4 Lecture 6 Done By Buthainahalmasaeed Batool shafi

4 Lecture #6 Done By : Buthainah-almasaeed & Batool shafi 4 -1
4 Biochemistry by Mary K. Campbell & Shawn O. Farrell 4 -2

4 The Three. Dimensional Structure of Proteins 4 -3
4 Protein Structure 1° structure: the sequence of amino acids in a polypeptide chain, read from the N-terminal end to the C-terminal end Ø Amino acid sequence (1° structure) of a protein determines its 3 D structure which determines its properties and its biological function. ØA striking example of the importance of primary structure is sickle-cell anemia, a disease caused by a change in one amino acid in each of two of the four chains of hemoglobin(Hb. S the β-chain of sickle cell Hb a valine residue has replaced a glutamic acid residue at position 6). 4 -4

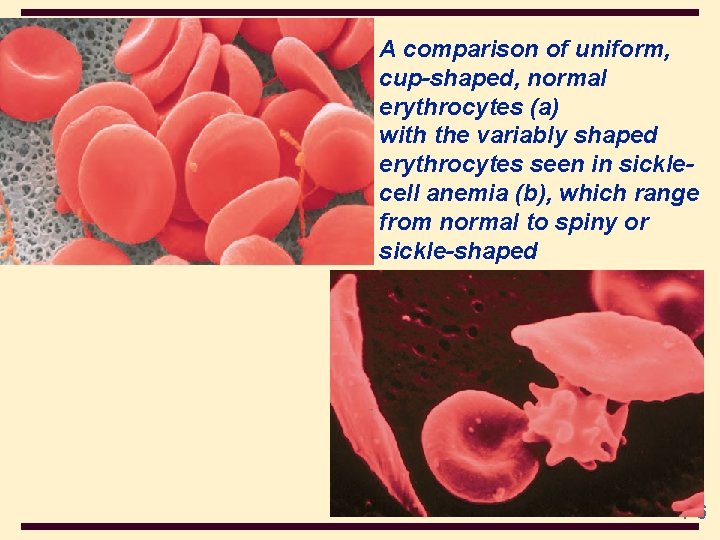
4 A comparison of uniform, cup-shaped, normal erythrocytes (a) with the variably shaped erythrocytes seen in sicklecell anemia (b), which range from normal to spiny or sickle-shaped 4 -6

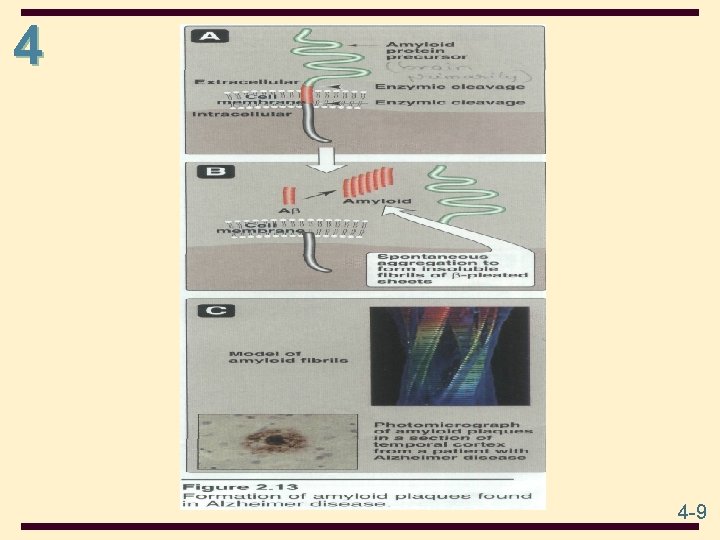
4 2° structure: the ordered 3 -dimensional arrangements (conformations) in localized regions of a polypeptide chain; refers only to H-bonded arrangement of the peptide backbone e. g. , -helix and -pleated sheet In Alzheimer’s disease patients, levels of βamyloid become elevated, and this protein undergoes a conformational transformation from a soluble α helix–rich state to a state rich in β sheet and prone to self-aggregation. 4 -7

