4 Electrical Conduction in Metals And Alloys Chapter







- Slides: 21


4 Electrical Conduction in Metals And Alloys Chapter 7 in the textbook 2

7. 1 Introduction and Survey Current density: Discuss flux equation: 3

7. 2 Conductivity-Classical Electron Theory Understanding of electrical conduction As Drude did, A free electron gas or plasma ; valence electrons of individual atoms in a crystal For a monovalent metal, What is plasma? : # of atoms / cm 3 : Avogadro constant (#/mole) d : density (gram/cm 3) M : atomic mass of element (gram/mole) One calculates about 1022 to 1023 atoms per cubic centimeter, i. e. , 1022 to 1023 free electrons per cm 3 for a monovalent metal. Mobility: ~ 102 to 103 cm 2/Vsec. Without electric field: the electrons move randomly so that no net velocity results. 4 Thermal velocity:

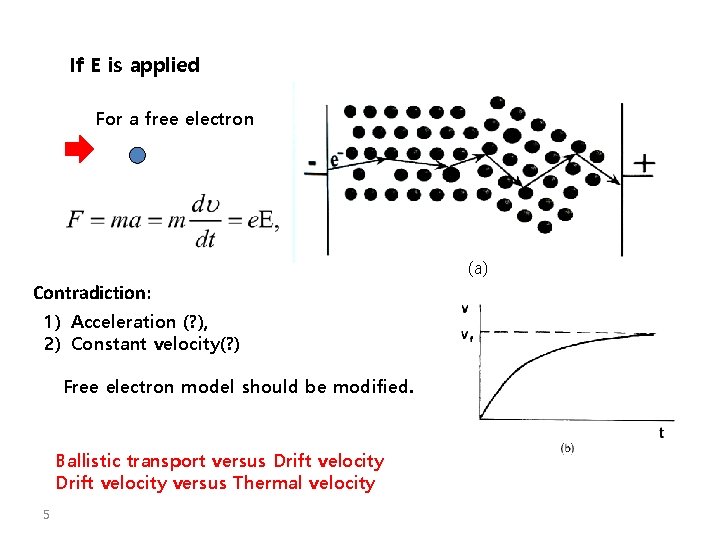
If E is applied For a free electron (a) Contradiction: 1) Acceleration (? ), 2) Constant velocity(? ) Free electron model should be modified. Ballistic transport versus Drift velocity versus Thermal velocity 5

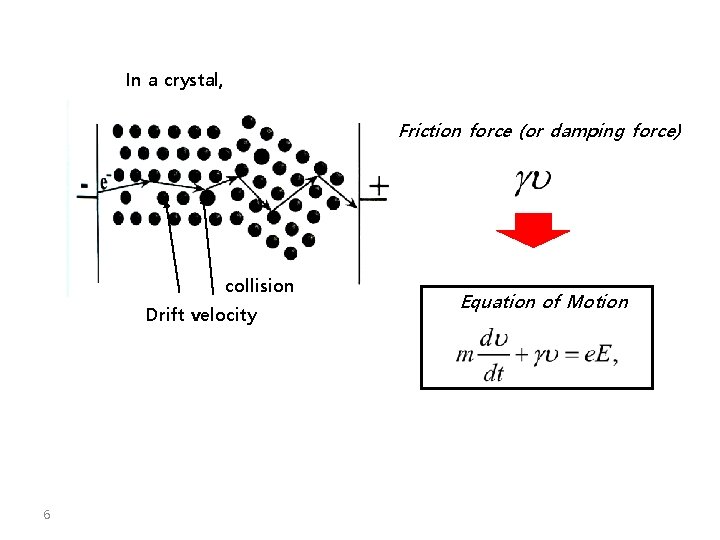
In a crystal, Friction force (or damping force) collision Drift velocity 6 Equation of Motion

