48 Year Old Female with Elevated Cholesterol Abnormal

48 Year Old Female with Elevated Cholesterol, Abnormal LFTs, Hypothyroidism, and Undiagnosed Diabetes Case Category: Diabetes History of present illness: 48 year old female with fatigue, difficulty sleeping and concentrating. History of elevated cholesterol (no prior treatment) and hypothyroidism (briefly treated with Synthroid).
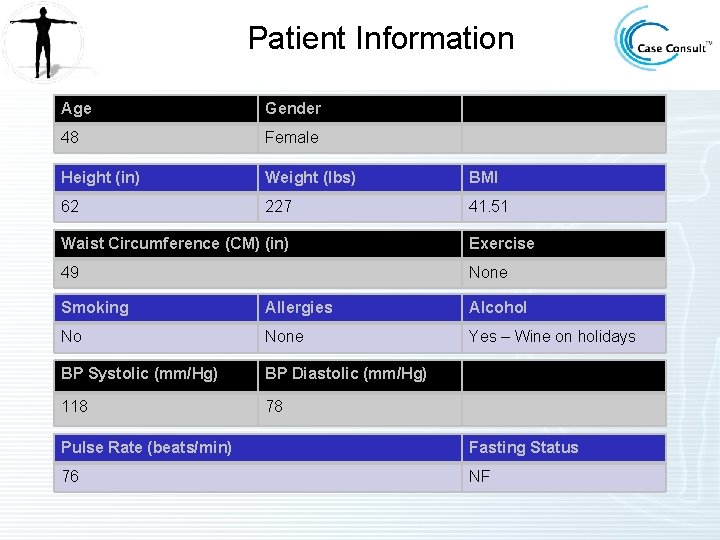
Patient Information Age Gender 48 Female Height (in) Weight (lbs) BMI 62 227 41. 51 Waist Circumference (CM) (in) Exercise 49 None Smoking Allergies Alcohol No None Yes – Wine on holidays BP Systolic (mm/Hg) BP Diastolic (mm/Hg) 118 78 Pulse Rate (beats/min) Fasting Status 76 NF
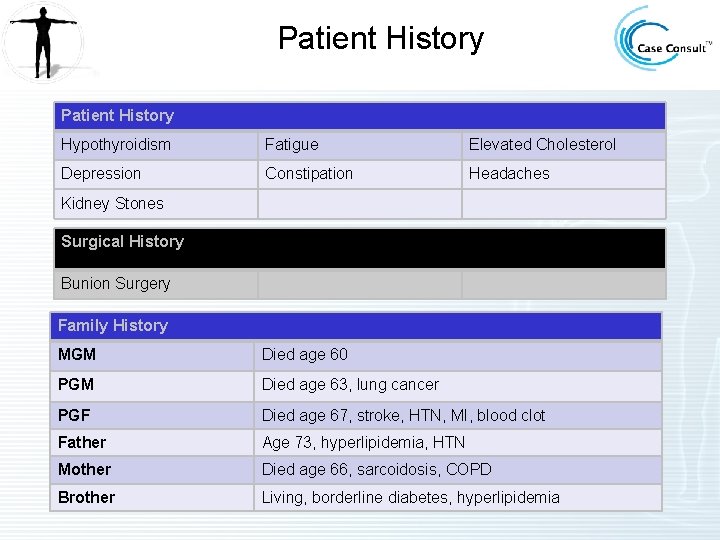
Patient History Hypothyroidism Fatigue Elevated Cholesterol Depression Constipation Headaches Kidney Stones Surgical History Bunion Surgery Family History MGM Died age 60 PGM Died age 63, lung cancer PGF Died age 67, stroke, HTN, MI, blood clot Father Age 73, hyperlipidemia, HTN Mother Died age 66, sarcoidosis, COPD Brother Living, borderline diabetes, hyperlipidemia

Current Medications Current Medication Prior to this Visit Vitamin D 3 2400 IU Daily
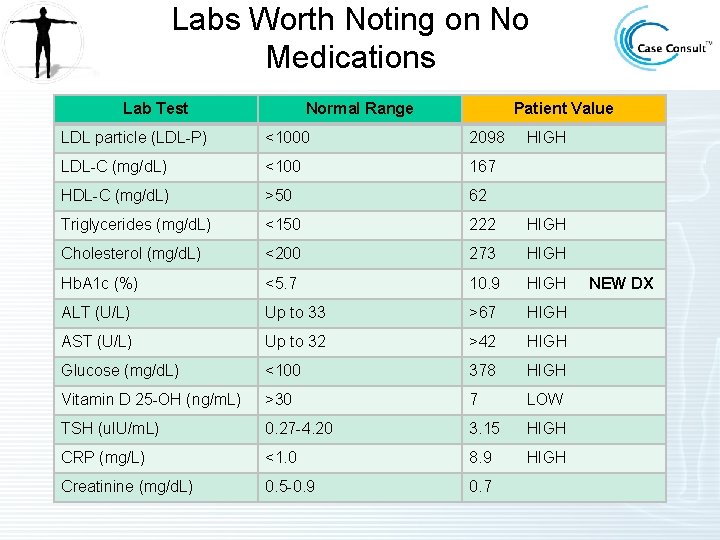
Labs Worth Noting on No Medications Lab Test Normal Range Patient Value LDL particle (LDL-P) <1000 2098 LDL-C (mg/d. L) <100 167 HDL-C (mg/d. L) >50 62 Triglycerides (mg/d. L) <150 222 HIGH Cholesterol (mg/d. L) <200 273 HIGH Hb. A 1 c (%) <5. 7 10. 9 HIGH ALT (U/L) Up to 33 >67 HIGH AST (U/L) Up to 32 >42 HIGH Glucose (mg/d. L) <100 378 HIGH Vitamin D 25 -OH (ng/m. L) >30 7 LOW TSH (u. IU/m. L) 0. 27 -4. 20 3. 15 HIGH CRP (mg/L) <1. 0 8. 9 HIGH Creatinine (mg/d. L) 0. 5 -0. 9 0. 7 HIGH NEW DX

