4 3 BALANCING EQUATIONS LEARNING OUTCOMES You Should
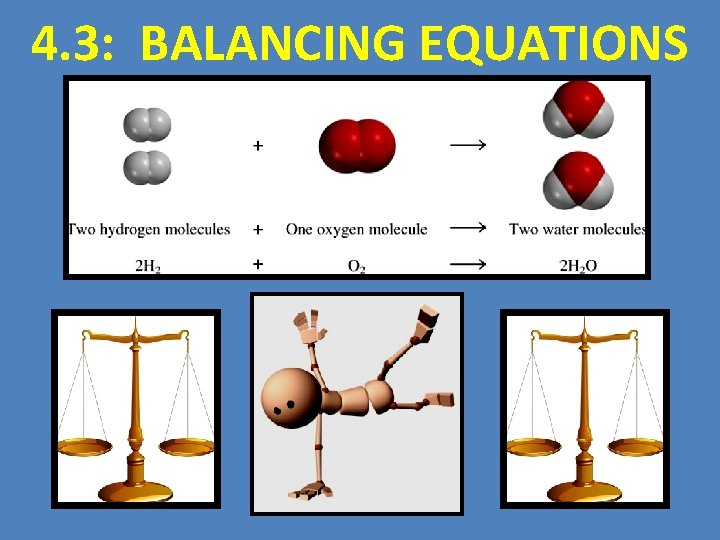
4. 3: BALANCING EQUATIONS

LEARNING OUTCOMES You Should Be Able To… 1. Define and explain the law of conservation of mass 2. Represent chemical reactions and the conservation of atoms, using molecular models 3. Write and balance (using the lowest whole number coefficients) chemical equations from formulae, word equations, or descriptions of experiments

VOCABULARY q Subscript q Coefficient q Law of Conservation of Mass q Molecule q Atom q Skeleton Equation q Balanced Equation q Word Equation

Balancing Equations • Chemical reactions result in chemical changes. – Chemical changes occur when new substances are created. – The original substance(s), called reactants, change into new substance(s) called products. Reactants (c) Mc. Graw Hill Ryerson 2007 Products See pages 202 - 203
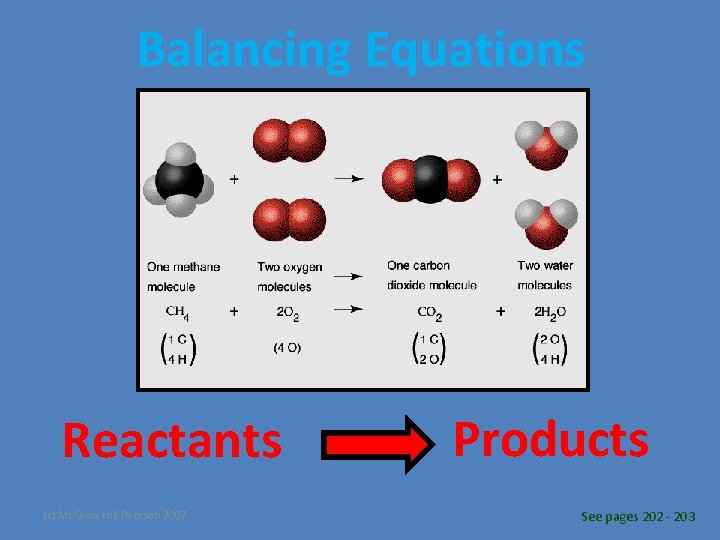
Balancing Equations Reactants (c) Mc. Graw Hill Ryerson 2007 Products See pages 202 - 203
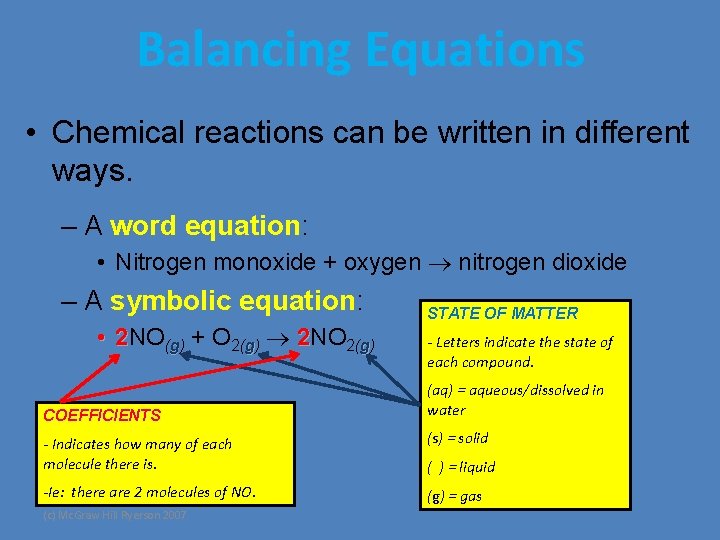
Balancing Equations • Chemical reactions can be written in different ways. – A word equation: • Nitrogen monoxide + oxygen nitrogen dioxide – A symbolic equation: • 2 NO(g) + O 2(g) 2 NO 2(g) COEFFICIENTS STATE OF MATTER - Letters indicate the state of each compound. (aq) = aqueous/dissolved in water - Indicates how many of each molecule there is. (s) = solid -Ie: there are 2 molecules of NO. (g) = gas (c) Mc. Graw Hill Ryerson 2007 ( ) = liquid

