5 1 Acids and Bases Acids and bases

5. 1 Acids and Bases • Acids and bases are very common. w Many familiar compounds are acids or bases. w Classification as acids or bases is based on chemical composition. • Acids and bases can be very dangerous! w Both can be very corrosive. § NEVER try to identify an acid or base by taste or touch! • The strength of acids and bases in measured on the p. H scale w p. H below 7 = acidic 7 = basic w 0 1 2 3 4 5 Acids p. H 7 = neutral 6 7 8 Neutral 9 10 p. H above 11 12 13 14 Bases See pages 220 - 222 w Each decrease of 1 on the p. H scale indicates 10 X more (c) Mc. Graw Hill Ryerson 2007 acidic

p. H Indicators • The p. H of acids and bases cannot be determined by sight. w Instead, p. H is measured by other chemicals called indicators, or by a p. H meter that measures the electrical conductivity of the solution. • p. H indicators change colour based on the solution they are placed in. w Litmus is the most common indictor, used on litmus paper. § Two colours of litmus paper: § Red = p. H below 7=acidic, Blue = p. H above 7=basic w Universal indicator contains many indicators that turn different colours at different p. H values (can be in liquid form, or on paper strips like litmus) w A p. H meter uses electrical probes to measure how solutions conduct electricity See pages 223 - 224 w Indicators change colour at different p. H values, so different (c) Mc. Graw Hill Ryerson 2007

Rules for Writing Formulas of Acids: • If you know a compound’s chemical formula, you may be able to identify it as an acid. w Acids often behave like acids only when dissolved in water w Therefore, acids are often written with subscript (aq) = aqueous = water • The chemical formula of an acid usually starts with Hydrogen (H-). w Acids with a carbon usually have the C written first. § Hydrogen + non metal • HCl(aq) = hydrochloric acid, § Hydrogen + polyatomic ion • HNO 3(aq) = nitric acid, § Carbon molecule + Hydrogen See pages 225 - 226 • CH 3 COOH(aq) = acetic acid (c) Mc. Graw Hill Ryerson 2007

Rules for Writing Names of Acids: • Naming acids: from chemical name → acid name w Hydrogen + ____ide = Hydro____ic acid § HF(aq) = _______________ = ______________ w Hydrogen + ____ate = ______ic acid § H 2 CO 3(aq) = ______________________________ w Hydrogen + ____ite = ______ous acid § H 2 SO 3(aq) = ____________________________

Bases • If you know a compound’s chemical formula, you may be able to identify it as a base. w Bases, like acids, often behave like bases only when dissolved in water w Therefore, bases are often written with subscript (aq) = aqueous = water • The chemical formula of a base usually ends with hydroxide (-OH). • Bases can be gentle or very caustic • Examples of common bases w w Na. OH(aq) Mg(OH)2(aq) Ca(OH)2(aq) NH 4 OH(aq) See page 227 (c) Mc. Graw Hill Ryerson 2007

Production of Ions • Acids and bases can conduct electricity because they release ions in solution. w Acids release hydrogen ions, H+(aq) w Bases release hydroxide ions OH–(aq) • The p. H of a solution refers to the concentration of ions it has. w Square brackets are used to signify concentration, [H+(aq)], [OH–(aq)] § High [H+(aq)] = low p. H, very acidic § High [OH–(aq)] = high p. H, very basic w A solution cannot have BOTH high [H+(aq)] and [OH–(aq)]; they cancel each other out and form water. This process is called neutraliztion. w H+(aq) + OH–(aq) H 2 O( ) See page 228 (c) Mc. Graw Hill Ryerson 2007
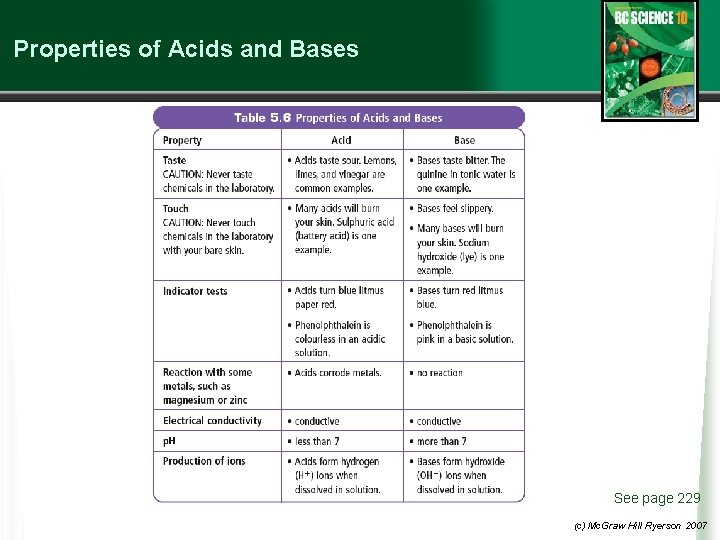
Properties of Acids and Bases See page 229 (c) Mc. Graw Hill Ryerson 2007
- Slides: 7