4 4 -9

4 Transmissible spongiform encephalopathies (TSEs), including Creutzfeldt-Jakob disease 4 -10

4 -Helix Characteristics: • • • coil of the helix is clockwise or right-handed there are 3. 6 amino acids per turn full turn distance is 5. 4 Å (pitch) each peptide bond is trans and planar C=O of each peptide bond is hydrogen bonded to the N-H of the fourth amino acid away • C=O…. . . H-N hydrogen bonds are parallel to helical axis • all R groups point outward from helix • R groups are not involved in the H-bonds 4 -11
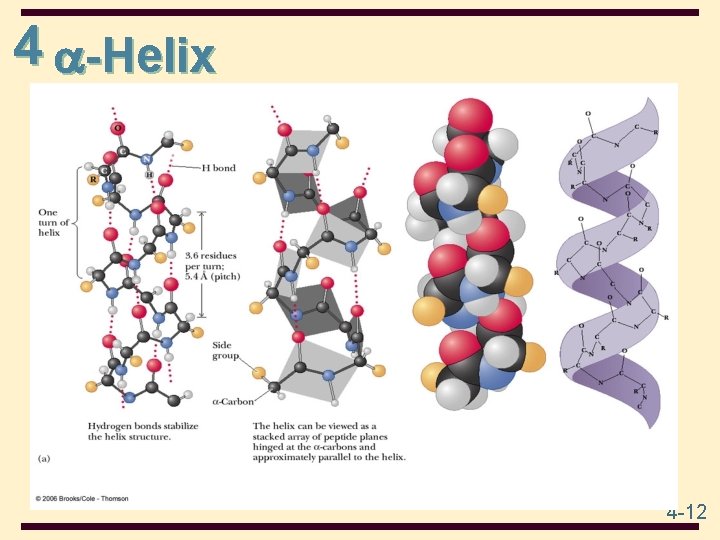
4 -Helix 4 -12

4 -Pleated Sheet • polypeptide chains are folded back on itself • polypeptide chains lie adjacent to one another; may be parallel or antiparallel • R-groups alternate, first above and then below plane • each peptide bond is trans and planar “straight lines” • C=O and N-H groups of each peptide bond are perpendicular to axis of the sheet • C=O…. . . H-N hydrogen bonds are between adjacent sheets and perpendicular to the direction of the sheet 4 -13
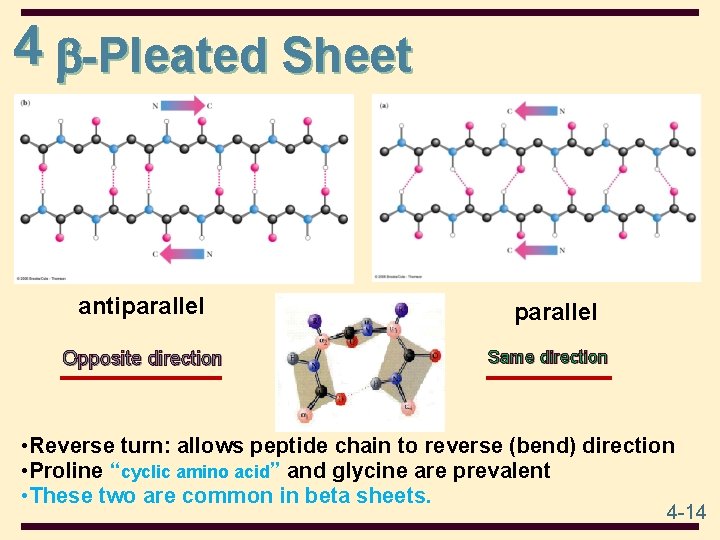
4 -Pleated Sheet antiparallel Opposite direction parallel Same direction • Reverse turn: allows peptide chain to reverse (bend) direction • Proline “cyclic amino acid” and glycine are prevalent • These two are common in beta sheets. 4 -14

4 • Tertiary (3°) structure: the 3 D arrangement in space of all atoms in a polypeptide chain • Bonds stabilize the 3° structure: Metal ion coordination, side chain H-bond, electrostatic attraction, disulfide bond, and hydrophobic interaction • Quaternary (4°) structure: the association of polypeptide chains. • Proteins are divided into two large classes based on their three-dimensional structure. Protein Classifications according to shape : (another classification) • fibrous proteins • globular proteins 4 -15

4 Fibrous Proteins • Fibrous proteins: contain polypeptide chains organized approximately parallel along a single axis. They • consist of long fibers or large sheets • tend to be mechanically strong • are insoluble in water and dilute salt solutions • Play an important structural roles in nature 4 -16

4 Secondary Structures and Properties of Fibrous Proteins Structure Characteristics α-Helix, cross-linked Tough, insoluble by disulfide protective structures of varying hardness and flexibility β-Conformation Soft, flexible Collagen triple helix filaments Silk fibroin High tensile strength, without stretch Examples of occurrence Keratin of hair, feathers, and nails Silk fibroin Collagen of tendons, bone matrix 4 -17

4 Collagen triple helix fibrous protein 3 polypeptide chains Collagen has an unusual amino acid composition and sequence: Glycine is found at almost every third residue ﻳﺴﺎﻋﺪ ﻋﻠﻰ ﺍﻟﺘﺼﺮﻳﻒ ﺑﺎﻟﻜﻠﻴﺔ Proline (Pro) makes up about 17% of collagen Collagen contains two uncommon derivative amino acids not directly inserted during translation. These amino acids are found at specific locations relative to glycine and are modified posttranslationally by different enzymes, both of which require vitamin C as a cofactor. 4 -18

4 Hydroxyproline (Hyp), derived from proline. Hydroxylysine (Hyl), derived from lysine (Lys). Depending on the type of collagen, varying numbers of hydroxylysines are glycosylated (mostly having disaccharides attached). Cortisol stimulates degradation of (skin) collagen into amino acids. 4 -19

