4 Furanocoumarin Psoralen R 1 R 2 H






















- Slides: 43

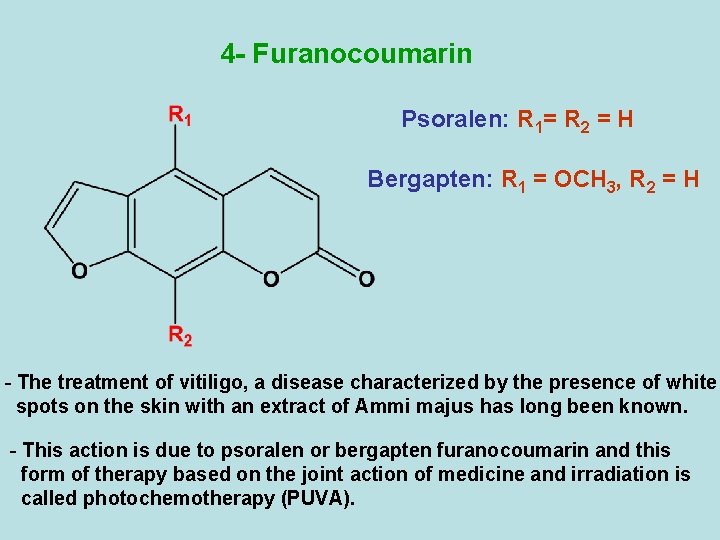
4 - Furanocoumarin Psoralen: R 1= R 2 = H Bergapten: R 1 = OCH 3, R 2 = H - The treatment of vitiligo, a disease characterized by the presence of white spots on the skin with an extract of Ammi majus has long been known. - This action is due to psoralen or bergapten furanocoumarin and this form of therapy based on the joint action of medicine and irradiation is called photochemotherapy (PUVA).

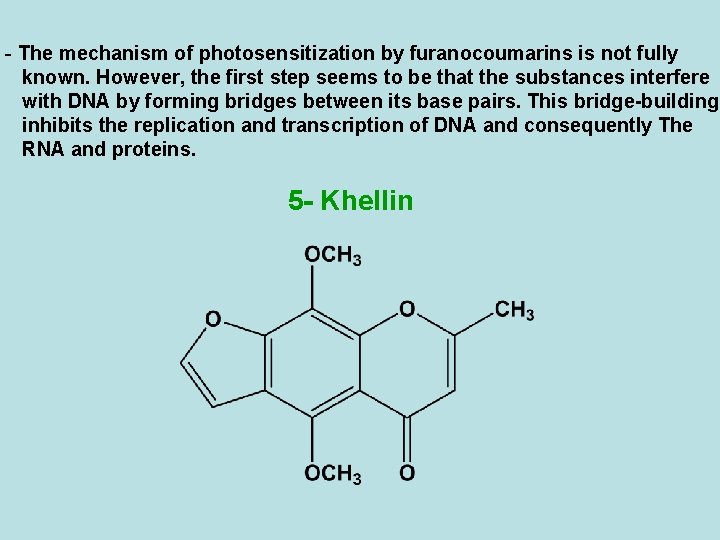
- The mechanism of photosensitization by furanocoumarins is not fully known. However, the first step seems to be that the substances interfere with DNA by forming bridges between its base pairs. This bridge-building inhibits the replication and transcription of DNA and consequently The RNA and proteins. 5 - Khellin

- Khellin is structurally related to the furanocoumarins but is a derivative of 5, 6 -benzo- 4 -pyrone (γ-chromone), rather than 5, 6 -benzo-2 -pyrone (α-chromone). - Its biosynthesis is however very different. - Khellin is present in Ammi visnaga fruit and has antispasmodic properties and was used to ease cramps in the stomach intestines, and urinary tract and for treatment of angina pectoris and asthma (visnagin). Nowadays, it is little used because of its unpleasent side effects as nausea and vomiting.

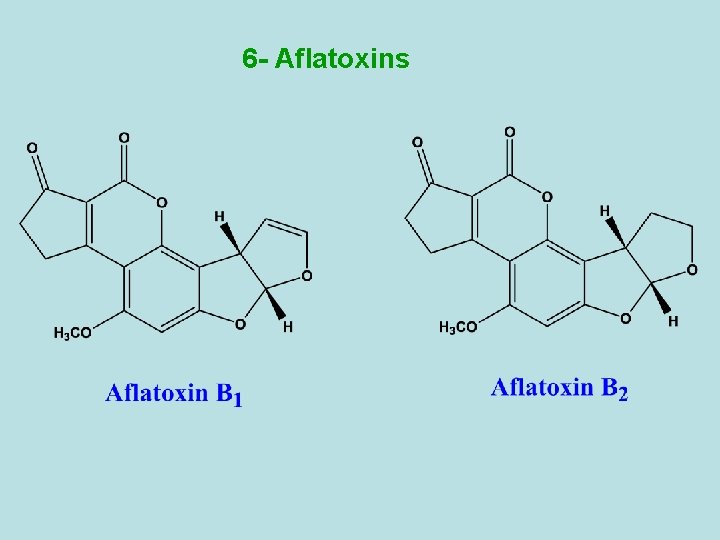
6 - Aflatoxins

- They are fungal metabolites produced by the moulds Aspergillus flavus which grow on foodstuffs such as cereals, beans and peas, coconuts. - Because of the high toxicity of the aflatoxins, food which are susceptible to mould infection must be carefully monitored for the presence of aflatoxins. - The aflatoxin family comprises eight compounds, aflatoxin B 1, B 2, G 1, G 2, M 1, M 2, GM 1 and GM 2 - They are highly poisonous substances which damage the liver. They are also carcinogenic especially aflatoxin B 1.

