron Arrangement in Atoms > END OF 5. 2 29 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.