4 • The best-known defect in collagen biosynthesis is Scurvy, a result of a dietary deficiency of vitamin C required by prolyl and lysyl hydroxylases. • The resulting deficit in the number of hydroxyproline and hydroxylysine residues undermines the conformational stability of collagen fibers, leading to bleeding gums, swelling joints, poor wound healing, and ultimately to death. • Menkes’syndrome, characterized by kinky hair and growth retardation, reflects a dietary deficiency of the copper required by lysyl oxidase, which catalyzes a key step in formation of the covalent cross-links that strengthen collagen fibers. `` • 4 -20

4 Genetic disorders of collagen biosynthesis include : • several forms of osteogenesis imperfecta, characterized by fragile bones. In Ehlers-Dahlos syndrome, a group of connective tissue disorders that involve impaired integrity of supporting structures, defects in the genes that encode α collagen-1, procollagen N-peptidase, or lysyl hydroxylase result in mobile joints and skin abnormalities. 4 -21
4 4 -22

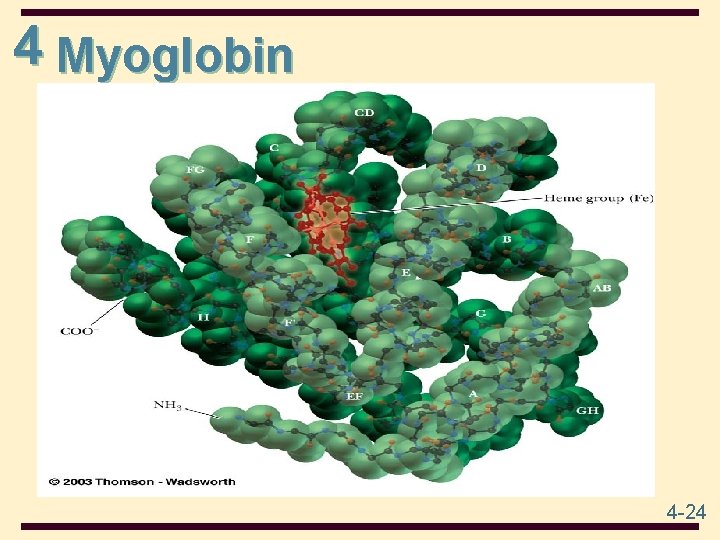
4 Globular Proteins • Globular proteins: proteins which are folded to a Globular proteins more or less spherical shape • they tend to be soluble in water and salt solutions • most of their polar side chains are on the outside and interact with the aqueous environment by hydrogen bonding and ion-dipole interactions • most of their nonpolar side chains are buried inside • nearly all have substantial sections of -helix and -sheet • Example • Myoglobin 4 -23
4 Myoglobin 4 -24

4 Myoglobin • • Function in O 2 storage in muscles a single polypeptide chain of 153 amino acids 8 polypeptide chains Compact with a single heme group in a hydrophobic pocket 8 regions of -helix; no regions of -sheet most polar side chains are on the surface nonpolar side chains are folded to the interior two His side chains are in the interior, involved with interaction with the heme group 4 -25