Law of Conservation of Mass • When a chemical reaction occurs, new compounds are created, BUT… – No new matter is created or destroyed; atoms are just No new matter is created or destroyed rearranged as the atoms change partners to form new rearranged change partners compounds. – If there are 3 atoms of oxygen in the reactants, there MUST be 3 atoms of oxygen in the products. – Number of each atom in reactants = number of each atom in products. • The law of conservation of mass: – Mass of reactants = mass of products If you could collect and measure all of the exhaust from this car, you would find that mass of reactants (gas + O 2) = mass of products (exhaust). (c) Mc. Graw Hill Ryerson 2007

Writing & Balancing Equations • The simplest form of chemical equation is a word equation. – Potassium metal + oxygen gas potassium oxide • A skeleton equation shows the formulas of the elements/compounds. – A skeleton equation shows which atoms are involved, but not how many molecules are involved. • K + O 2 K 2 O
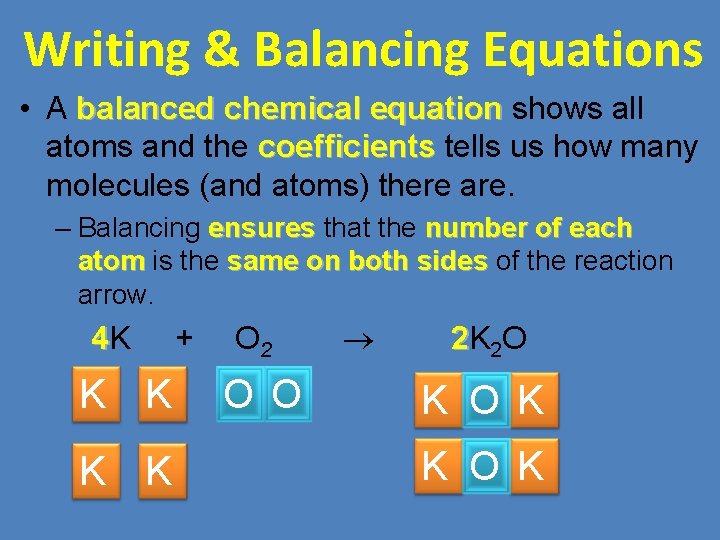
Writing & Balancing Equations • A balanced chemical equation shows all balanced chemical equation atoms and the coefficients tells us how many coefficients molecules (and atoms) there are. – Balancing ensures that the number of each ensures atom is the same on both sides of the reaction atom same on both sides arrow. 4 K + K K O 2 O O 2 K 2 O K K O K