Cyanophore glycosides

- Many plants contain glycosides that form hydrogen cyanide on hydrolysis and are therefore known as cyanogenic glycosides. . Detaction of cyanogenic glycosides 1 - Guignard´s reaction - When the drug is warmed with water at 40°C or boiled with acidulated water, HCN is liberated and converted sodium picrate (yellow) to sodium picramate (red colour). 2 - Drug + Alcoholic KOH + Fe. SO 4 + Fe. Cl 3 then add HCl soln. , a prussian blue colour appeared. 3 - Drug + mercurous nitrate give black ppt. (Hg).

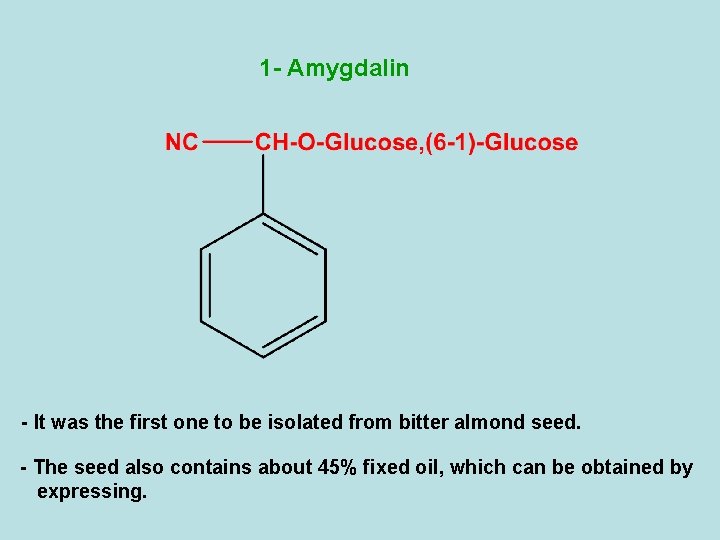
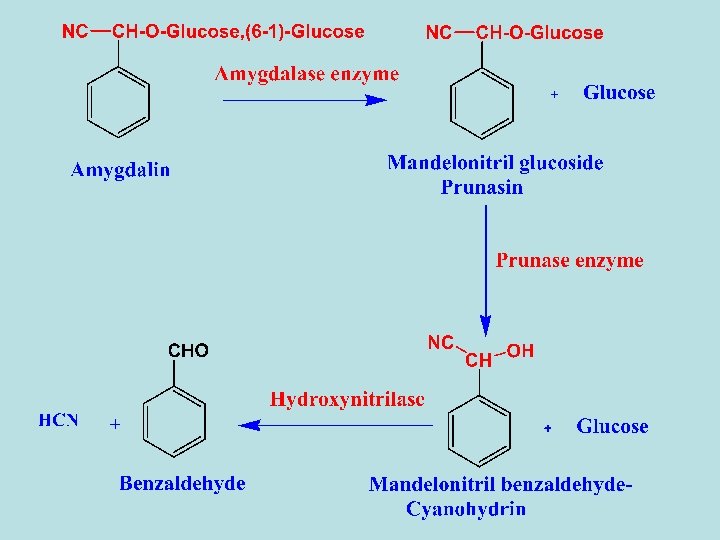
1 - Amygdalin - It was the first one to be isolated from bitter almond seed. - The seed also contains about 45% fixed oil, which can be obtained by expressing.

- The residue left after expressing the fixed oil gives bitter almond oil on steam distillation. - Bitter almond oil consists mainly of benzaldehyde but it also contain 2 -4 % HCN. - Bitter almond and bitter almond oil are used as spices, but this use is not entirely safe because of the hydrogen cyanide content.

1 - Enzymatic hydrolysis - Almonds contain a mixture of enzymes called emulsin which consists of amygdalin hydrolase, prunasin hydrolase and Hydroxynitrilase. When bitter almond are crushed in the presence of water amygdalin hydrolysis in catalyst of these enzymes to yield glucose, hydrogen cyanide and benzaldehyde. 2 - Acidic Hydrolysis - By acid hydrolysis amygdalin yields benzaldehyde, 2 molcules of β-DGlucose and 1 mole of HCN.