Questions to Consider • Question 1: Multiple untreated conditions? Assess motivation to start several therapies. New diabetes diagnosis is a very important secondary cause for abnormal lipids/lipoproteins/triglycerides. This should be focus of initial treatment. • Question 2: Diet review is critical. Avoid simple sugars, carbohydrates, and alcohol. • Question 3: Any evidence of end organ disease from diabetes? Has patient ever had any renal issues? Is creatinine normal? Seeing an ophthalmologist regularly?

Criteria for the Diagnosis of Diabetes A 1 C ≥ 6. 5% OR Fasting plasma glucose (FPG) ≥ 126 mg/d. L (7. 0 mmol/L) OR 2 -h plasma glucose ≥ 200 mg/d. L (11. 1 mmol/L) during an OGTT OR A random plasma glucose ≥ 200 mg/d. L (11. 1 mmol/L) ADA. I. Classification and Diagnosis. Diabetes Care 2012; 35(suppl 1): S 12. Table 2.

Labs on No Medications (1 of 3)
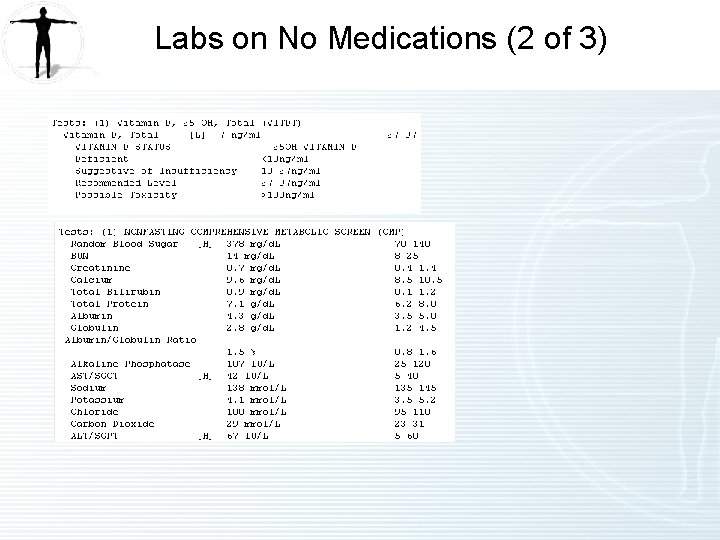
Labs on No Medications (2 of 3)

Labs on No Medications (3 of 3)

