4 3 MODERN ATOMIC THEORY VOCABULARY energy levels

4. 3 MODERN ATOMIC THEORY Ø VOCABULARY: Ø energy levels Ø electron cloud Ø orbital Ø electron configuration Ø ground state Ø OBJECTIVES: Ø Describe Bohr’s model of the atom and the evidence for energy levels. Ø Explain how the electron cloud model represents the behavior and locations of electrons in atoms. 10/17/2021 4. 3 Modern Atomic Theory 1

BOHR’S MODEL OF THE ATOM Ø Niels Bohr (1885 -1962) Ø Danish physicist Ø Worked with Rutherford Ø Agreed with Rutherford’s model of a nucleus surrounded by large volume of space. However, Bohr’s model focused on electrons. Ø Main focus of modern atomic model: describing the arrangement of electrons in an atom 10/17/2021 4. 3 Modern Atomic Theory 2
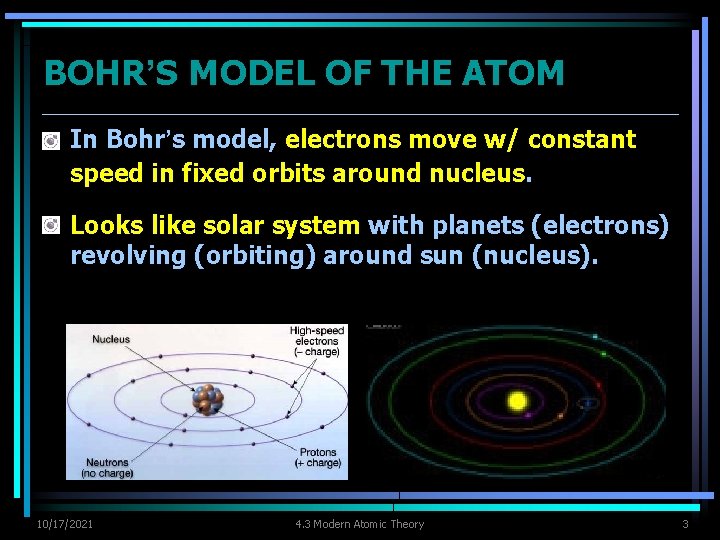
BOHR’S MODEL OF THE ATOM Ø In Bohr’s model, electrons move w/ constant speed in fixed orbits around nucleus. Ø Looks like solar system with planets (electrons) revolving (orbiting) around sun (nucleus). 10/17/2021 4. 3 Modern Atomic Theory 3

BOHR’S MODEL OF THE ATOM Ø In Bohr’s model, electrons move w/ constant speed in fixed orbits around nucleus. Ø Looks like solar system with planets (electrons) revolving (orbiting) around sun (nucleus). 10/17/2021 4. 3 Modern Atomic Theory 4

BOHR’S MODEL OF THE ATOM Ø Atomic structure animation table for first 11 elements of the Periodic Table Ø http: //web. visionlearning. com/custom/chemistry/animations /CHE 1. 3 -an-animations. shtml 10/17/2021 4. 3 Modern Atomic Theory 5

BOHR’S MODEL OF THE ATOM: ENERGY LEVELS Ø Each electron has a specific amount of energy. Ø Energy levels - possible energies that electrons in an atom can have Ø An electron in an atom can move from one energy level to another when the atom gains or loses energy. 10/17/2021 4. 3 Modern Atomic Theory 6

BOHR’S MODEL OF THE ATOM: ENERGY LEVELS Ø http: //www. colorado. edu/physics/2000/quantumzone/bohr. html Ø Larger orbit to smaller orbit – loses energy (squiggle shoots out) Ø Smaller orbit to larger orbit – gains energy (squiggle comes in). 10/17/2021 4. 3 Modern Atomic Theory 7

BOHR’S MODEL OF THE ATOM: EVIDENCE FOR ENERGY LEVELS Ø Scientist can measure… Ø energy gained when electrons absorb energy & move to a higher energy level. Ø energy released when electron returns to a lower energy level. Ø Light is usually emitted when electrons move to lower energy levels. 10/17/2021 4. 3 Modern Atomic Theory 8

