4 8 Shapes of Molecules Determined by number

4. 8 Shapes of Molecules • Determined by number of valence electrons of the central atom • 3 -D shape a result of bonded pairs and lone pairs of electrons • Use VSEPR theory (valence-shell-electron-pair repulsion Timberlake Lecture. PLUS 1

Using the VSEPR Model 1. Draw the electron-dot structure 2. Identify the central atom 3. Count the total number of electron pairs around central atom 4. Predict the electron shape 5. Predict the shape of the molecule using the bonding atoms Timberlake Lecture. PLUS 2
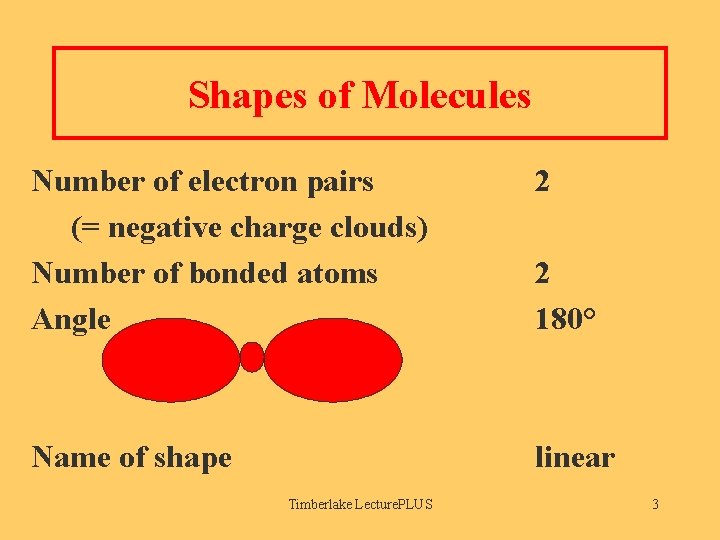
Shapes of Molecules Number of electron pairs (= negative charge clouds) Number of bonded atoms Angle 2 Name of shape linear Timberlake Lecture. PLUS 2 180° 3
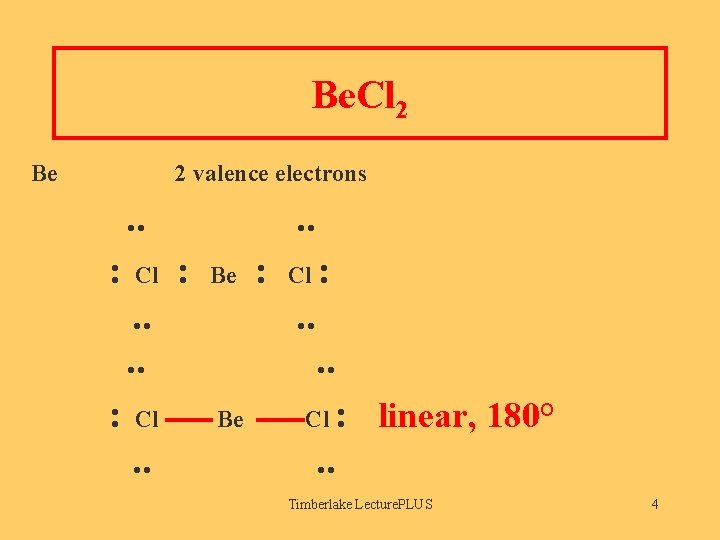
Be. Cl 2 Be 2 valence electrons . . : Cl . . : Be . . : Cl . . : Cl : . . Be . . Cl : linear, 180°. . Timberlake Lecture. PLUS 4