NMR Lipoprofile • Insert NMR Lipo. Profile 03112010 TP 61 Insert Page 2

Initial Treatment & Management • Advised low glycemic, low saturated fat diet and daily exercise. • Start metformin ER 750 mg 2 -3 tablets daily with slow titration as tolerated. • Start Byetta 10 mcg injectable twice daily. (Start with 5 mcg BID x 1 month and then increase to 10) • Start Crestor 5 mg/day due to >60 % reduction LDL needed. • Start Fish Oil OTC 2000 mg with EPA and DHA at meal. • Increase Vitamin D 3 to 5000 IU daily and 50, 000 IU/weekly for vitamin D deficiency. • Follow-up with PCP regarding elevated TSH.
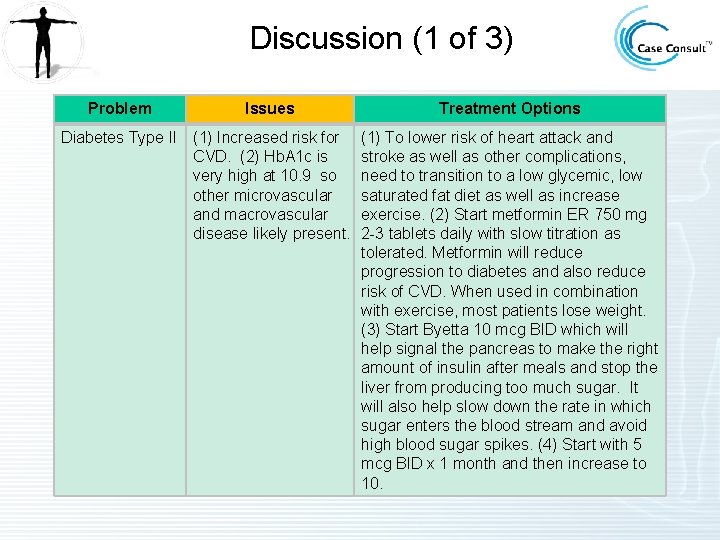
Discussion (1 of 3) Problem Issues Diabetes Type II (1) Increased risk for CVD. (2) Hb. A 1 c is very high at 10. 9 so other microvascular and macrovascular disease likely present. Treatment Options (1) To lower risk of heart attack and stroke as well as other complications, need to transition to a low glycemic, low saturated fat diet as well as increase exercise. (2) Start metformin ER 750 mg 2 -3 tablets daily with slow titration as tolerated. Metformin will reduce progression to diabetes and also reduce risk of CVD. When used in combination with exercise, most patients lose weight. (3) Start Byetta 10 mcg BID which will help signal the pancreas to make the right amount of insulin after meals and stop the liver from producing too much sugar. It will also help slow down the rate in which sugar enters the blood stream and avoid high blood sugar spikes. (4) Start with 5 mcg BID x 1 month and then increase to 10.
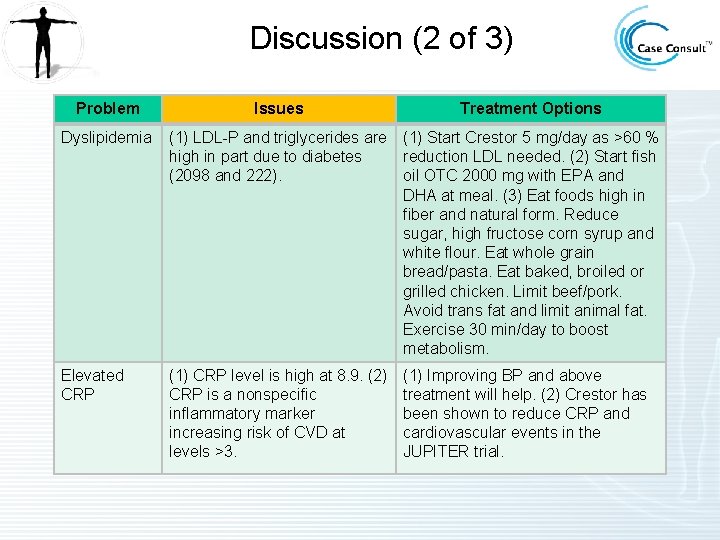
Discussion (2 of 3) Problem Issues Treatment Options Dyslipidemia (1) LDL-P and triglycerides are high in part due to diabetes (2098 and 222). (1) Start Crestor 5 mg/day as >60 % reduction LDL needed. (2) Start fish oil OTC 2000 mg with EPA and DHA at meal. (3) Eat foods high in fiber and natural form. Reduce sugar, high fructose corn syrup and white flour. Eat whole grain bread/pasta. Eat baked, broiled or grilled chicken. Limit beef/pork. Avoid trans fat and limit animal fat. Exercise 30 min/day to boost metabolism. Elevated CRP (1) CRP level is high at 8. 9. (2) CRP is a nonspecific inflammatory marker increasing risk of CVD at levels >3. (1) Improving BP and above treatment will help. (2) Crestor has been shown to reduce CRP and cardiovascular events in the JUPITER trial.
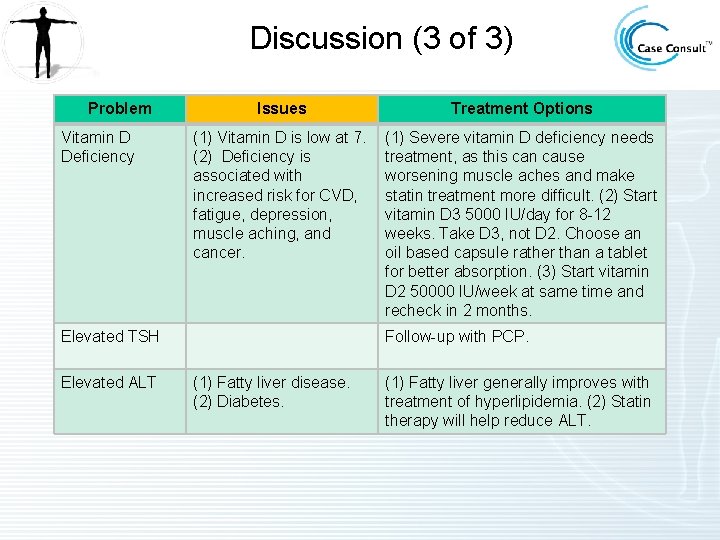
Discussion (3 of 3) Problem Vitamin D Deficiency Issues Treatment Options (1) Vitamin D is low at 7. (2) Deficiency is associated with increased risk for CVD, fatigue, depression, muscle aching, and cancer. (1) Severe vitamin D deficiency needs treatment, as this can cause worsening muscle aches and make statin treatment more difficult. (2) Start vitamin D 3 5000 IU/day for 8 -12 weeks. Take D 3, not D 2. Choose an oil based capsule rather than a tablet for better absorption. (3) Start vitamin D 2 50000 IU/week at same time and recheck in 2 months. Elevated TSH Elevated ALT Follow-up with PCP. (1) Fatty liver disease. (2) Diabetes. (1) Fatty liver generally improves with treatment of hyperlipidemia. (2) Statin therapy will help reduce ALT.

Follow Up – 1 on Crestor 5, Byetta 10 BID, Metformin 1500 • Type II Diabetes – Improved. – Hb. A 1 c lowered to 7. 5. – As Byetta is hard to dose with meals and she is skipping on occasion, switch to Victoza once daily injectable. She is an excellent candidate for GLP 1 agonist as glucose is on average at 180. – Increase metformin to 2250 mg/day. • Dyslipidemia – Improved. – On Crestor 5 mg/day. – LDL-P is down to 1140 from 2098. LDL-C was 167 and is now 67. Triglycerides are down to 131 from 222. Continue low dose for now as tolerating. • Vitamin D Deficiency – Improved. – Levels increased at 2400 IU/day from 7 to 41. – Increase to 5000 IU/day. – Continue therapy. • Elevated TSH – Unchanged. – Consulted specialist. Thyroid ultrasound showed nondescript, nodules left lobe. – Referred to endocrinologist.