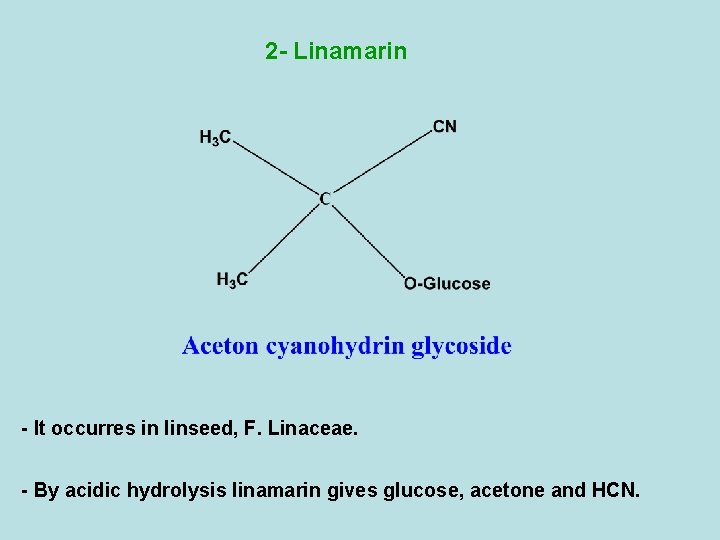
2 - Linamarin - It occurres in linseed, F. Linaceae. - By acidic hydrolysis linamarin gives glucose, acetone and HCN.

Thioglycosides Sulphonated glycosides

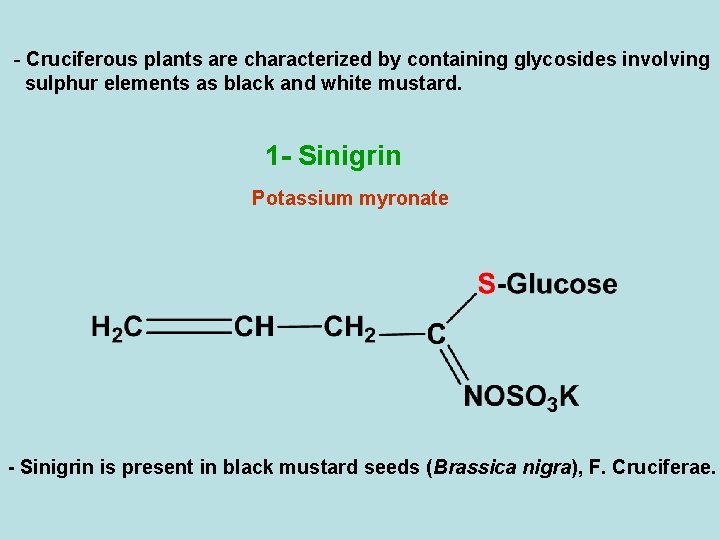
- Cruciferous plants are characterized by containing glycosides involving sulphur elements as black and white mustard. 1 - Sinigrin Potassium myronate - Sinigrin is present in black mustard seeds (Brassica nigra), F. Cruciferae.

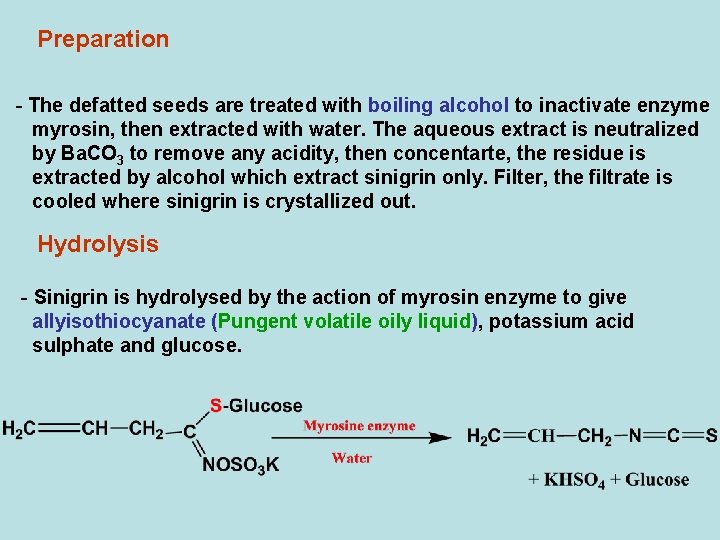
Preparation - The defatted seeds are treated with boiling alcohol to inactivate enzyme myrosin, then extracted with water. The aqueous extract is neutralized by Ba. CO 3 to remove any acidity, then concentarte, the residue is extracted by alcohol which extract sinigrin only. Filter, the filtrate is cooled where sinigrin is crystallized out. Hydrolysis - Sinigrin is hydrolysed by the action of myrosin enzyme to give allyisothiocyanate (Pungent volatile oily liquid), potassium acid sulphate and glucose.

Uses - It is used as locally as counter irritant. - It is used for treatment of rheumatism. - Used as condiment. Chemical tests - Allylisothiocyanate + mixture of (sodium nitroprusside, NH 4 OH + Br 2) gives a blue spots of thiourea. - Allylisothiocyanate + Ag. NO 3 + K 2 Cr 2 O 7 gives yellow spots against red back ground.