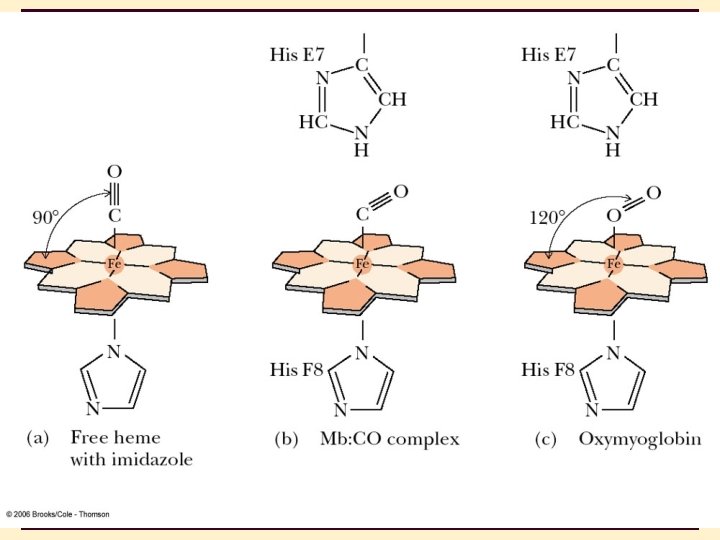
4 Heme is a prosthetic group made from one protoporphyrin ring and iron in the center protoporphyrin ring is made from 4 pyrrole rings Fe(II) of heme has 6 coordinates sites; 4 sites interact with N atoms of protoporphyrin , 1 with N of a His side chain, and 1 with either an O 2 molecule or an N of the second His side chain 4 -26
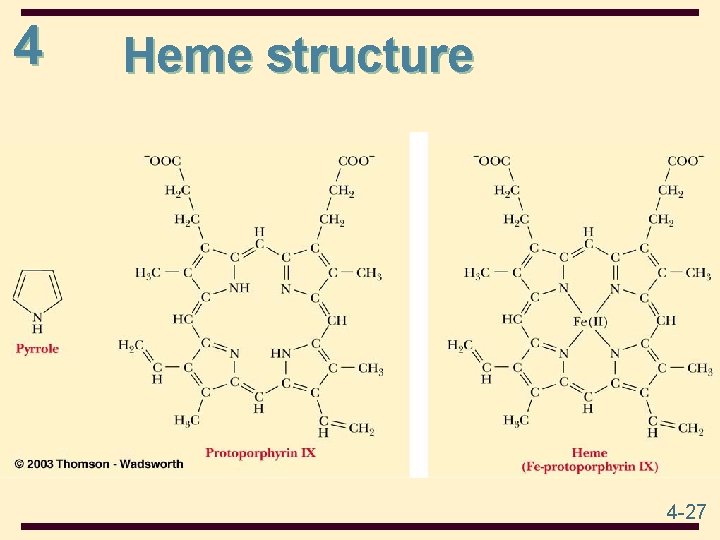
4 Heme structure 4 -27
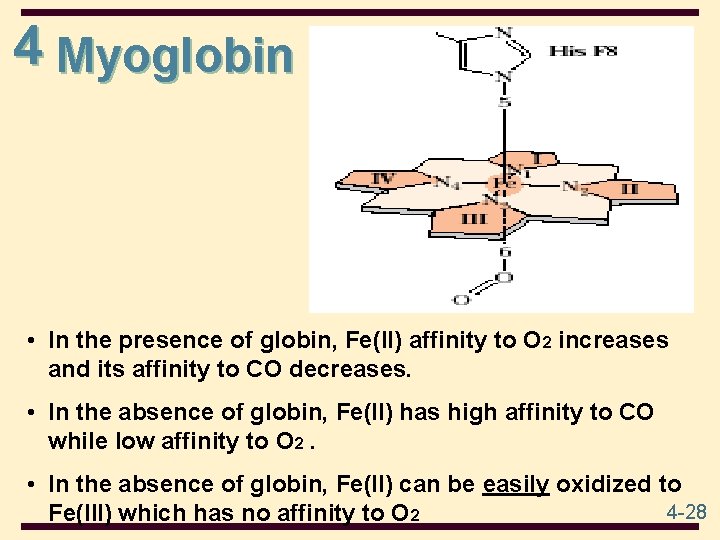
4 Myoglobin • In the presence of globin, Fe(II) affinity to O 2 increases and its affinity to CO decreases. • In the absence of globin, Fe(II) has high affinity to CO while low affinity to O 2. • In the absence of globin, Fe(II) can be easily oxidized to 4 -28 Fe(III) which has no affinity to O 2
4 4 -29

4 Quaternary Structure • Quaternary (4°) structure: the association of polypepetide monomers into multisubunit proteins (dimer, trimer, tetramer, etc) • examples we will see in this course 4 -30
4 Ouaternary str is tow monemer jount together to • make multisubunit The last example in rhe table (insulin) >>6 • subunite is not true Insulin is 2 polypeptied chain for -chains • 4 -31
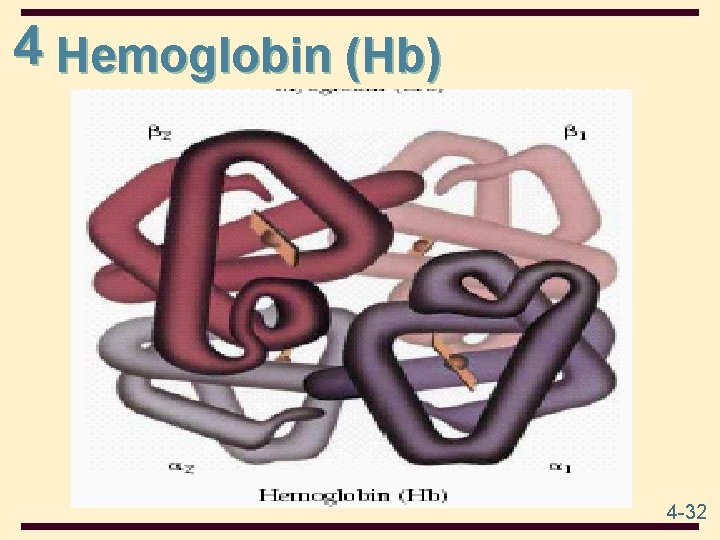
4 Hemoglobin (Hb) 4 -32