Follow Up Labs on Crestor 5, Byetta 10, Metformin 1500

Follow-Up 2 on Crestor 5, Victoza 1. 8 and Metformin 2250 (1 of 2) • Vitamin B 12 Deficiency – Vitamin B 12 is low at 304. – Cause of B 12 deficiency may be due to metformin, bacterial flora changes that induce food cobalamin malabsorption, poor dietary intake of foods high in B 12, and some chronic diseases. – Treatment of B 12 deficiency is important to avoid health changes. – Start sublingal 1000 mcg/day and recheck. – Foods high in B 12 that are also heart healthy include sockeye salmon, plain yogurt, milk and roasted chicken. • Type II Diabetes – Improved. – Hb. A 1 c is now 5. 7. – On Victoza but having issues with injection. – Try Cycloset 0. 8 mg/day as a new option of diabetes management as it does not lower glucose to the extent of causing hypoglycemia and works well to treat insulin resistance. Also beneficial on lipids and has been shown to reduce CVD events in the safety/efficacy trial.

Follow-Up 2 on Crestor 5, Victoza 1. 8 and Metformin 2250 (2 of 2) • Vitamin D Deficiency – Suspect Improved. – Levels not checked today. • Dyslipidemia – Deteriorated. – Very high stress likely contributing to symptoms and labs worsening. – On Crestor 5 mg, Metformin 2250 mg, Victoza 1. 8 mg and OTC fish oil 4 g/day. – LDL-P is up to 1181 from 661. – Advise higher dose of Crestor to 10 mg/day. – Meet with dietitian to improve diet. • Elevated TSH – Unchanged. – Seeing endocrinologist. No treatment indicated. • Elevated ALT – Improved as expected with lipid and diabetes treatment. • Sleep Apnea – Advise work-up for sleep apnea due to disturbed sleep and increased cardiovascular risk. – May not respond well to Cycloset if sleep pattern not normal.

Follow-Up Labs on Crestor 5, Victoza 1. 8 and Metformin 2250

Follow-Up 3 On Crestor 10, Cycloset 0. 8 and Metformin 2250 • Diabetes Type 2 – Improved. – Hb. A 1 c is normal at 5. 6. – Tolerating Cycloset. – Continue therapy. • Vitamin D Deficiency – Deteriorated. – Taking Vitamin D 3 10, 000 IU/day. – Consider using name brand restarting 50, 000 IU/weekly. • Dyslipidemia – Improved. – LDL-P is now 486 compared to 1181 on Crestor 10 mg, Cycloset 0. 8 mg, and Omega 3. • B 12 Deficiency – Improved on supplement. – Continue due to ongoing use of high dose metformin. • Elevated CRP – Improved. • Fatigue – Improved with treatment of vitamin D deficiency, vitamin B 12 deficiency, and diabetes.
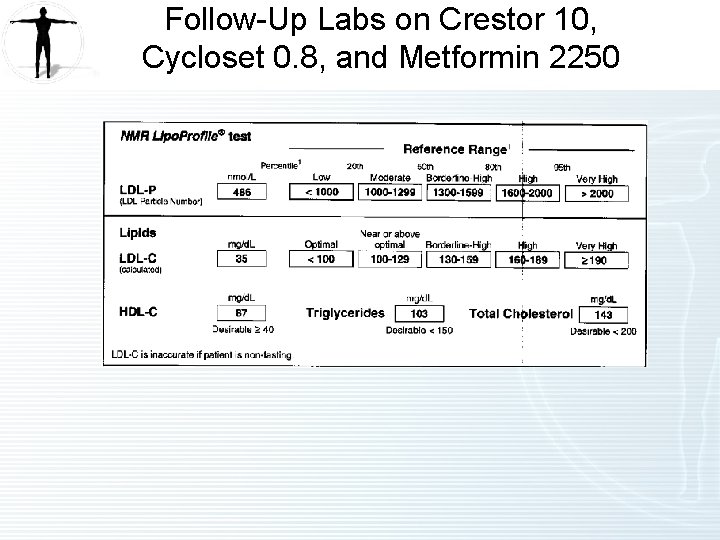
Follow-Up Labs on Crestor 10, Cycloset 0. 8, and Metformin 2250