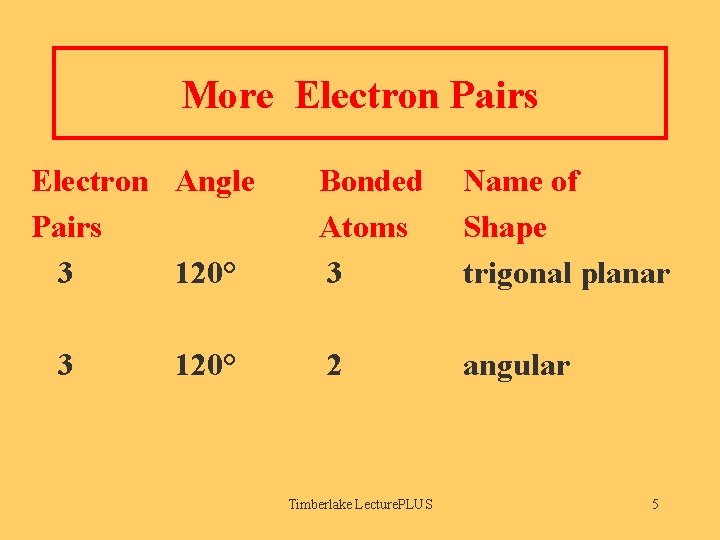
More Electron Pairs Electron Angle Pairs 3 120° Bonded Atoms 3 Name of Shape trigonal planar 2 angular Timberlake Lecture. PLUS 5
Electron Shape with 3 Pairs Timberlake Lecture. PLUS 6
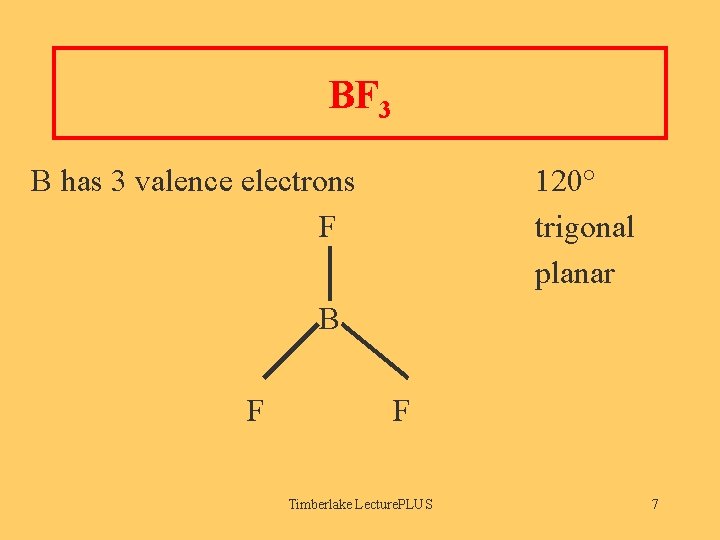
BF 3 B has 3 valence electrons F 120° trigonal planar B F F Timberlake Lecture. PLUS 7
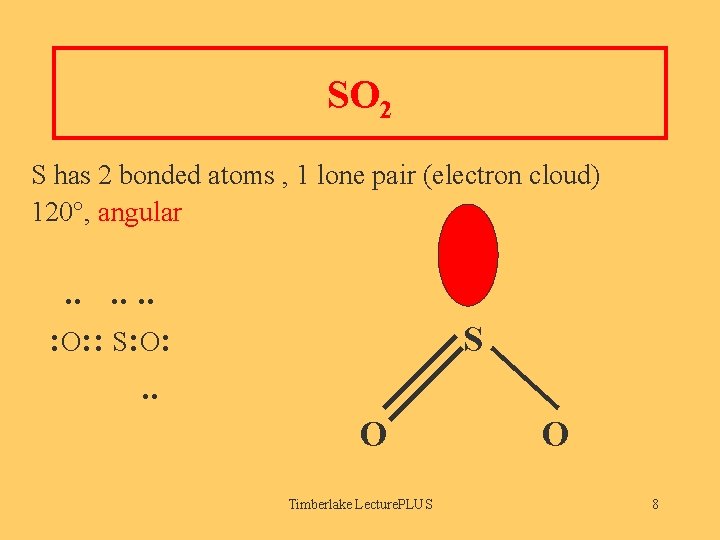
SO 2 S has 2 bonded atoms , 1 lone pair (electron cloud) 120°, angular . . . : O: : S: O: . . S O Timberlake Lecture. PLUS O 8
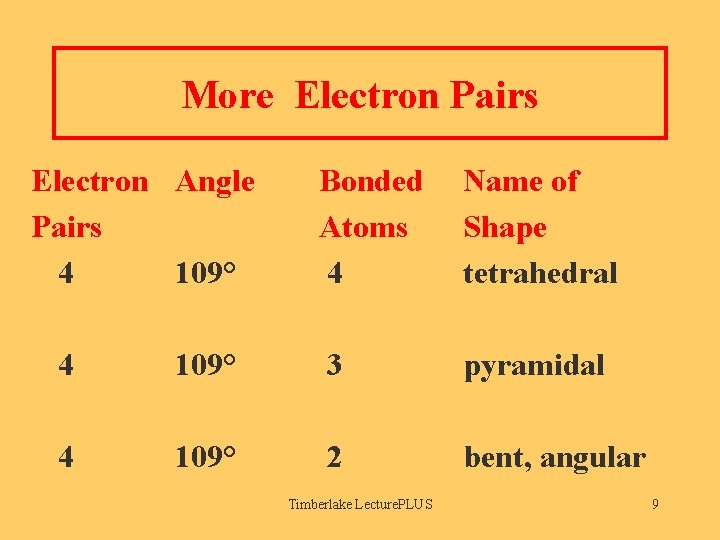
More Electron Pairs Electron Angle Pairs 4 109° Bonded Atoms 4 Name of Shape tetrahedral 4 109° 3 pyramidal 4 109° 2 bent, angular Timberlake Lecture. PLUS 9
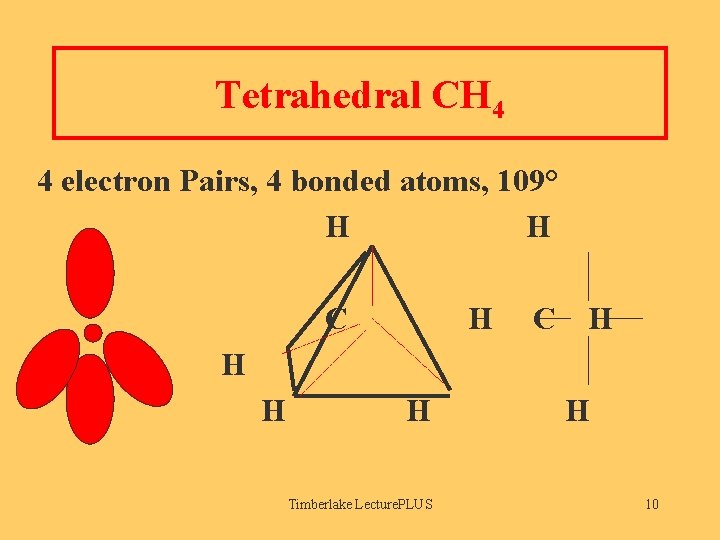