4 Oxygen Binding of Hb • Hb is a tetramer of two -chains (141 amino acids each) and two -chains (153 amino acids each); 2 2 • each chain has 1 heme group; hemoglobin can bind up to 4 molecules of O 2 • binding is cooperative; when one O 2 is bound, it becomes easier for the next O 2 to bind (positive cooperativity) • the function of hemoglobin is to transport oxygen • the structure of oxygenated Hb (loaded) is different from that of unoxygenated Hb (unloaded) • H+, CO 2, Cl-, and 2, 3 -bisphoglycerate (BPG) affect the ability of Hb to bind and transport oxygen 4 -33
4 4 -34 Fig. 4 -28, p. 106
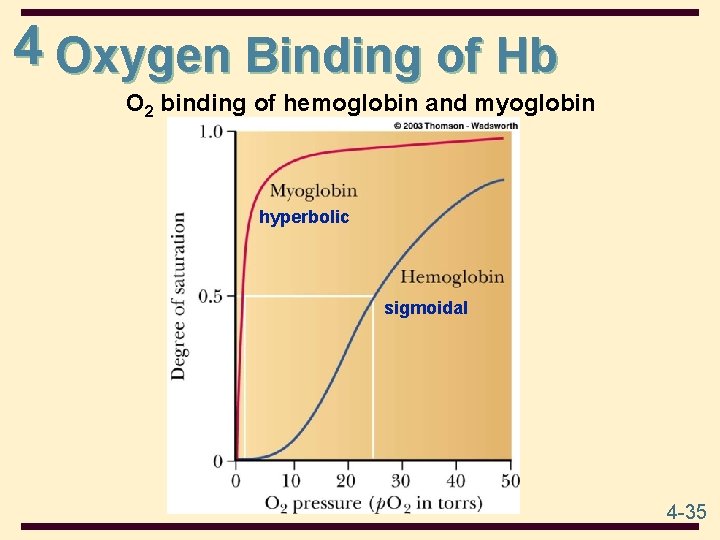
4 Oxygen Binding of Hb O 2 binding of hemoglobin and myoglobin hyperbolic sigmoidal 4 -35

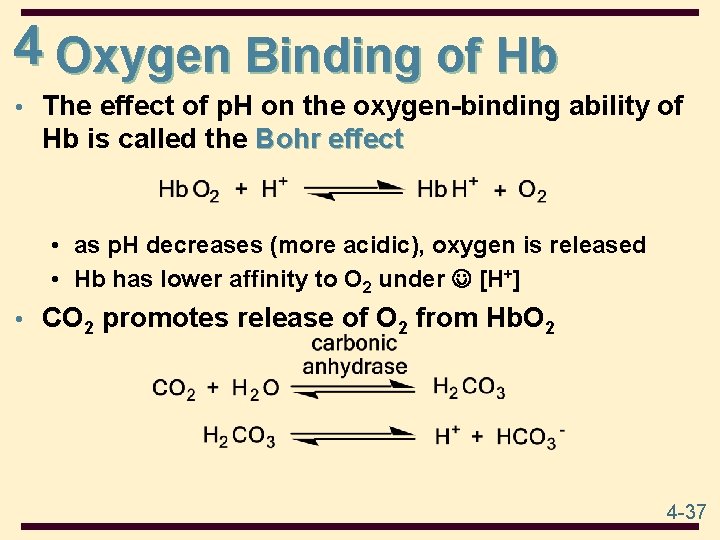
4 Oxygen Binding of Hb • The effect of p. H on the oxygen-binding ability of Hb is called the Bohr effect • as p. H decreases (more acidic), oxygen is released • Hb has lower affinity to O 2 under [H+] • CO 2 promotes release of O 2 from Hb. O 2 4 -37
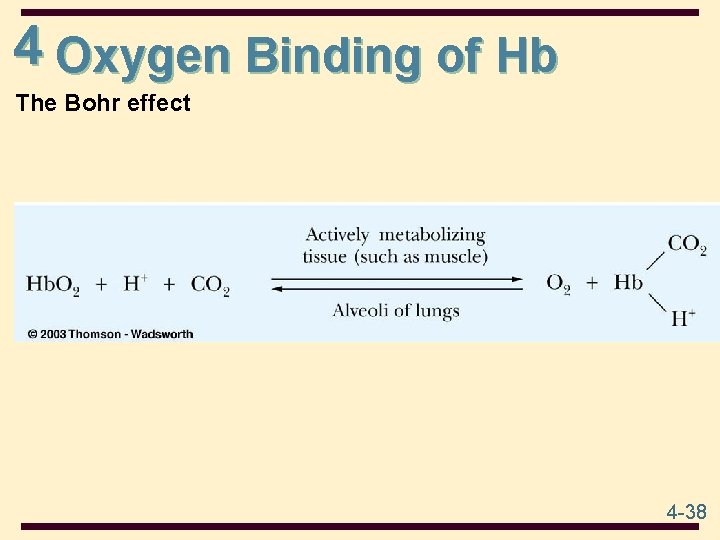
4 Oxygen Binding of Hb The Bohr effect 4 -38