Clinical Pearls • Aggressive treatment of diabetes with medications that also help lipoprotein abnormalities is key. Here we have example of appropriate use of GLP-1 agonist in addition to metformin at initial visit when diabetes first diagnosed as Hb. A 1 C >10. • Twice daily therapy is often associated with lack of compliance. There are several options now for either once daily GLP-1 agonists or now once weekly Byetta (Bydureon) 2/2012. • Cycloset is an excellent option for diabetes management when injectable is either not desired or not effective. Use of these above combination therapies may be very effective and avoid need for starting insulin. Cycloset is weight neutral and GLP-1 agonists cause weight loss vs. the weight gain that may occur with insulin.
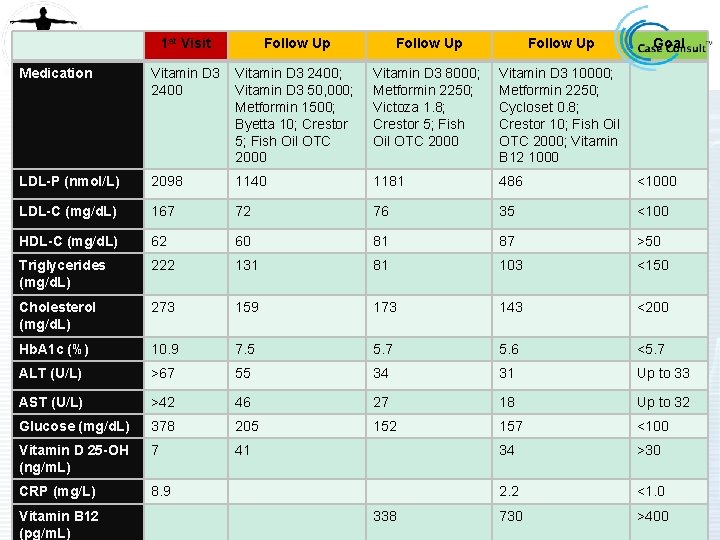
Follow Up Case Summary 1 st Visit Follow Up Medication Vitamin D 3 2400; Vitamin D 3 50, 000; Metformin 1500; Byetta 10; Crestor 5; Fish Oil OTC 2000 Vitamin D 3 8000; Metformin 2250; Victoza 1. 8; Crestor 5; Fish Oil OTC 2000 Vitamin D 3 10000; Metformin 2250; Cycloset 0. 8; Crestor 10; Fish Oil OTC 2000; Vitamin B 12 1000 LDL-P (nmol/L) 2098 1140 1181 486 <1000 LDL-C (mg/d. L) 167 72 76 35 <100 HDL-C (mg/d. L) 62 60 81 87 >50 Triglycerides (mg/d. L) 222 131 81 103 <150 Cholesterol (mg/d. L) 273 159 173 143 <200 Hb. A 1 c (%) 10. 9 7. 5 5. 7 5. 6 <5. 7 ALT (U/L) >67 55 34 31 Up to 33 AST (U/L) >42 46 27 18 Up to 32 Glucose (mg/d. L) 378 205 152 157 <100 Vitamin D 25 -OH (ng/m. L) 7 41 34 >30 CRP (mg/L) 8. 9 2. 2 <1. 0 730 >400 Vitamin B 12 (pg/m. L) 338 Follow Up Goal

Diabetes Care: Initial Evaluation • A complete medical evaluation should be performed to – Classify the diabetes – Detect presence of diabetes complications – Review previous treatment, glycemic control in patients with established diabetes – Assist in formulating a management plan – Provide a basis for continuing care • Perform laboratory tests necessary to evaluate each patient’s medical condition ADA. V. Diabetes Care 2012; 35(suppl 1): S 16.

Components of the Comprehensive Diabetes Evaluation (1) Medical history (1) • Age and characteristics of onset of diabetes (e. g. , DKA, asymptomatic laboratory finding) • Eating patterns, physical activity habits, nutritional status, and weight history; growth and development in children and adolescents • Diabetes education history • Review of previous treatment regimens and response to therapy (A 1 C records) ADA. V. Diabetes Care 2012; 35(suppl 1): S 17. Table 7.

Components of the Comprehensive Diabetes Evaluation (2) Medical history (2) • Current treatment of diabetes, including medications and medication adherence, meal plan, physical activity patterns, and readiness for behavior change • Results of glucose monitoring and patient’s use of data • DKA frequency, severity, and cause • Hypoglycemic episodes – Hypoglycemia awareness – Any severe hypoglycemia: frequency and cause ADA. V. Diabetes Care 2012; 35(suppl 1): S 17. Table 7.

Components of the Comprehensive Diabetes Evaluation (3) Medical history (3) • History of diabetes-related complications – Microvascular: retinopathy, nephropathy, neuropathy • Sensory neuropathy, including history of foot lesions • Autonomic neuropathy, including sexual dysfunction and gastroparesis – Macrovascular: CHD, cerebrovascular disease, PAD – Other: psychosocial problems*, dental disease* *See appropriate referrals for these categories. ADA. V. Diabetes Care 2012; 35(suppl 1): S 17. Table 7.

Components of the Comprehensive Diabetes Evaluation (4) Physical examination (1) • Height, weight, BMI • Blood pressure determination, including orthostatic measurements when indicated • Fundoscopic examination* • Thyroid palpation • Skin examination (for acanthosis nigricans and insulin injection sites) *See appropriate referrals for these categories. ADA. V. Diabetes Care 2012; 35(suppl 1): S 17. Table 7.

Components of the Comprehensive Diabetes Evaluation (5) Physical examination (2) • Comprehensive foot examination – – Inspection Palpation of dorsalis pedis and posterior tibial pulses Presence/absence of patellar and Achilles reflexes Determination of proprioception, vibration, and monofilament sensation ADA. V. Diabetes Care 2012; 35(suppl 1): S 17. Table 7.

Components of the Comprehensive Diabetes Evaluation (6) Laboratory evaluation • A 1 C, if results not available within past 2– 3 months • If not performed/available within past year – Fasting lipid profile, including total, LDL, and HDL cholesterol and triglycerides – Liver function tests – Test for urine albumin excretion with spot urine albumin-tocreatinine ratio – Serum creatinine and calculated GFR – Thyroid-stimulating hormone in type 1 diabetes, dyslipidemia, or women over age 50 years ADA. V. Diabetes Care 2012; 35(suppl 1): S 17. Table 7.