Counting Atoms to Balance Equations • Using the law of conservation of mass, we can count atoms to balance the number of count atoms to balance atoms in chemical equations. – Word equation: methane + oxygen water + carbon dioxide – Skeleton equation: CH 4 + O 2 H 2 O + CO 2 • To balance the compounds, take note of how many atoms of each element occur on each side of the reaction arrow. (c) Mc. Graw Hill Ryerson 2007 See Page 207
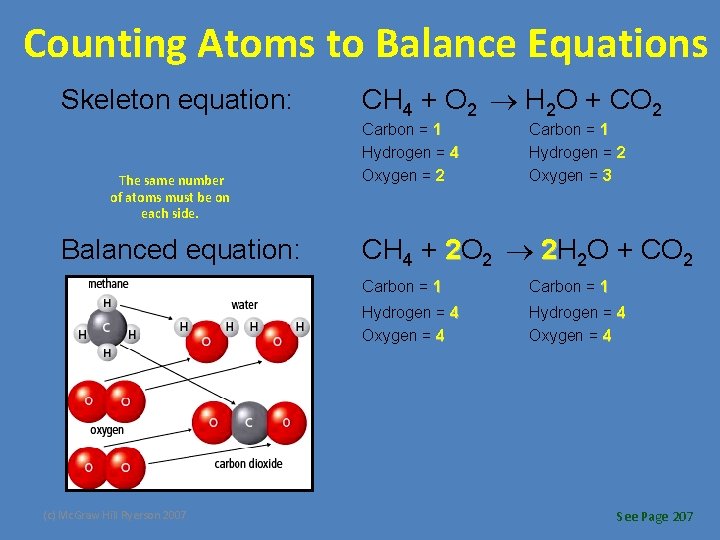
Counting Atoms to Balance Equations Skeleton equation: The same number of atoms must be on each side. Balanced equation: (c) Mc. Graw Hill Ryerson 2007 CH 4 + O 2 H 2 O + CO 2 Carbon = 1 Hydrogen = 4 Oxygen = 2 Carbon = 1 Hydrogen = 2 Oxygen = 3 CH 4 + 2 O 2 2 H 2 O + CO 2 Carbon = 1 Hydrogen = 4 Oxygen = 4 See Page 207
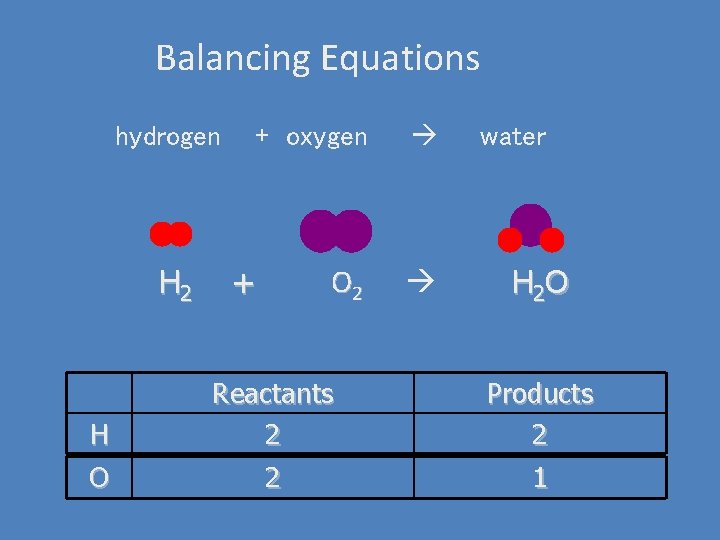
Balancing Equations hydrogen H 2 H O + oxygen O 2 + water H 2 O Reactants 2 Products 2 2 1

Balancing Equations + oxygen hydrogen peroxide X hydrogen H 2 + O 2 H 2 O 2 YOU CANNOT CHANGE THE SUBSCRIPTS H O Reactants 2 2 Products 2 2
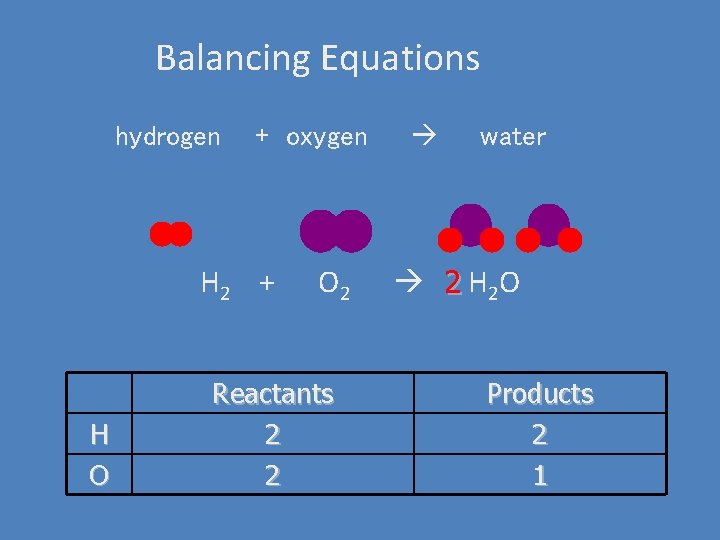
Balancing Equations hydrogen + oxygen H 2 + H O O 2 Reactants 2 2 water 2 H 2 O Products 2 1

Balancing Equations hydrogen + oxygen H 2 + H O O 2 Reactants 2 2 water 2 H 2 O Products 4 2