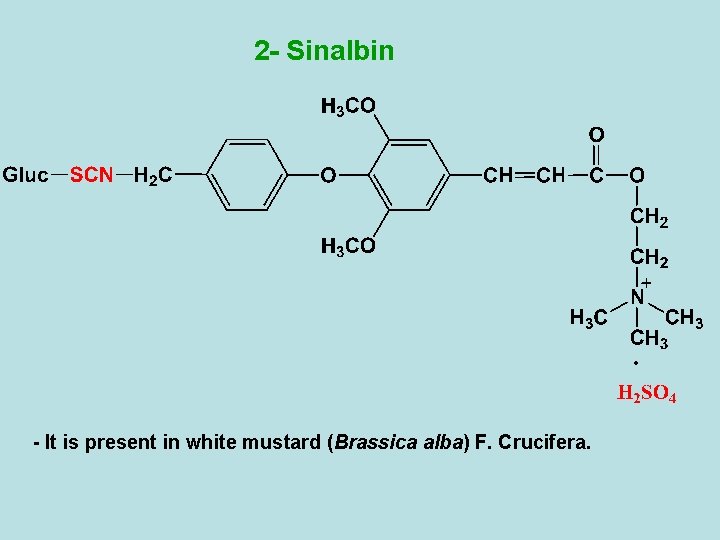
2 - Sinalbin - It is present in white mustard (Brassica alba) F. Crucifera.


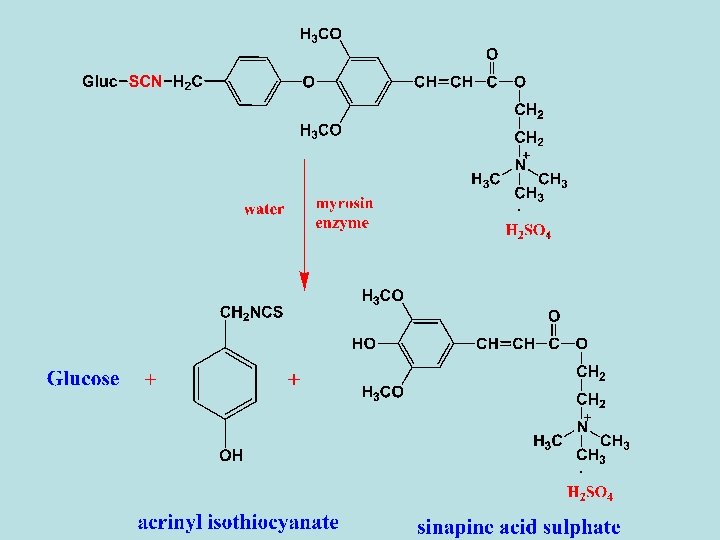
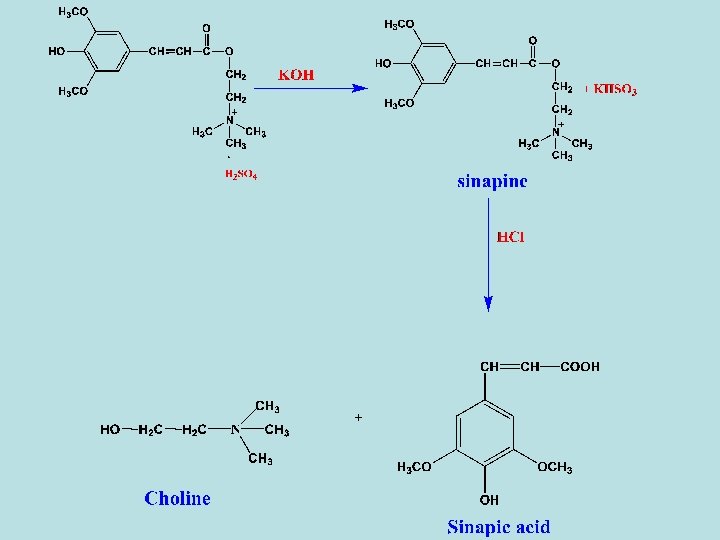
- Enzymatic hydrolysis of sinalbin with myrosin enzyme in the presence of water gives acrinyl isothiocyanate (non-volatile oily liquid), sinapine acid sulphate and glucose. - Sinapine acid sulphate can be converted to Uses - The same as sinigrin. cholin as following


Cardiac glycosides

- Cardiac glycosides are a group of natural products characterized by their specific effect on myocardial contraction and atrioventricular conduction. - In large dose they are toxic and bring about cardiac arrest in systol, but in lower dose thay are important drugs in the treatment of congestive heart failure. - Cardiac glycosides have a direct carditonic action on the myocardium, resulting in an increase in the force of contraction. - The increased contractility is caused by inhibition of the membranebound sodium-potassium activated ATPase, leading to an increase in the intracellular stores of calcium.