4 Oxygen Binding of Hb Summary of the Bohr effect Lungs Actively Metabolizing Muscle Higher p. H than actively Lower p. H due to production of H+ metabolizing tissue Hemoglobin binds O 2 Hemoglobin releases H+ Hemoglobin binds H + 4 -39

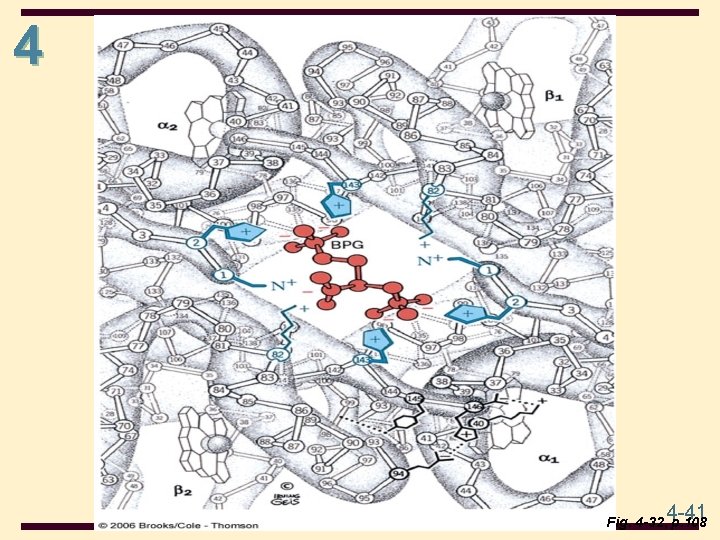
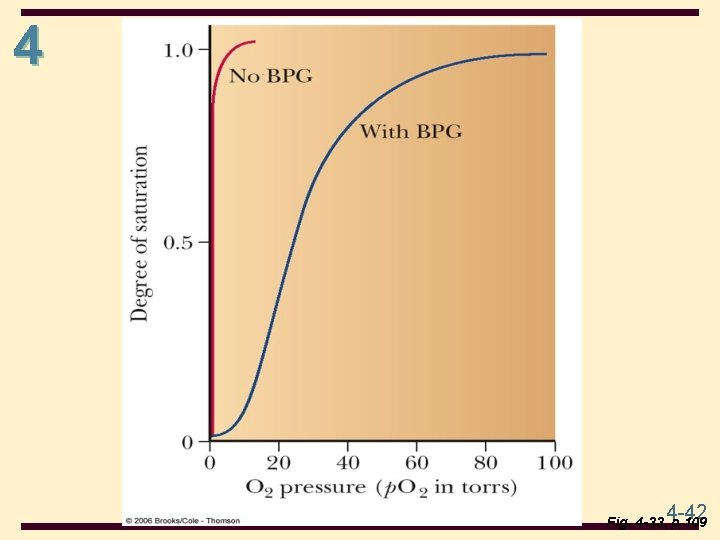
4 Hemoglobin (Hb) • Hemoglobin in blood is bound to BPG • interaction is electrostatic, between negative charges on BPG and positive side chains (e. g. , Lys, Arg) of hemoglobin • BPG promotes O 2 dissociation • Hb stripped of BPG remains saturated with O 2 4 -40
4 4 -41 Fig. 4 -32, p. 108
4 4 -42 Fig. 4 -33, p. 109
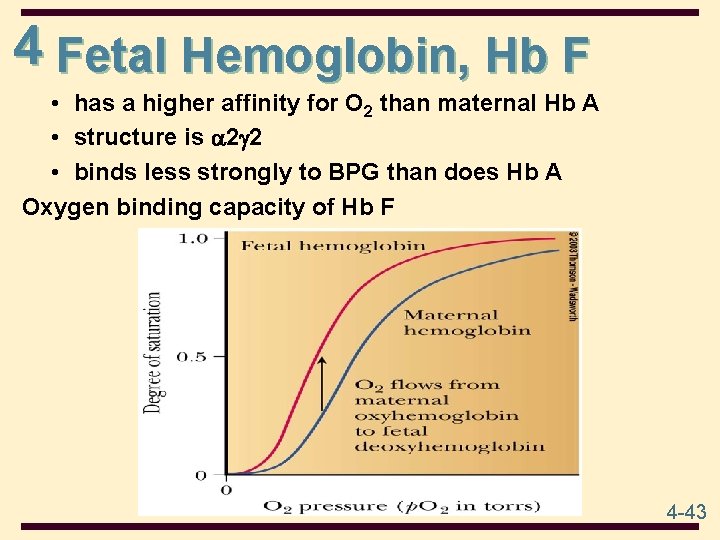
4 Fetal Hemoglobin, Hb F • has a higher affinity for O 2 than maternal Hb A • structure is 2 g 2 • binds less strongly to BPG than does Hb A Oxygen binding capacity of Hb F 4 -43