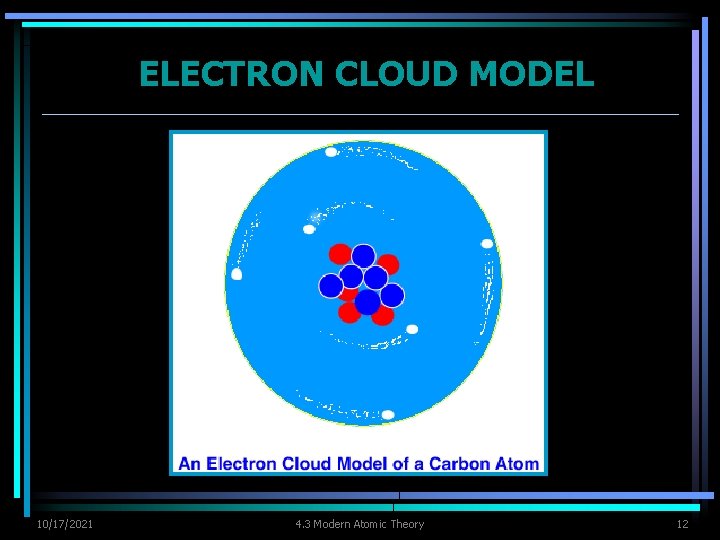
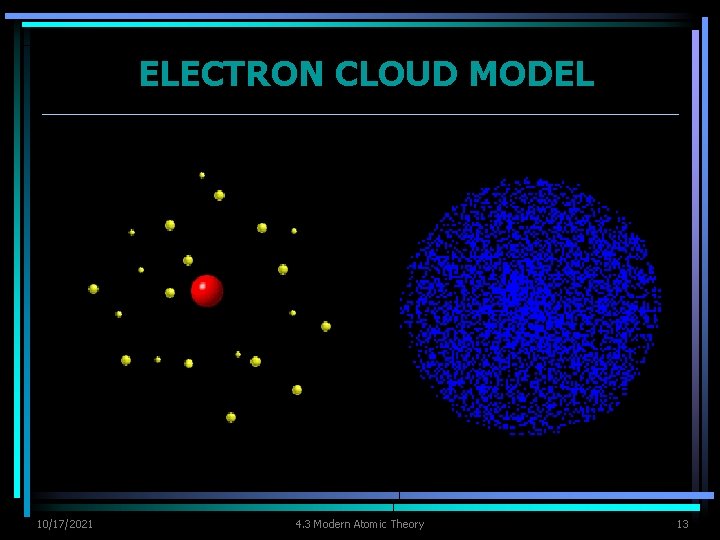
ELECTRON CLOUD MODEL Ø As scientist made more discoveries, Bohr’s model was improved. Ø Bohr’s model Ø correct in introducing concept of energy levels Ø incorrect in assuming electrons moved like planets do in our solar system Ø Can’t be sure at a specific moment where electrons are located because electrons move in less predictable ways & energy levels cannot be used to describe actual location of an electron. 10/17/2021 4. 3 Modern Atomic Theory 9

ELECTRON CLOUD MODEL Ø Electron cloud - visual model of the most likely locations for electrons in an atom. Ø cloud is denser at locations where probability of finding an electron is high Ø Scientist use the electron cloud model to describe the possible locations of electrons around the nucleus. 10/17/2021 4. 3 Modern Atomic Theory 10

ELECTRON CLOUD MODEL Ø Example: propeller moving Ø know blades are located somewhere in blur but you can’t be exactly sure where each blade is located at specific moment in time. 10/17/2021 4. 3 Modern Atomic Theory 11
ELECTRON CLOUD MODEL 10/17/2021 4. 3 Modern Atomic Theory 12
ELECTRON CLOUD MODEL 10/17/2021 4. 3 Modern Atomic Theory 13