Components of the Comprehensive Diabetes Evaluation (7) Referrals • Eye care professional for annual dilated eye exam • Family planning for women of reproductive age • Registered dietitian for MNT • Diabetes self-management education • Dentist for comprehensive periodontal examination • Mental health professional, if needed ADA. V. Diabetes Care 2012; 35(suppl 1): S 17. Table 7.
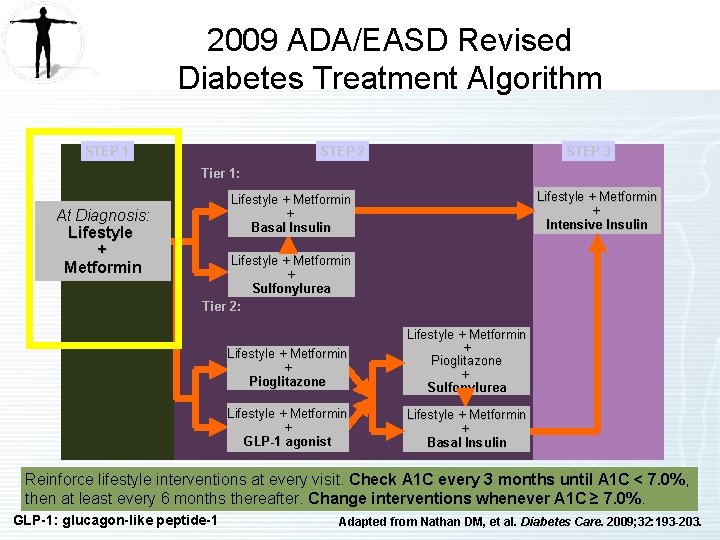
2009 ADA/EASD Revised Diabetes Treatment Algorithm STEP 1 STEP 2 STEP 3 Tier 1: At Diagnosis: Lifestyle + Metformin + Intensive Insulin Lifestyle + Metformin + Basal Insulin Lifestyle + Metformin + Sulfonylurea Tier 2: Lifestyle + Metformin + Pioglitazone + Sulfonylurea Lifestyle + Metformin + GLP-1 agonist Lifestyle + Metformin + Basal Insulin Reinforce lifestyle interventions at every visit. Check A 1 C every 3 months until A 1 C < 7. 0%, then at least every 6 months thereafter. Change interventions whenever A 1 C ≥ 7. 0%. GLP-1: glucagon-like peptide-1 Adapted from Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203.
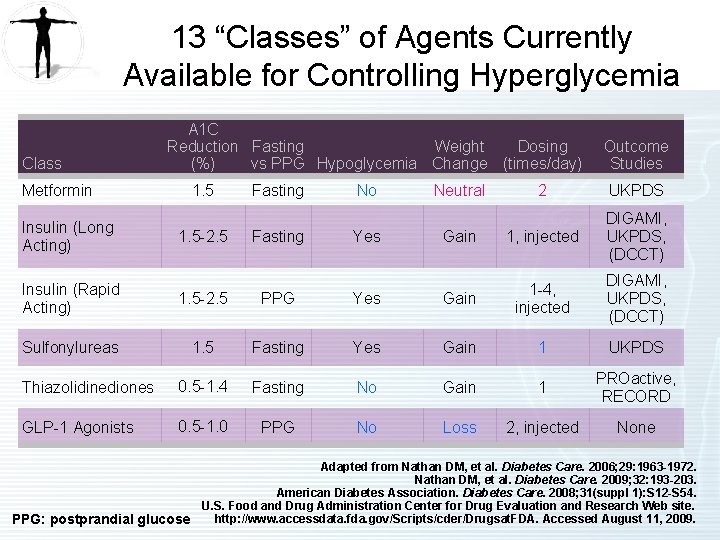
13 “Classes” of Agents Currently Available for Controlling Hyperglycemia Class Metformin Insulin (Long Acting) A 1 C Reduction Fasting Weight Dosing (%) vs PPG Hypoglycemia Change (times/day) 1. 5 -2. 5 Fasting No Yes Neutral Outcome Studies 2 UKPDS Gain 1, injected DIGAMI, UKPDS, (DCCT) Insulin (Rapid Acting) 1. 5 -2. 5 PPG Yes Gain 1 -4, injected Sulfonylureas 1. 5 Fasting Yes Gain 1 UKPDS Thiazolidinediones 0. 5 -1. 4 Fasting No Gain 1 PROactive, RECORD GLP-1 Agonists 0. 5 -1. 0 PPG No Loss 2, injected None Adapted from Nathan DM, et al. Diabetes Care. 2006; 29: 1963 -1972. Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203. American Diabetes Association. Diabetes Care. 2008; 31(suppl 1): S 12 -S 54. U. S. Food and Drug Administration Center for Drug Evaluation and Research Web site. http: //www. accessdata. fda. gov/Scripts/cder/Drugsat. FDA. Accessed August 11, 2009. PPG: postprandial glucose
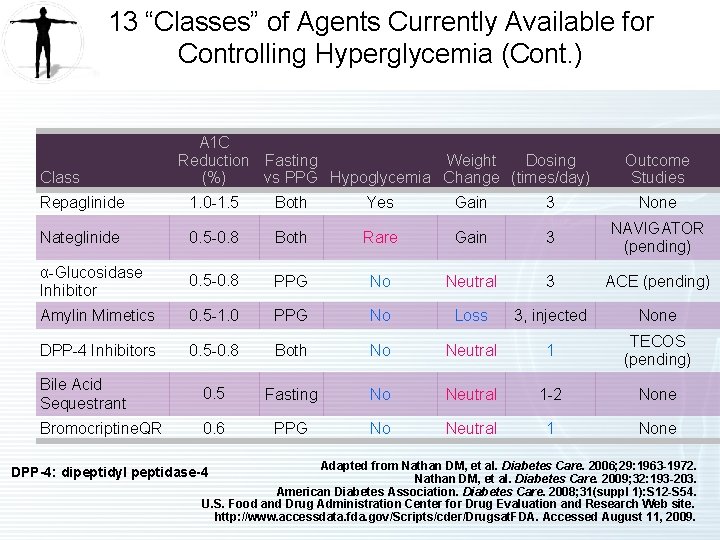
13 “Classes” of Agents Currently Available for Controlling Hyperglycemia (Cont. ) Class A 1 C Reduction Fasting Weight Dosing (%) vs PPG Hypoglycemia Change (times/day) Outcome Studies Repaglinide 1. 0 -1. 5 Both Yes Gain 3 None Nateglinide 0. 5 -0. 8 Both Rare Gain 3 NAVIGATOR (pending) α-Glucosidase Inhibitor 0. 5 -0. 8 PPG No Neutral 3 ACE (pending) Amylin Mimetics 0. 5 -1. 0 PPG No Loss 3, injected None DPP-4 Inhibitors 0. 5 -0. 8 Both No Neutral 1 TECOS (pending) Bile Acid Sequestrant 0. 5 Fasting No Neutral 1 -2 None Bromocriptine. QR 0. 6 PPG No Neutral 1 None Adapted from Nathan DM, et al. Diabetes Care. 2006; 29: 1963 -1972. Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203. American Diabetes Association. Diabetes Care. 2008; 31(suppl 1): S 12 -S 54. U. S. Food and Drug Administration Center for Drug Evaluation and Research Web site. http: //www. accessdata. fda. gov/Scripts/cder/Drugsat. FDA. Accessed August 11, 2009. DPP-4: dipeptidyl peptidase-4