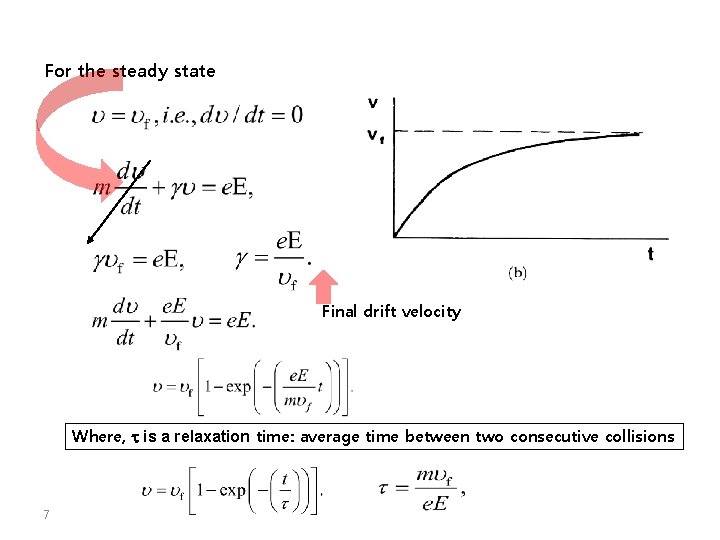
For the steady state Final drift velocity Where, t is a relaxation time: average time between two consecutive collisions 7

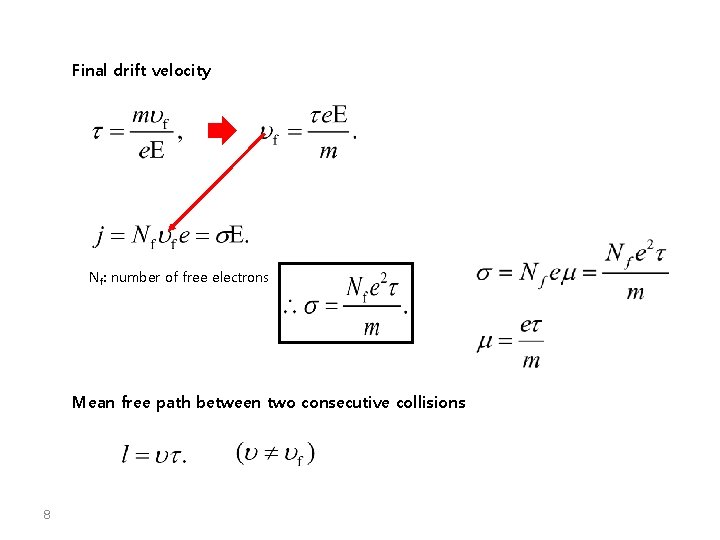
Final drift velocity Nf: number of free electrons Mean free path between two consecutive collisions 8

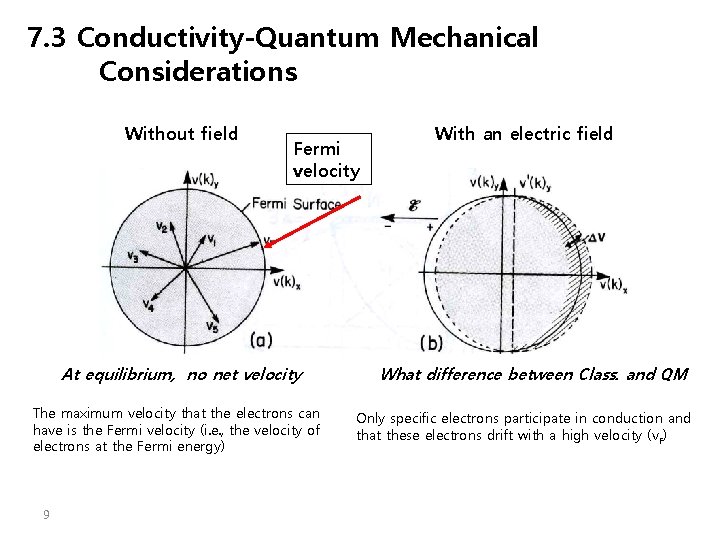
7. 3 Conductivity-Quantum Mechanical Considerations Without field Fermi velocity At equilibrium, no net velocity The maximum velocity that the electrons can have is the Fermi velocity (i. e. , the velocity of electrons at the Fermi energy) 9 With an electric field What difference between Class. and QM Only specific electrons participate in conduction and that these electrons drift with a high velocity (v. F)