Balancing Equations hydrogen + oxygen 2 H 2(g) + H O water O 2 (g) 2 H 2 O (l) Reactants 4 2 Products 4 2

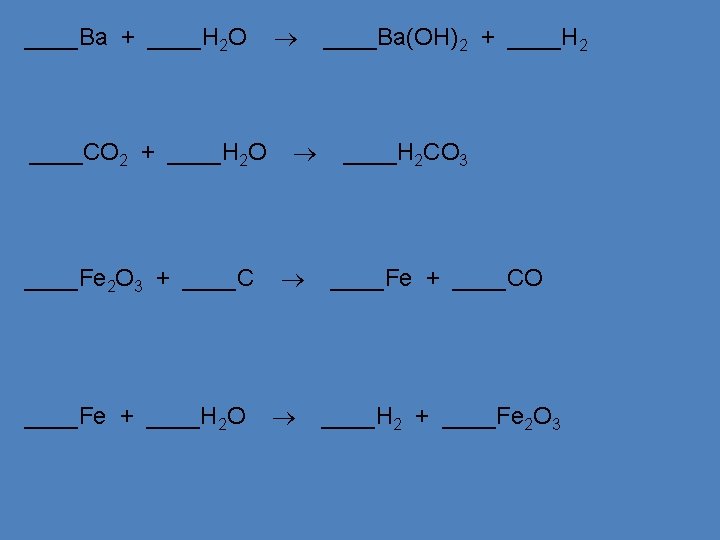
Birkeland’s Big Bag ‘O Balancing Tricks • Balance chemical equations by following these steps: – Trial and error will work but can be very inefficient. • USE A TABLE (write atoms underneath reactants and products) USE A TABLE • If they look the same on both sides of the equation, treat 2– polyatomic ions (such as SO polyatomic ions as a group 4 ) as a group & balance them as such. • If ‘OH’ and H 2 O are in the equation, write water as HOH. • Balance one compound at a time & Balance one compound at a time rewrite the # of atoms in rewrite the # of atoms the chart as things change • Only add coefficients; NEVER change subscripts!!! Only add coefficients • If H and O appear in more than one place, attempt to balance H and O them LAST – Balance everything that isn’t ‘H’ or ‘O’ 1 st. – Balance the ‘H’s 2 nd to last. – Balance the ‘O’s last. – Always double-check after you think you are finished. Always double-check – CHECK YOUR ANSWERS!!! See pages 209 - 211 (c) Mc. Graw Hill Ryerson 2007

http: //www. authorstream. com/Presentation/kellymdeters-86103 -section 2 -7 -balancing-chemical-equations-education-ppt-powerpoint/

• Balance the following: – Fe + Br 2 Fe. Br 3 – Sn(NO 2)4 + K 3 PO 4 KNO 2 + Sn 3 (PO 4)4 – C 2 H 6 + O 2 CO 2 + H 2 O

Turn Word Equations into Balanced Equations • If you don’t transform your word into a skeleton equation properly, you won’t be able to balance the equation correctly. – Change chemical names into chemical formulas. 4 types: types • Simple ionic compounds • Multivalent ionic compounds • Ionic compounds with polyatomic ions • Covalent compound • Be careful of diatomic elements -remember the special seven!! seven H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2 See page 208 (c) Mc. Graw Hill Ryerson 2007

Turn Word Equations into Balanced Equations w Several common covalent molecules containing hydrogen have common names that you should know and MEMORIZE!! § methane = CH 4 § glucose = C 6 H 12 O 6 § ethane = C 2 H 6 § ammonia = NH 3 See page 208 (c) Mc. Graw Hill Ryerson 2007

Turn Word Equations into Balanced Equations Example #1: Word Equation: Solutions of lead nitrate react with potassium iodide to produce solid lead iodide and a solution of potassium nitrate. Skeleton Equation: Pb(NO 3)2(aq) + KI(aq) Pb. I 2(s) + KNO 3(aq) Balanced Equation: Pb(NO 3)2(aq) + 2 KI(aq) Pb. I 2(s) + 2 KNO 3(aq) Example #2: Word Equation: Copper reacts with hydrogen nitrate to produce copper (II) nitrate plus hydrogen. Skeleton Equation: Cu + H(NO 3) Cu(NO 3)2 + H 2 Balanced Equation: Cu + 2 H(NO 3) Cu(NO 3)2 + H 2 See page 208 (c) Mc. Graw Hill Ryerson 2007
- Slides: 23