Selecting an Individualized Glycemic Regimen From Tier 1 or Tier 2 Tier 1 Agents Tier 2 Agents • When metformin is not tolerated or is contraindicated: • When avoidance of hypoglycemia is important: – Glomerular filtration rate < 30 m. L/min, Creatinine >1. 5 • When a large reduction in A 1 C is needed: – A 1 C reduction ≥ 1. 5% • When medication cost is a hardship – Certain occupations (eg, drivers, heavy equipment operators, using power tools, public safety jobs) – Certain populations (eg, history of hypoglycemia, alcohol use, elderly patients, renal insufficiency) • When promotion of weight loss is important Adapted from Nathan DM, et al. Diabetes Care. 2009; 32: 193 -203. Lassmann-Vague V. Diabetes Metab. 2005; 31: 5 S 53 -5 S 57.
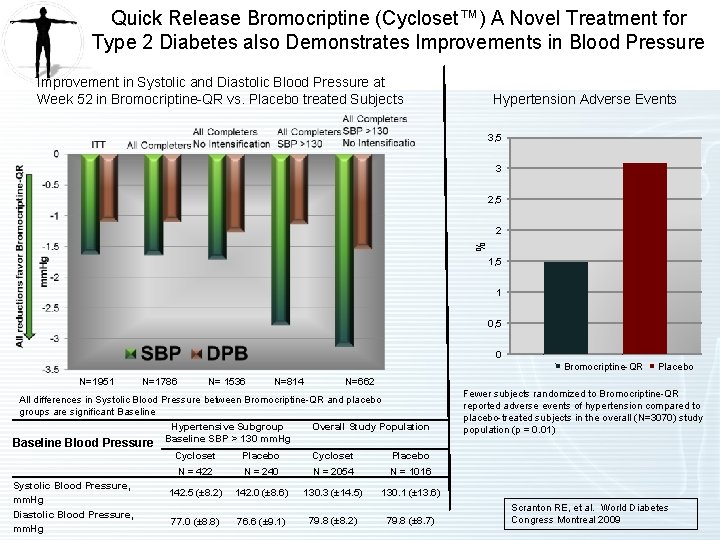
Quick Release Bromocriptine (Cycloset™) A Novel Treatment for Type 2 Diabetes also Demonstrates Improvements in Blood Pressure Improvement in Systolic and Diastolic Blood Pressure at Week 52 in Bromocriptine-QR vs. Placebo treated Subjects Hypertension Adverse Events 3, 5 3 2, 5 % 2 1, 5 1 0, 5 0 Bromocriptine-QR N=1951 N=1786 N= 1536 N=814 N=662 All differences in Systolic Blood Pressure between Bromocriptine-QR and placebo groups are significant Baseline Blood Pressure Systolic Blood Pressure, mm. Hg Diastolic Blood Pressure, mm. Hg Hypertensive Subgroup Baseline SBP > 130 mm. Hg Overall Study Population Cycloset Placebo N = 422 N = 240 N = 2054 N = 1016 142. 5 (± 8. 2) 142. 0 (± 8. 6) 130. 3 (± 14. 5) 130. 1 (± 13. 6) 77. 0 (± 8. 8) 76. 6 (± 9. 1) Placebo 79. 8 (± 8. 2) 79. 8 (± 8. 7) Fewer subjects randomized to Bromocriptine-QR reported adverse events of hypertension compared to placebo-treated subjects in the overall (N=3070) study population (p = 0. 01) Scranton RE, et al. World Diabetes Congress Montreal 2009
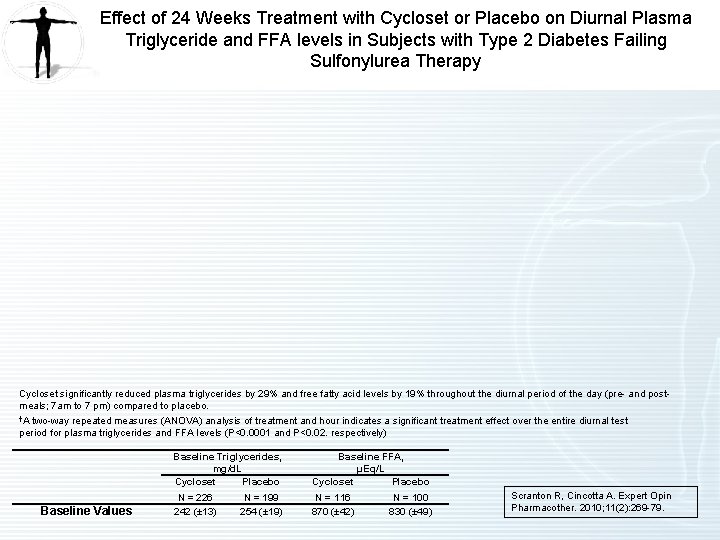
Effect of 24 Weeks Treatment with Cycloset or Placebo on Diurnal Plasma Triglyceride and FFA levels in Subjects with Type 2 Diabetes Failing Sulfonylurea Therapy Cycloset significantly reduced plasma triglycerides by 29% and free fatty acid levels by 19% throughout the diurnal period of the day (pre- and postmeals; 7 am to 7 pm) compared to placebo. †A two-way repeated measures (ANOVA) analysis of treatment and hour indicates a significant treatment effect over the entire diurnal test period for plasma triglycerides and FFA levels (P<0. 0001 and P<0. 02. respectively) Baseline Values Baseline Triglycerides, mg/d. L Cycloset Placebo Baseline FFA, µEq/L Cycloset Placebo N = 226 242 (± 13) N = 116 870 (± 42) N = 199 254 (± 19) N = 100 830 (± 49) Scranton R, Cincotta A. Expert Opin Pharmacother. 2010; 11(2): 269 -79.
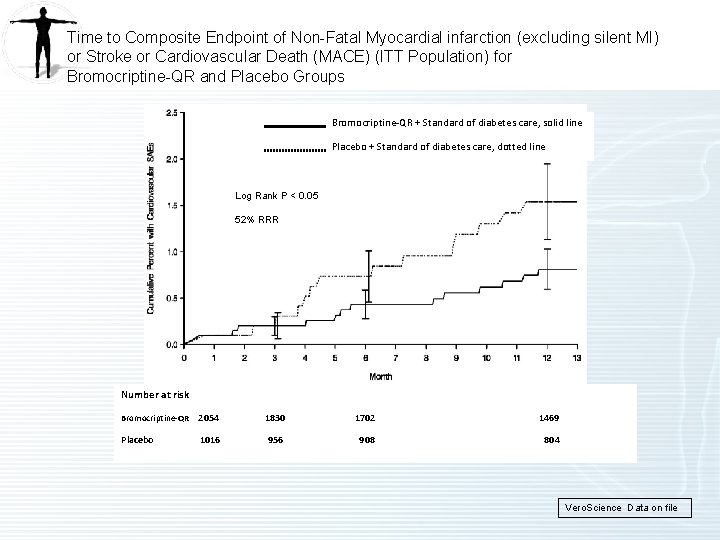
Time to Composite Endpoint of Non-Fatal Myocardial infarction (excluding silent MI) or Stroke or Cardiovascular Death (MACE) (ITT Population) for Bromocriptine-QR and Placebo Groups Bromocriptine-QR + Standard of diabetes care, solid line Placebo + Standard of diabetes care, dotted line Log Rank P < 0. 05 52% RRR Number at risk Bromocriptine-QR 2054 1830 1702 1469 Placebo 1016 956 908 804 Vero. Science Data on file

