4 2 b Naming Covalent Molecules A Naming

4. 2 b Naming Covalent Molecules

A) Naming COVALENT Molecules • Covalent Compounds: composed of 2 parts: a non-metal and another non-metal A binary covalent compound contains two non-metal elements joined together by one or more covalent bonds.

Different from naming Ionic molecules because: • Need to add a prefix saying how many of each atom you have Same as Ionic: • change the end of second name to idefrom naming Ionic molecules because: • When using polyatomic ions, the name doesn’t change

Name: • When naming a binary covalent compound, prefixes' are used to indicate how many atoms of each element are present. • The second element’s name ends with the suffix “-ide. ” • No prefix is used if there is just one atom of the first element. • Do not reduce the name.
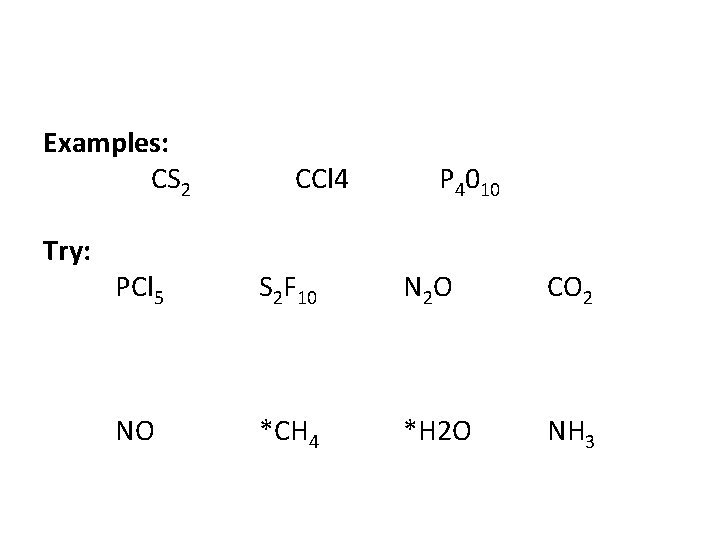
Examples: CS 2 Try: CCl 4 P 4010 PCl 5 S 2 F 10 N 2 O CO 2 NO *CH 4 *H 2 O NH 3

B) Writing COVALENT Formulas Chemical formula: • When writing the chemical formula, subscripts are used to indicate the number of atoms present. • The exact number of atoms is always shown in the formula. • Unlike the formula for an ionic compound, the subscripts do not always represent the smallest whole number formula.

Examples: • Dinitrogen trioxide • Sulfur hexafluoride • Carbon tetaiodide • • Try: Oxygen difluoride Diiodine hexachloride Phosphorus pentabromide
- Slides: 7