- They are widely distributed in some families as apocyanaceae as strophanthus seed or Scrophulariaceae as digitalis leaves or Liliaceae as squill. - Chemically or structurally, the aglycones of cardiac glycosides are steroids, which is a group of tetracyclic saturated secondray alcohol in which the side chain is an unsaturated lactone ring, which can be either 5 - or 6 -membered attached to C-17 of the steroidal skeleton. - The various aglyones differ mainly in the numbers and positions of the hydroxyl substituents and in the extent of oxidation of the C-19 methyl group. - Almost all the known aglycones have two β-orinted hydroxyl groups, one at C-3 and the other at C-14.


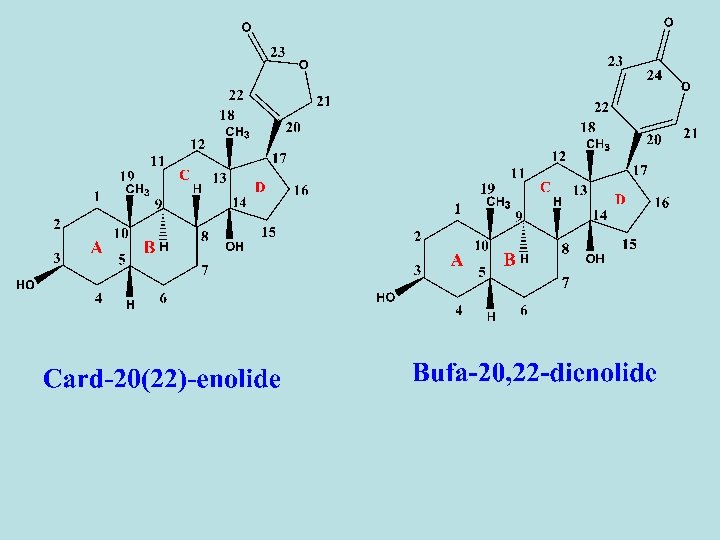
- On the basis of the lacton-ring structure, theses aglycones classify into two groups, A- Cardenolides in which the lactone ring is five membered, and consequently the aglycone contain 23 carbons. As digitalis glycosides. Their absorbance are at 220 nm in UV. B- Bufadienolides in which the lactone ring is six membered, and consequently the aglycone contain 24 carbons. As squill glycosides. Their absorbance are at 300 nm in UV. - The structural activity of cardiac glycosides depends on: 1 - Nature of aglycone part (No. Of OH groups). 2 - nature of lactone ring (5 or 6 membered ring) 3 - Presence or absence of double bonds in the lactone part. 4 - Sugar moiety.

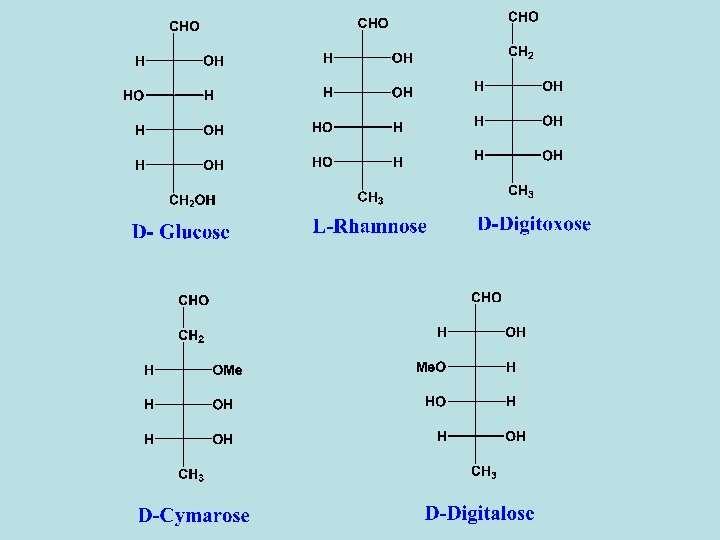
Sugars of cardiac glycosides - The activity of the cardiac glycosides is considerably modified by the sugar moiety. - Combination of sugar with the aglycone increases both the potency and toxicity of the active principle. - The sugar may affect certain physical properties such as water solubility and diffusion througn semipermeable membranes and consequently the rate of absorption and transportion of the compound to the site of action. - Some of the sugar units found in cardiac glycosides are normal as glucose or methyl pentose as rhamnose and others are deoxy sugar as digitoxose, cymarose and sarmentose which are 2 -deoxy sugars. In some sugars the C-3 hydroxyl group is methylated as cymarose, digitalose and sarmentose