References (1 of 2) • • • Diagnosis and classification of diabetes mellitus. Diabetes Care. Jan 2010; 33 Suppl 1: S 62 -9. Standards of medical care in diabetes. Diabetes Care. Jan 2012; 35 Suppl S 20. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet. Sep 12 1998; 352(9131): 854 -65. Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002; 346: 393 -403. Gerstein HC, Yusuf S, Bosch J, et al. Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: a randomized controlled trial. Lancet. 2006; 368: 1096 -1105. Orchard TJ, Temprosa M, Goldberg R, et al. The effect of metformin and intensive lifestyle intervention on the metabolic syndrome: the Diabetes Prevention Program randomized trial. Ann Intern Med 2005; 142: 611 -619. Gaziano JM, Cincotta AH, O’Connor CM, et al. Randomized clinical trial of quick-release bromocriptine among patients with type 2 diabetes on overall safety and cardiovascular outcomes. Diabetes Care. 2010 Jul; 33(7): 1503 -8. Pili H, Ohashi S, Matsuda M, et al. Bromocriptine: a novel approach to the treatment of type 2 diabetes. Diabetes Care 2000 Aug; 23(8): 1154 -61. Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes Care. 2006 Aug; 29(8): 1963 -72.

References (2 of 2) • • • Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes Care. 2009 Jan; 32(1): 193 -203. Bolen S, Feldman L, Vassy J, et al. Systematic review: comparative effectiveness and safety of oral medications for type 2 diabetes. Ann Intern Med. 2007 Dec 18; 147(12): 887. Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. 2008 Nov 20; 359(21): 2195 -207.
- Slides: 41