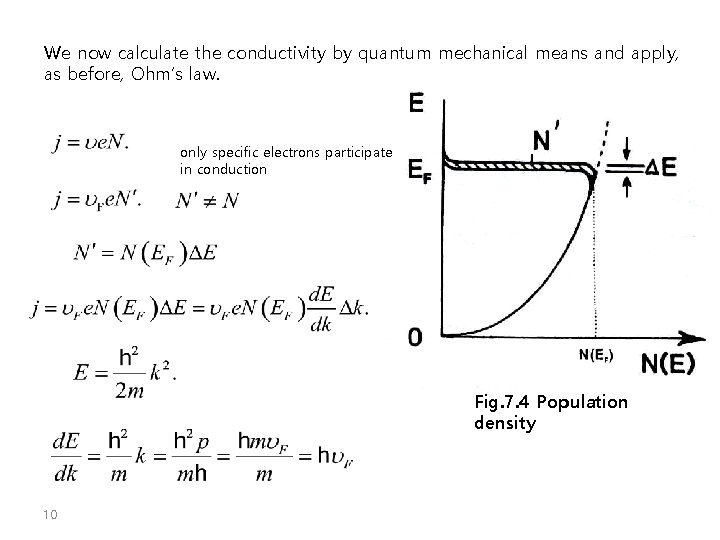
We now calculate the conductivity by quantum mechanical means and apply, as before, Ohm’s law. only specific electrons participate in conduction Fig. 7. 4 Population density 10

Population density at EF When the electric field vector points in the v(k)x direction: 11 Fig. 7. 5

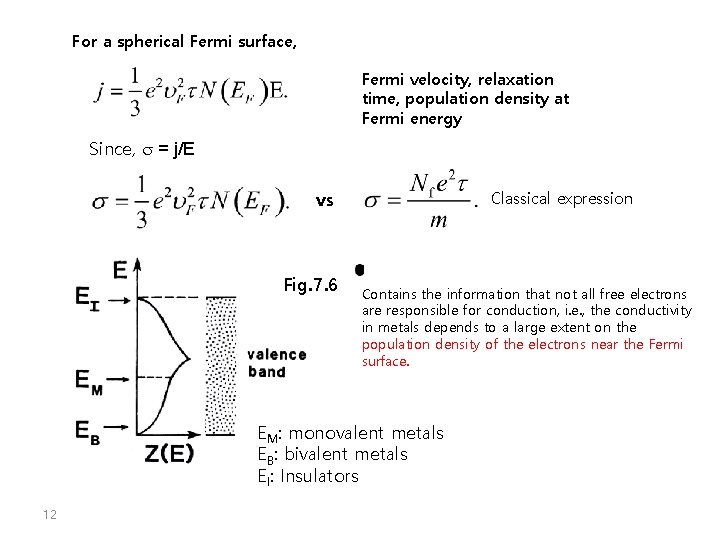
For a spherical Fermi surface, Fermi velocity, relaxation time, population density at Fermi energy Since, s = j/E vs Fig. 7. 6 Classical expression Contains the information that not all free electrons are responsible for conduction, i. e. , the conductivity in metals depends to a large extent on the population density of the electrons near the Fermi surface. EM: monovalent metals EB: bivalent metals EI: Insulators 12