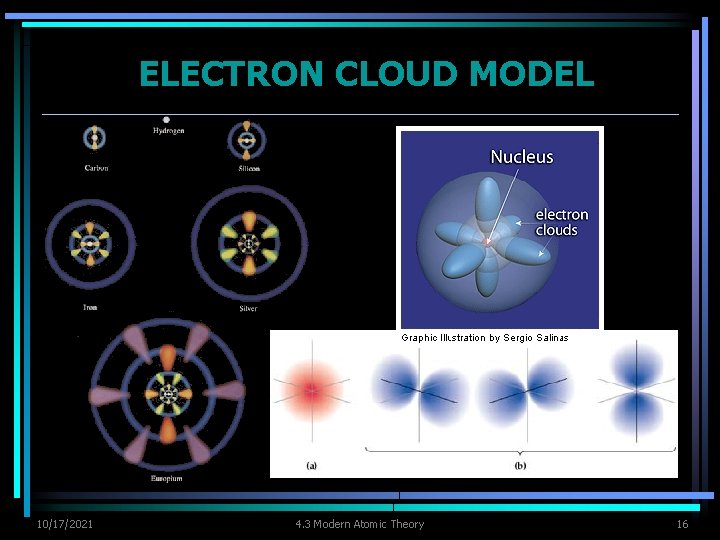
ATOMIC ORBITALS Ø Electron cloud represents all the orbitals in an atom. Ø Orbital - region of space around nucleus where an electron is likely to be found Ø An electron cloud is a good approximation of how electrons behave in their orbitals. 10/17/2021 4. 3 Modern Atomic Theory 14

ATOMIC ORBITALS Ø Lowest energy level (least energy) has 1 orbital Ø Highest energy level has more than one orbital Ø Max number of electrons in an energy level is twice # of orbitals Ø Each orbital can contain two electrons at most. 10/17/2021 4. 3 Modern Atomic Theory 15
ELECTRON CLOUD MODEL 10/17/2021 4. 3 Modern Atomic Theory 16

ELECTRON CONFIGURATION Ø Configuration is an arrangement of objects in a given space. Ø Some configurations more stable than others (less likely to change) 10/17/2021 4. 3 Modern Atomic Theory 17

ELECTRON CONFIGURATION Ø Electron configuration - arrangement of electrons in the orbitals of an atom Ø The most stable electron configuration is the one in which the electrons are in orbitals with the lowest possible energies. Ø Ground state - all electrons in an atom have the lowest possible energies 10/17/2021 4. 3 Modern Atomic Theory 18

ELECTRON CONFIGURATION Ø David’s Wizzy Periodic Table - Click on Element to See Atom Structure http: //www. colorado. edu/physics/2000/applets/a 2. html 10/17/2021 4. 3 Modern Atomic Theory 19

SUMMARY Ø In the past , scientist believed that electrons circled the nucleus the same way the planets circled the sun. Today however, scientists know that there is no exact path of an electron. The quick moving electrons form a “cloud” around the nucleus. Ø In the modern atomic theory, electrons are arranged into energy levels, or shells. 10/17/2021 4. 3 Modern Atomic Theory 20

FLAME TEST Ø To prepare for tomorrow’s lab you have to read section 4. 3 and the lab handout. 10/17/2021 4. 3 Modern Atomic Theory 21
Ø Bohr’s Atom http: //www. visionlearning. com/library/flash_viewer. php? oid=1347 Ø periodic table 11 elements http: //web. visionlearning. com/custom/chemistry/animations/CHE 1. 3 -an-atoms. shtml Ø http: //www. visionlearning. com/library/index. php Ø http: //www. visionlearning. com/library/module_viewer. php? mid=51&l=&c 3= Ø Ø common isotopes http: //web. visionlearning. com/custom/chemistry/overheads/CHE 1. 3 -oh-isotopes. shtml Ø Brainpop. com 10/17/2021 4. 3 Modern Atomic Theory 22
- Slides: 22