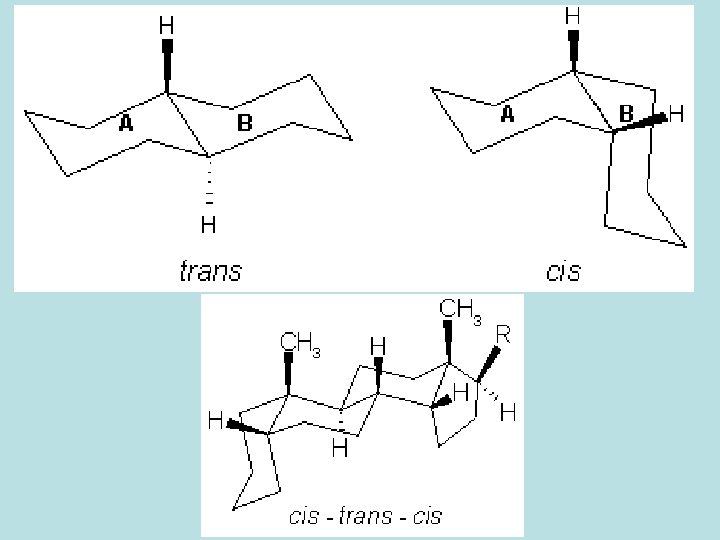
- The sugar moiety is attached almost via the C-3 hydroxyl group of the aglycone. Notes on the activity. - Opening of the lactone ring renders the glycoside inactive. - If the C-3 hydroxyl group is α-orinted, the effect is weaker than that of the β-isomer. - The activity is strongest if the 17 -lactone ring is β-orinted and the A/B, C/D ring junctions are both cis.


Colour test for cardiac glycoside 1 - Liebermann test (for sterols) - One drop of conc. H 2 SO 4 is added to the solution of the glycoside in glacial acetic acid. A change in colour from red to violet to blue and finally green colour appear. 2 - Legal test (for unsaturated lactone ring) - A small quantity of the glycoside is dissolved in a few drops of pyridine. A drop of 2% sodium nitroprussid and a drop of 20% Na. OH solution are then added, a deep red colour constituentes a positive test.

3 - Antimony tricholride test (for both cardenolides or bufadienolides) - Cardiac glycosides + antimony trichloride (in CHCl 3) gives blue or violet colour. 4 - Baljet test - Cardiac glycosides or aglycone + baljet Reagent (1% aqueous solution of picric acid and 10% Na. OH) gives orange or red colour. 5 - keller killiani test (for 2 -Deoxy sugars) - The glycoside is dissolved in glacial acetic acid containing trace of Fe. Cl 3. Conc. H 2 SO 4 containing the same amount of Fe. Cl 3 is added to form the lower layer. An intense blue colour develops at the surface between the two layer.

6 - Raymond´s test (for active CH 2 (C-21) in the cardenolide) - A small amount of glycoside in 1 ml 50% alcohol + 0. 1 ml 1% m-dinitrobenzene (in ethanol) + 2 -3 drops of 20% Na. OH soln. , a violet or blue colour is appeared. 7 - Kedd´s reagent (modified Raymond reagent) - Kedd´s reagent formed of equal volumes of 2% m-dinitrobenzene (in ethanol) and 5. 7% (w/v) aqueous solution of KOH. - Alc. Extract + Kedd´s reagent gives blue or violet colour. - The colour fades in 1 -2 hours, and this reagent can use as a spraying reagent. - It is used for quantitative determination of cardenolides.

Acidic and Enzymatic hydrolysis - The glycosidic linkages are cleaved by acid hydrolysis giving aglycones and sugars. - Partial acid hydrolysis may be used for the cleavage of the glycosidic linkages involving deoxy sugar. - Specific enzymes are present in the plants containing cardioactive glycosides. Special precautions to avoid hydrolysis must be taken during drying and storage process Reaction with alkali 1 - Mild alkali hydrolysis removes the O-acetyl group attached to one of the sugar units as in case of lanatoside A, B, C, D. , etc. . . 2 - Strong alkali solution will affect on the lactone ring, where the lactone ring is opened. Once opened, it does not reform to yield the original lactone.

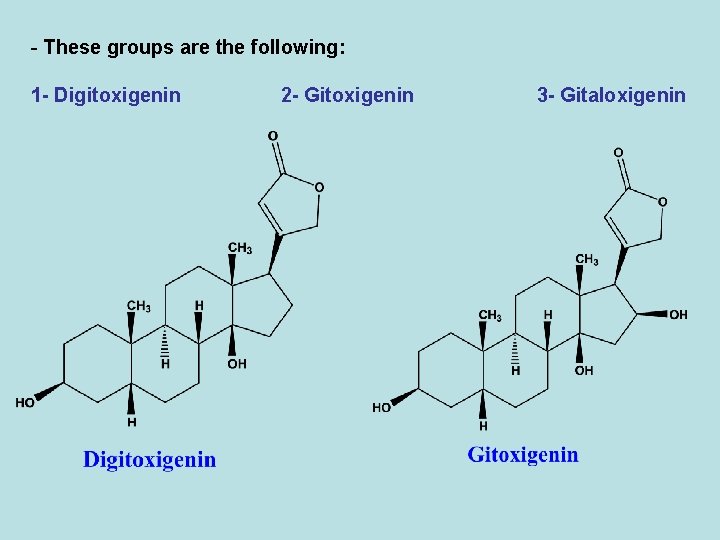
1 - Digitalis glycosides - The leaves of Digitalis purpurea contain about 30 different glycosides, most of them are cardiac glycosides. - On the other hand, around 70 different glycosides have been isolated from D. Lanata leaf. Most of them are cardiac glycosides. - D. Lanata is three times potent than D. Purpurea. - The enzymes present in the leaves of the Digitalis degrade the sugar moieties of the glycosides, so that the dried leaves (crude drug) may contain both sugar-rich primary glycosides and secondary glycosides. - The cardiac glycosides of Digitalis can be divided into three groups according to the structure of the aglycone.