4 Adaptation to High Altitude • Physiologic changes that accompany prolonged exposure to high altitude include an increase in the number of erythrocytes and in their concentrations of hemoglobin and of BPG. • Elevated BPG lowers the affinity of Hb. A for O 2 (decreases P 50), which enhances release of O 2 at the tissues. 4 -44

4 Initial inefficient response to low oxygen pressure • There is an increase in breathing and heart rate to as much as double, even while resting. Pulse rate and blood pressure go up sharply as our hearts pump harder to get more oxygen to the cells. These are stressful changes, especially for people with weak hearts. 4 -45

4 • Later, a more efficient response normally develops as acclimatization takes place. Additional red blood cells and capillaries are produced to carry more oxygen. The lungs increase in size to facilitate the osmosis of oxygen and carbon dioxide. There is also an increase in the vascular network of muscles which enhances the transfer of gases. 4 -46
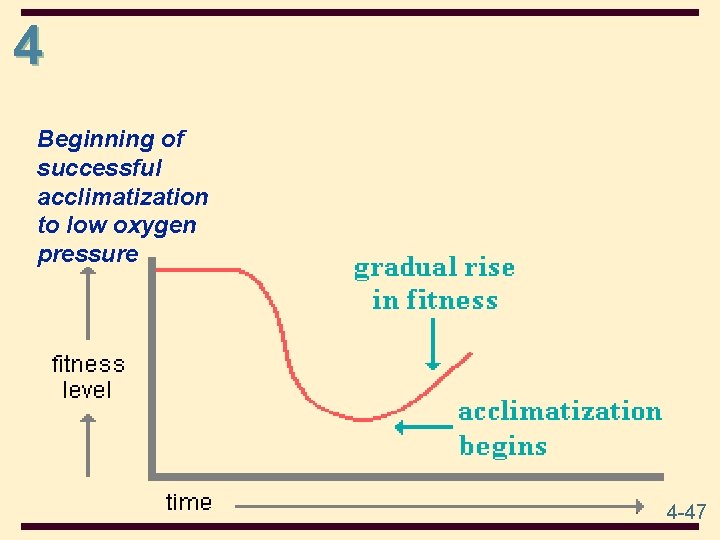
4 Beginning of successful acclimatization to low oxygen pressure 4 -47

4 • However, successful acclimatization rarely results in the same level of physical and mental fitness that was typical of altitudes close to sea level. Strenuous exercise and memorization tasks still remain more difficult. In addition, the rate of miscarriages is usually higher at altitudes above two miles because fetuses receive less oxygen from their mothers. 4 -48
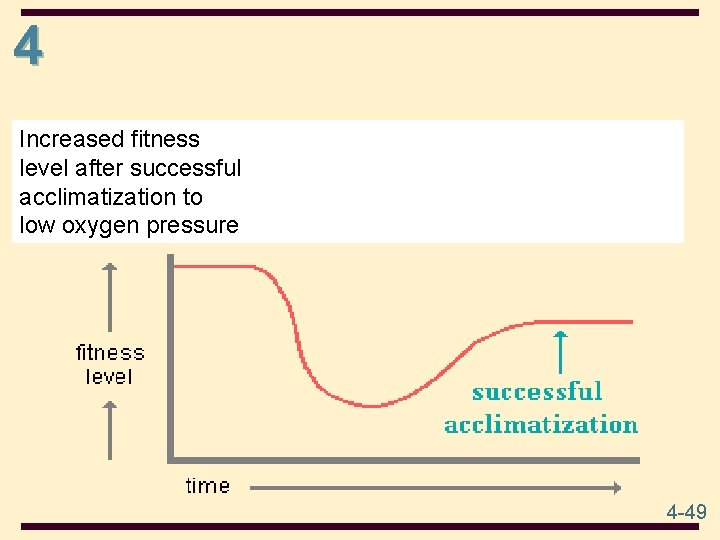
4 Increased fitness level after successful acclimatization to low oxygen pressure 4 -49

4 • On returning to sea level after successful acclimatization to high altitude, the body usually has more red blood cells and greater lung expansion capability than needed. Since this provides athletes in endurance sports with a competitive advantage, the U. S. maintains an Olympic training center in the mountains of Colorado. Several other nations also train their athletes at high altitude for this reason. However, the physiological changes that result in increased fitness are short term at low altitude. In a matter of weeks, the body returns to a normal fitness level. 4 -50
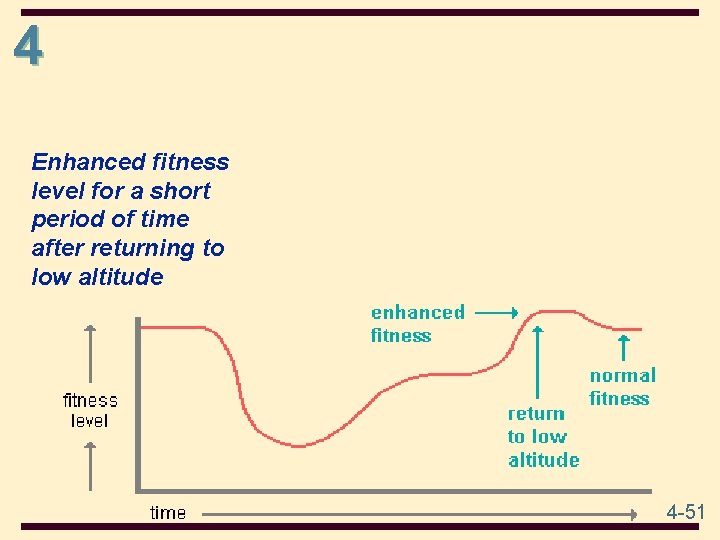
4 Enhanced fitness level for a short period of time after returning to low altitude 4 -51