Classical Concept: QM: Since, s = j/E vs 13 Classical expression

7. 4 Experimental Results and Their Interpretation 7. 4. 1 For Pure Metals Linear temperature coefficient of resistivity Matthiessen’s rule Ideal resistivity Residual resistivity 14 Good indicator to check the quality of materials. One definition of metal – the resistivity goes up with the temperature

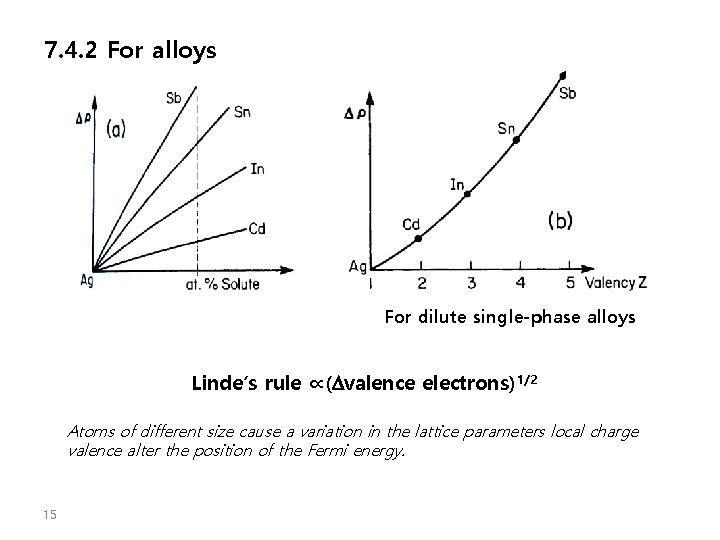
7. 4. 2 For alloys For dilute single-phase alloys Linde’s rule ∝(Dvalence electrons)1/2 Atoms of different size cause a variation in the lattice parameters local charge valence alter the position of the Fermi energy. 15

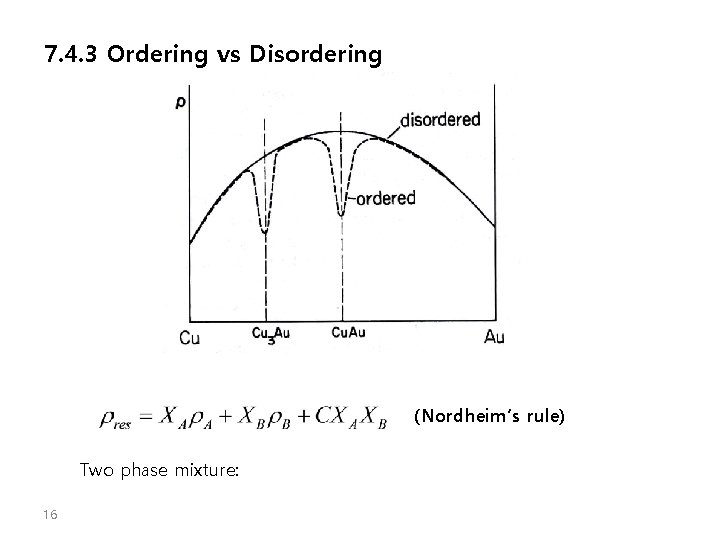
7. 4. 3 Ordering vs Disordering (Nordheim’s rule) Two phase mixture: 16