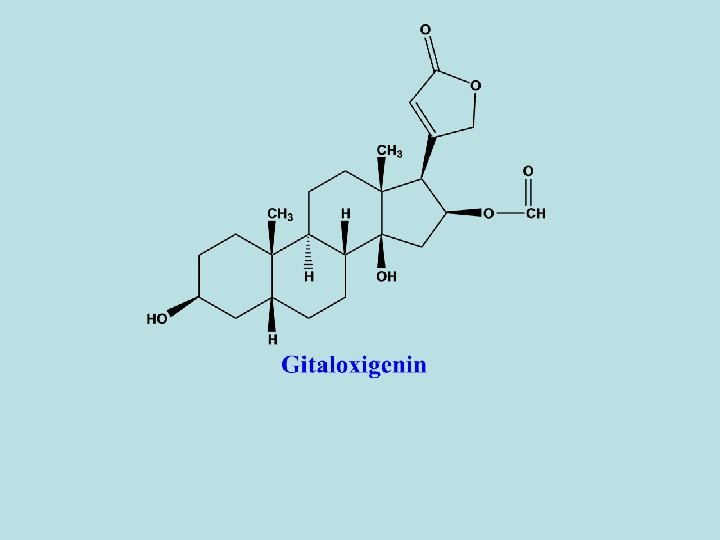
- These groups are the following: 1 - Digitoxigenin 2 - Gitoxigenin 3 - Gitaloxigenin


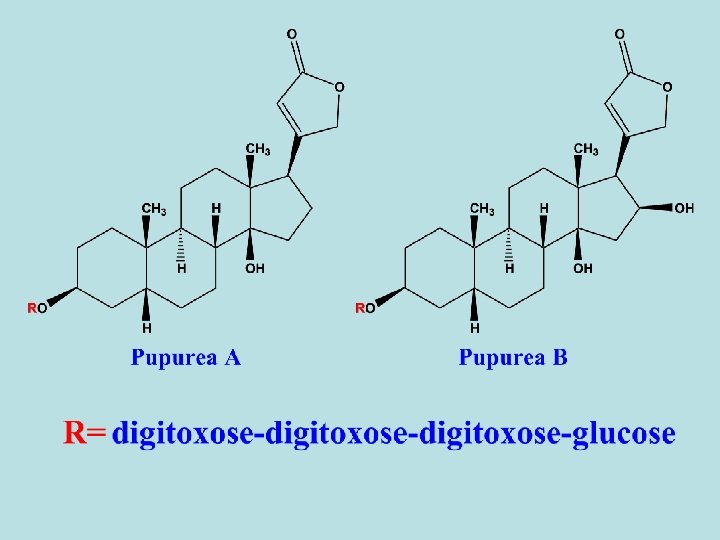
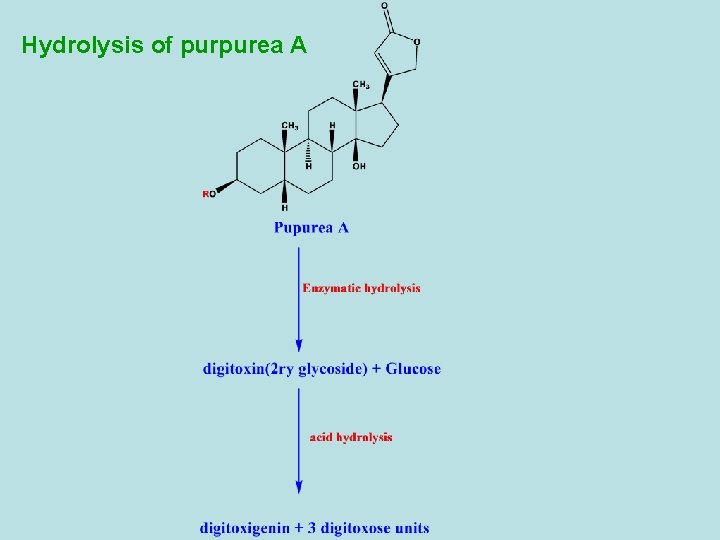
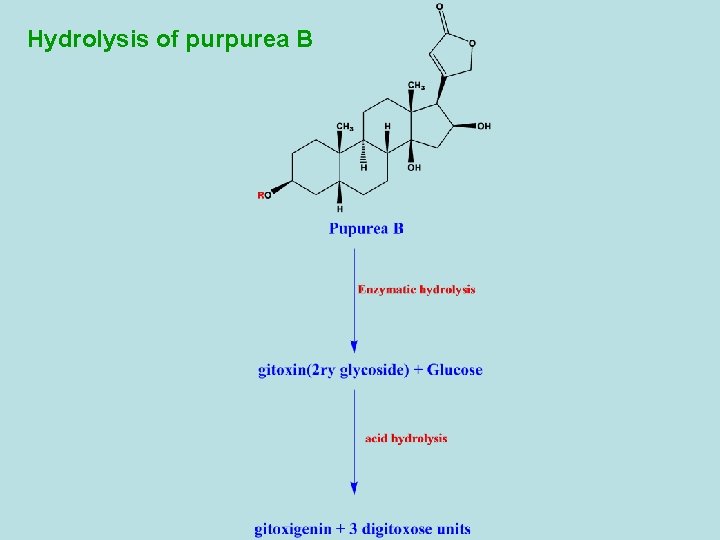
-Digitalis purpurea contains differnt types of primary and secondary cardiac glycosides. - 1 ry glycosides as purpurea A and B. - 2 ry glycosides are resulted from partial hydrolysis (during drying) as digitoxin and gitoxin. - Purpurea A and B are frequently referred as deacetyl lanatoside A and B. - Besides the cardiac glycosides, digitalis leaf contains also sponins. - Digitoxin is the main 2 ry glycoside of D. Purpurea and its effect starts very slowly and reaches its maximum 8 -12 hours after being taken orally. The excretion of the compound is also slow, so that risk of overdosage through cumulation is high. So, the dosage must be determined individually to prevent cumulation.