4 BIOMEDICAL IMPLICATIONS Myoglobinuria • Following massive crush injury, myoglobin released from damaged muscle fibers colors the urine dark red. • Myoglobin can be detected in plasma following a myocardial infarction. Anemias • Anemias, reductions in the number of red blood cells or of hemoglobin in the blood, can reflect impaired synthesis of hemoglobin (eg, in iron deficiency; or impaired production of erythrocytes (eg, in folic acid or vit. B 12 def. ) 4 -52

4 Glycosylated Hemoglobin (Hb. A 1 c) When blood glucose enters the erythrocytes it glycosylates the amino group of lysine residues and the amino terminals of hemoglobin. The fraction of hemoglobin glycosylated, normally about 5%, is proportionate to blood glucose concentration. Since the half-life of an erythrocyte is typically 60 days, the level of glycosylated hemoglobin (Hb. A 1 c) reflects the mean blood glucose conc. over the preceding 6– 8 weeks. Measurement of Hb. A 1 c therefore provides valuable information for management of diabetes mellitus. 4 -53

4 Hemoglobin (Hb. S) • In sickle cell, Val replaces the β 6 Glu of Hb. B, creating a “sticky patch” that has a complement on deoxy. Hb (but not on oxy. Hb). • Deoxy. Hb. S polymerizes at low O 2 concentrations, forming fibers that distort erythrocytes into sickle shapes. • Alpha and beta thalassemias are anemias that result from reduced production of α and β subunits of Hb. A, respectively 4 -54

4 Factors Directing Folding • Noncovalent interactions, such as • hydrogen bonding between polar side chains, e. g. , Ser and Thr • hydrophobic interaction between nonpolar side chains, e. g. , Val and Ile • electrostatic attraction between side chains of opposite charge, e. g. , Lys and Glu • electrostatic repulsion between side chains of like charge, e. g. , Lys and Arg, Glu and Asp • Formation of disulfide (-S-S-) covalent bonds between side chains of cysteines 4 -55

4 Denaturation • Denaturation: the unfolding of the protein the loss of biological activity • Native protein (active) denatured protein (inactive) • Denaturation can be brought about by 1. Heat 2. Large changes in p. H, which alter charges on side chains, e. g. , -COO- to -COOH or -NH + to -NH 3. Detergents such as sodium dodecyl sulfate (SDS) which disrupt hydrophobic interactions 4. Urea or guanidine HCL, which form H-bonds with protein that are stronger than those within the protein disrupt H-bonding 5. Mercaptoethanol, which reduces disulfide bonds. 4 -56
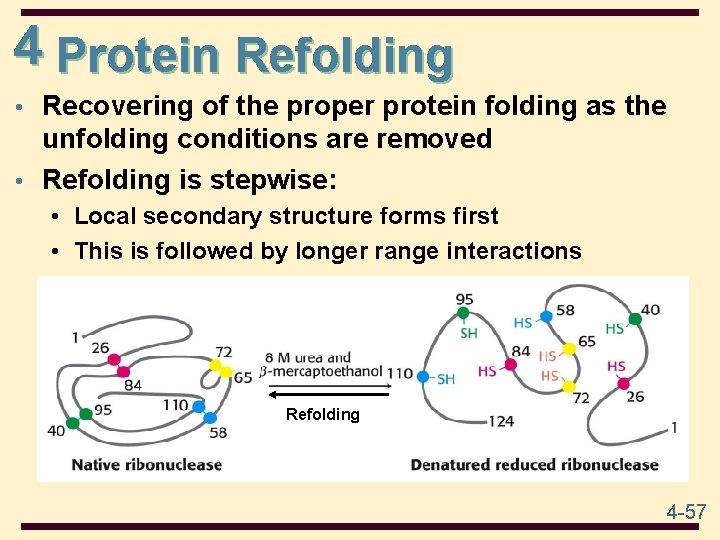
4 Protein Refolding • Recovering of the proper protein folding as the unfolding conditions are removed • Refolding is stepwise: • Local secondary structure forms first • This is followed by longer range interactions Refolding 4 -57
4 End 4 -58
- Slides: 58