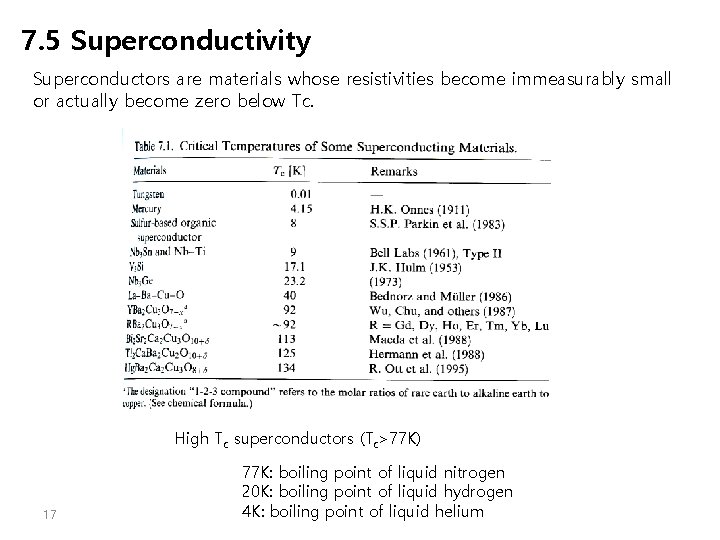
7. 5 Superconductivity Superconductors are materials whose resistivities become immeasurably small or actually become zero below Tc. High Tc superconductors (Tc>77 K) 17 77 K: boiling point of liquid nitrogen 20 K: boiling point of liquid hydrogen 4 K: boiling point of liquid helium

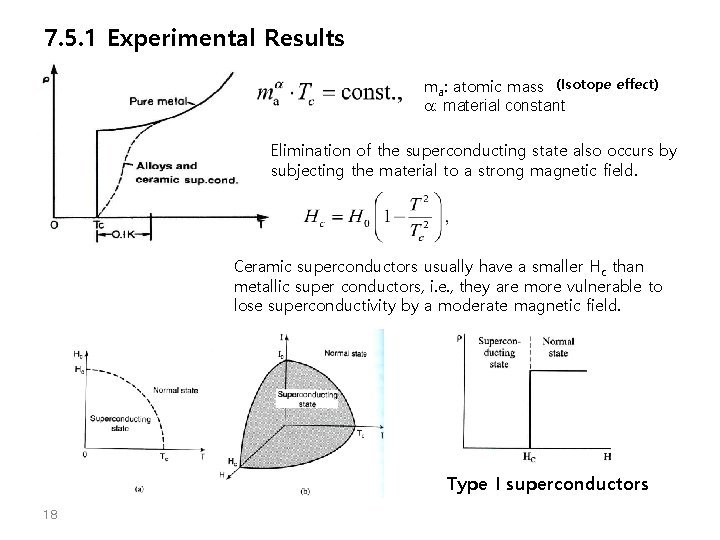
7. 5. 1 Experimental Results ma: atomic mass (Isotope effect) a: material constant Elimination of the superconducting state also occurs by subjecting the material to a strong magnetic field. Ceramic superconductors usually have a smaller Hc than metallic super conductors, i. e. , they are more vulnerable to lose superconductivity by a moderate magnetic field. Type I superconductors 18

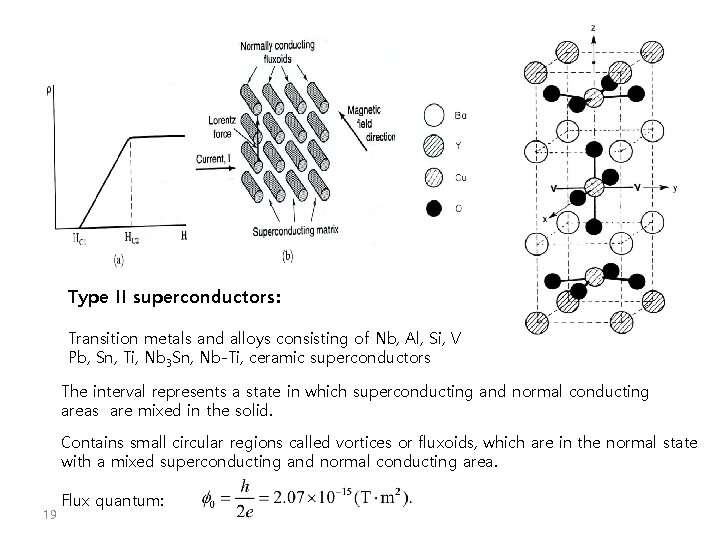
Type II superconductors: Transition metals and alloys consisting of Nb, Al, Si, V Pb, Sn, Ti, Nb 3 Sn, Nb-Ti, ceramic superconductors The interval represents a state in which superconducting and normal conducting areas are mixed in the solid. Contains small circular regions called vortices or fluxoids, which are in the normal state with a mixed superconducting and normal conducting area. 19 Flux quantum:

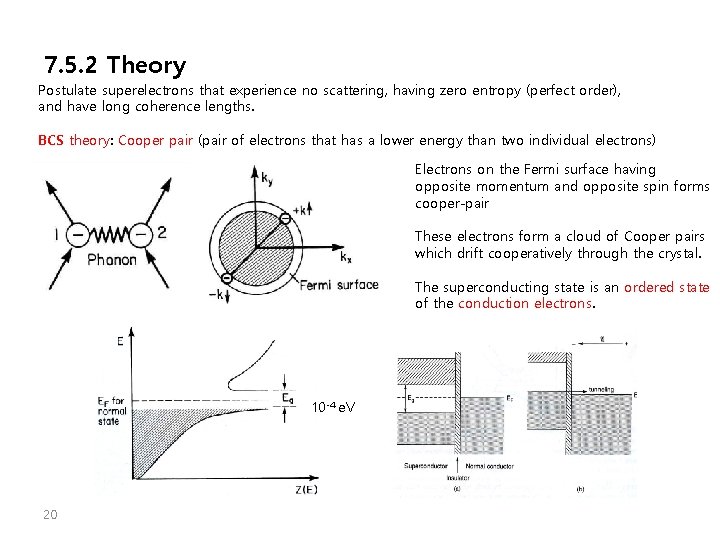
7. 5. 2 Theory Postulate superelectrons that experience no scattering, having zero entropy (perfect order), and have long coherence lengths. BCS theory: Cooper pair (pair of electrons that has a lower energy than two individual electrons) Electrons on the Fermi surface having opposite momentum and opposite spin forms cooper-pair These electrons form a cloud of Cooper pairs which drift cooperatively through the crystal. The superconducting state is an ordered state of the conduction electrons. 10 -4 e. V 20

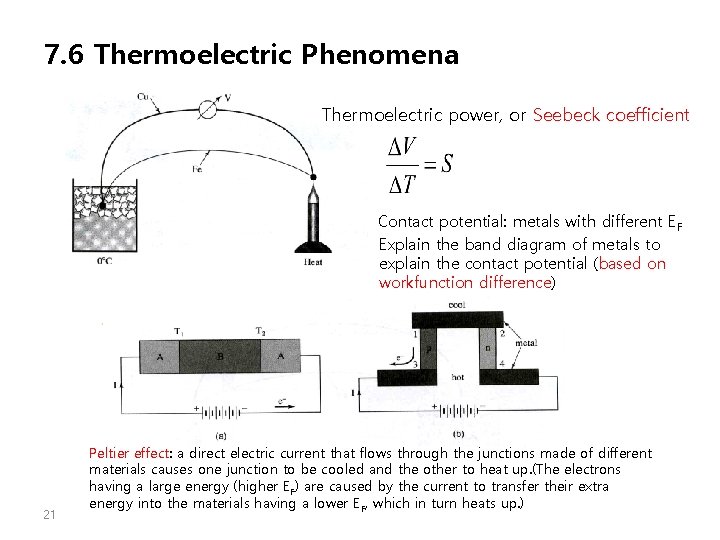
7. 6 Thermoelectric Phenomena Thermoelectric power, or Seebeck coefficient Contact potential: metals with different EF Explain the band diagram of metals to explain the contact potential (based on workfunction difference) 21 Peltier effect: a direct electric current that flows through the junctions made of different materials causes one junction to be cooled and the other to heat up. (The electrons having a large energy (higher EF) are caused by the current to transfer their extra energy into the materials having a lower EF, which in turn heats up. )