Hydrolysis of purpurea A

Hydrolysis of purpurea B

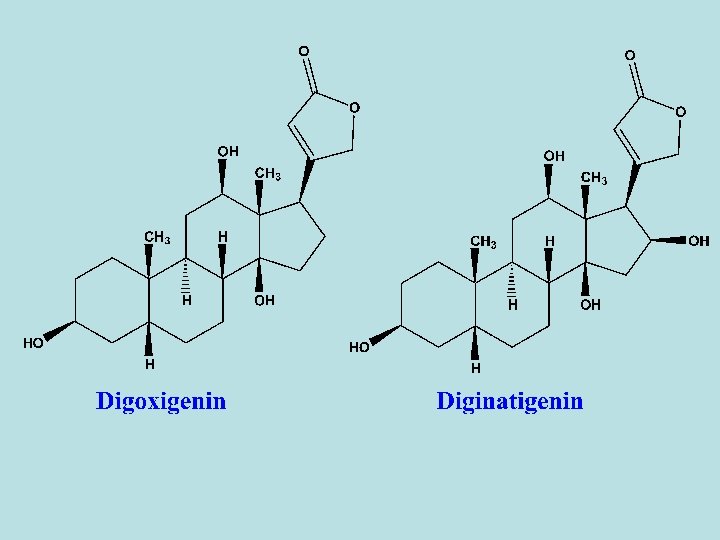
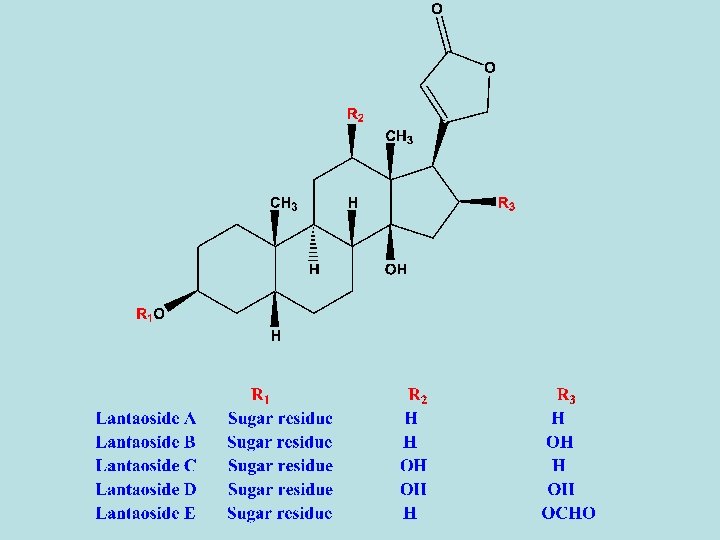
- D. lanata leaves contain five 1 ry glycosides, lanatoside A- E. - All of these glycosides differ from that of purpurea glycosides in that the third digitoxose sugar in the sugar moiety contain an acetyl group. - So, sugar residue are digitoxose-acetyldigitoxose-glucose. - Lanatoside A: its aglycone is digitoxigenin. - Lanatoside B: its aglycone is gitoxigenin. - Lanatoside C: its aglycone is digoxigenin. - Lanatoside D: its aglycone is diginatigenin. - Lanatoside E: its aglycone is gitaloxigenin. - Alkaline hydrolysis give rise to deacetyl lanatoside A (purpurea A), deacetyl lanatoside B (purpurea B) and deacetyl lantoside C – E.