Tetrahedral CH 4 4 electron Pairs, 4 bonded atoms, 109° H H C H H Timberlake Lecture. PLUS H 10
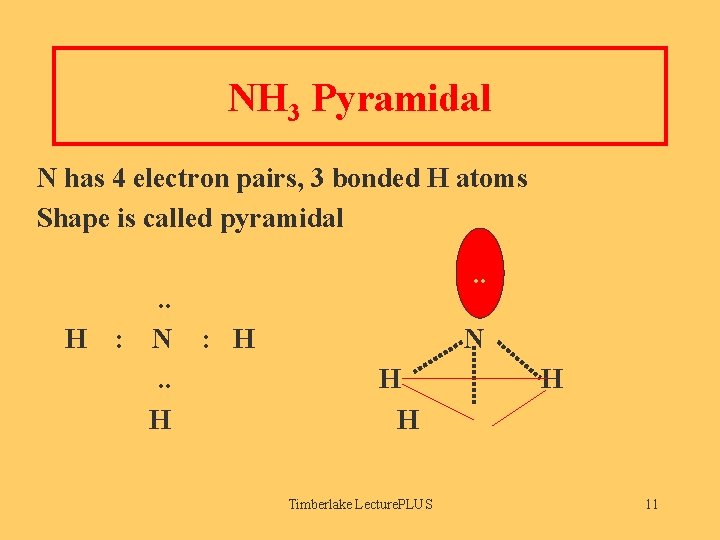
NH 3 Pyramidal N has 4 electron pairs, 3 bonded H atoms Shape is called pyramidal H . . : N. . H . . : H N H H Timberlake Lecture. PLUS H 11
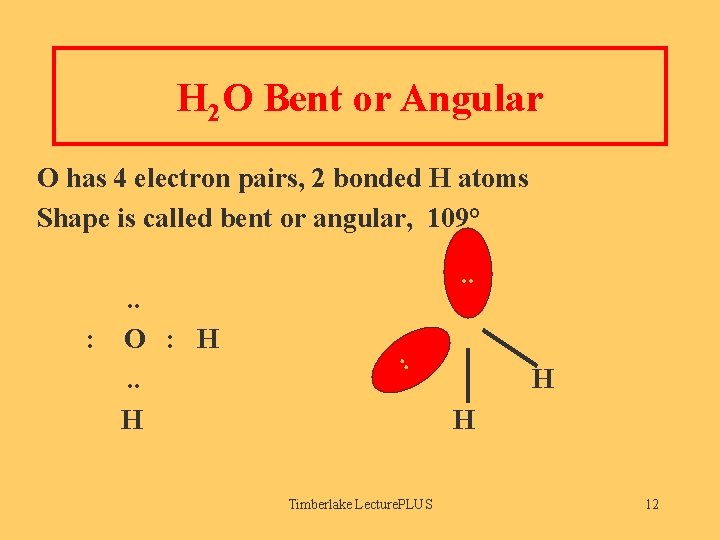
H 2 O Bent or Angular O has 4 electron pairs, 2 bonded H atoms Shape is called bent or angular, 109° O. . : O : H. . H . . H H Timberlake Lecture. PLUS 12

Learning Check Determine the molecular geometry of each of the following: A. CCl 4 1) tetrahedral 2) pyramidal 3) angular B. SO 3 1) trigonal planar 2) pyramidal 3) angular C. PCl 3 Lecture. PLUS 1) trigonal planar Timberlake 2) pyramidal 3) angular 13

Solution Determine the molecular geometry of each of the following: A. CCl 4 1) tetrahedral B. SO 3 1) trigonal planar C. PCl 3 2) pyramidal Timberlake Lecture. PLUS 14
- Slides: 14