44 Sc production with 18 Me V cyclotron






- Slides: 8

44 Sc production with 18 Me. V cyclotron and study of scandium-labeled peptide based ligands for clinical use Thanks to: MD, Ph. D Maija Radzina Rīga Stradiņš University, Radiology Research laboratory

1. To develop economically efficient middle energy (18 Me. V) cyclotron based technology for production and purification of 44 Sc radionuclide as a medical diagnostic isotope in comparison with generator based technology 2. To provide clinical study platform for 44 Sc radionuclide based imaging in neuroendocrine tumors 3. Theranostic pairing – 44 Sc + 47 Sc and their validation in middle energy cyclotrone based technology 2

44 Sc and 47 Sc as matched pair for molecular imaging • 44 Sc is a promising radionuclide for positron emission tomography (PET/CT) with 177 Lu, 90 Y, or 47 Sc as therapeutic counterparts 1 • 44 Sc • due to the half life (T 1/2 = 3. 92 h) almost 4 times longer than half life of 68 Ga (T 1/2 = 67. 71 min) it is an attractive candidate for development of novel PETradiopharmaceuticals. 47 Sc Mean range in tissue 810 µm 177 Lu 90 Y • 47 Sc – – 670 µm 3900 µm can be utilized in radiotherapy using the same vector molecules 1 Hoehr C. , at. al. 44 g. Sc production using a water target on a 13 Me. V cyclotron, Nuclear Medicine and Biology 41, 2014, 401 – 406; Matched Radionuclide Pairs for Imaging and Therapy (edited by A. Bockish) Eur J Nucl Med Mol Imaging, Vol 38, Suppl 1, June 2011 3

Phase I Develop economicaly efficient cyclotron manufactured 44 Sc as a diagnostic radionuclide and labeling technique 1. 44 Sc liquid and solid target design and adjustment for 44 Sc production with 18 Me. V cyclotron; 2. Production and optimization of 44 Sc production using solid target versus using liquid target; 3. Transfer of crude 44 Sc from cyclotron to “hot lab” from solid target versus from liquid target; 4

4. Obtain pure, noncarrier added 44 Sc by chemical purification versus mass separation with the help of MEDICIS; 6. Develop technology and method for radiolabeling of DOTA conjugated peptide (TOC & TATE) with 44 Sc. Product purification and formulation; 5. Quality check of cyclotron-produced and purified 44 Sc against reference sample of 44 Sc from MEDICIS; 7. Quality control – physical and chemical analysis of 44 Sc-DOTA conjugated peptide-based ligand for cancer diagnostics; Phase I activities may be initiated by the beginning of 2019 5

Phase II Verification of use in clinical medicine 1. Clinical study: • neuroendocrine tumour patients (30 patients in 3 years project) for clinical validation of 44 Sc-DOTATOC • study in other malignancy patient groups (30 patients) for validation of 44 Sc diagnostic radiopharmaceuticals • licenced medical PET/CT facility of RSU Nuclear medicine clinic is able to perform in-human imaging studies and host the clinical study with 44 Sc- DOTA peptides and cancer binding active substances on a neuroendocrine malignancy patient cohort for the proof of concept Phase II activities may be initiated by the beginning of 2020 6

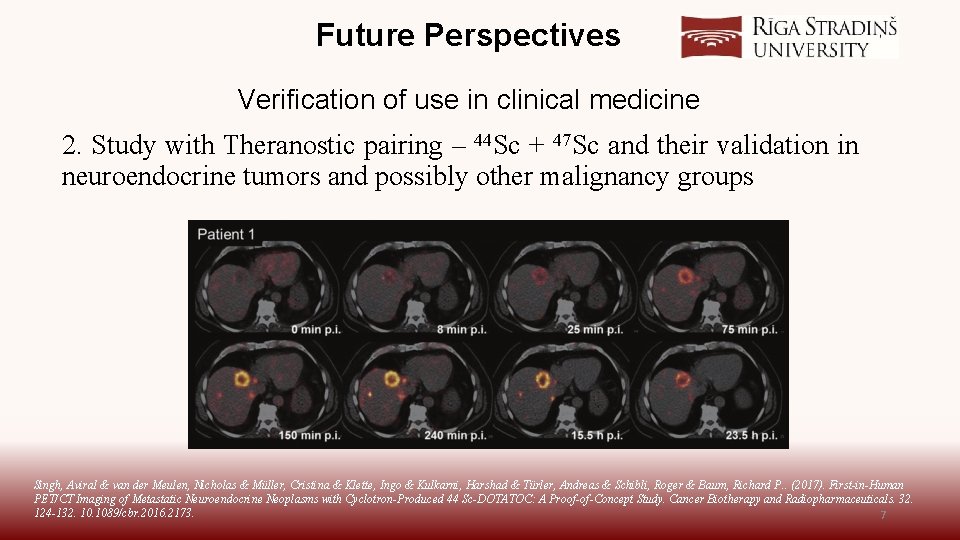
Future Perspectives Verification of use in clinical medicine 2. Study with Theranostic pairing – 44 Sc + 47 Sc and their validation in neuroendocrine tumors and possibly other malignancy groups Singh, Aviral & van der Meulen, Nicholas & Müller, Cristina & Klette, Ingo & Kulkarni, Harshad & Türler, Andreas & Schibli, Roger & Baum, Richard P. . (2017). First-in-Human PET/CT Imaging of Metastatic Neuroendocrine Neoplasms with Cyclotron-Produced 44 Sc-DOTATOC: A Proof-of-Concept Study. Cancer Biotherapy and Radiopharmaceuticals. 32. 124 -132. 1089/cbr. 2016. 2173. 7

For PRISMAS-MAP • Medical and clinical basis • Well established and world class medical expertise and know-how • Infrastructure – Cyclotron 8