340 B Overview and Update Ryan Witz VP

340 B Overview and Update Ryan Witz VP, Healthcare Financing Initiatives

Agenda • Overview of the 340 b program • CA Governor’s Executive Order • Proposed Federal Changes

340 B Drug Discount Program

340 B Drug Discount Program created by Congress in 1992 to help eligible safety -net “providers to stretch scarce Federal resources as far as possible, reaching more eligible patients and providing more comprehensive services” H. R. Rep. No. 102 -384(II), at 12 (1992).

340 B Drug Discount Program Overview ● Provides financial relief from high prescription costs to certain safety net hospitals ● Requires drug manufacturers participating in Medicaid to sell outpatient drugs at discounted rates to healthcare organizations that care for a significant number of uninsured and low-income patients There are 180 340 B hospitals across over 1, 800 sites in CA ● Allows hospitals to provide free or discounted drugs to low-income patients and expand health services ● Program has undergone significant changes and remains threatened

340 B Drug Discount Program • Administered by the Office of Pharmacy Affairs (OPA) within the Health Resources and Services Administration (HRSA) • The 340 B program requires manufacturers that participate in Medicaid and Medicare Part B to provide discounts to 340 B providers on outpatient drugs • California Medicaid (Medi-Cal) is a mandatory carve-in state

340 B Drug Discount Program 340 B-Eligible Providers Types
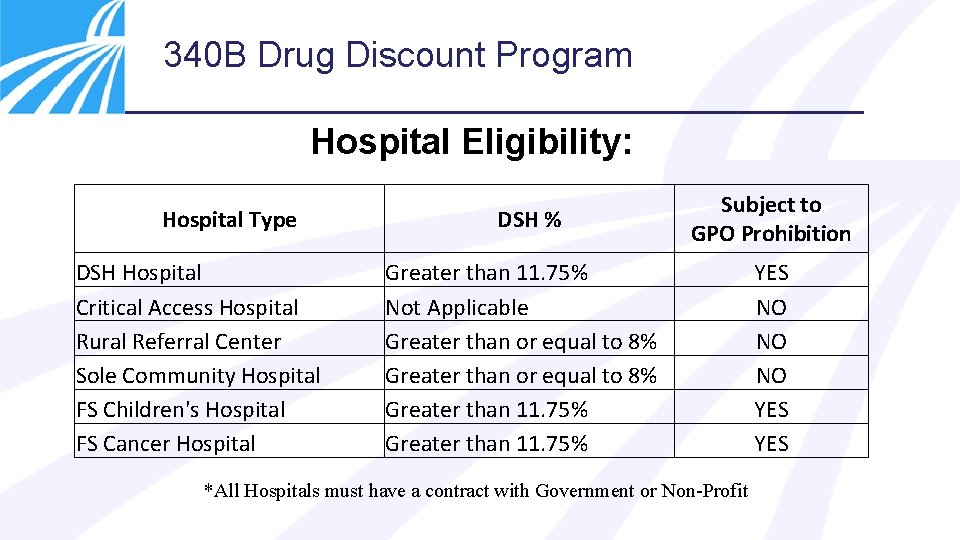
340 B Drug Discount Program Hospital Eligibility: Hospital Type DSH Hospital Critical Access Hospital Rural Referral Center Sole Community Hospital FS Children's Hospital FS Cancer Hospital DSH % Subject to GPO Prohibition Greater than 11. 75% Not Applicable Greater than or equal to 8% Greater than 11. 75% *All Hospitals must have a contract with Government or Non-Profit YES NO NO NO YES

340 B Drug Discount Program • Disproportionate share hospitals, free standing children’s hospitals, and free standing cancer hospitals may not use a group purchasing organization (GPO) or other group purchasing arrangement when purchasing covered outpatient drugs • Complying with the GPO prohibition is a statutory requirement • Hospitals can still use a GPO for inpatient drugs

340 B Drug Discount Program • State’s can choose whether to “Carve-in” or “Carve-out” the requirements for Covered Entity’s participation in Medicaid • California requires 340 B Covered Entities to carve-in for Medi-Cal • 340 B Hospital must list on OPA Medicaid Exclusion File Database all Medicaid billing numbers or National Provider Identifiers used • Hospitals must indicate on the OPA website they will not bill Medicaid FFS in the carve-out states. • Medi-Cal FFS reimbursement is limited to AAC + a dispensing fee

340 B Drug Discount Program • Medi-Cal managed care has become California’s delivery system of choice for Medicaid, with over 10. 6 million enrolled in a plan • ACA expanded Medicaid Agencies’ ability to claim rebates on Medicaid managed care • Significant challenges exist for the state with duplicate discounts • Medi-Cal MCP reimbursement is not limited to AAC + a dispensing fee

340 B Drug Discount Program • In-House vs. Contract Pharmacies • Growth of contract pharmacy arrangements • 2010 – 1, 300 arrangements • 2017 – 20, 000 arrangements • In 2017, 30% of CE’s utilize contract pharmacies • Duplicate discount issues https: //www. gao. gov/assets/700/692697. pdf

340 B Program Under Attack • State and Federal proposals to reduce the size of the program and increase transparency • Moratorium on new 340 B DSH hospitals (340 B PAUSE Act) • Onerous reporting requirements • Elimination of contract pharmacy arrangements in 340 B • California proposal to prohibit 340 B drugs from being dispensed to Medi-Cal managed care and fee-for-service beneficiaries

CA Governor’s Executive Order

Governor Newsom’s Executive Order on Drug Pricing ● Governor Newsom signed an executive order on drug pricing as his first act as governor ● Instructs DHCS to transition all pharmacy benefits for Medi-Cal managed care beneficiaries to fee-for-service by January 2021 ● This transition would impact reimbursement for 340 B-covered pharmacy drugs

Governor Newsom’s Executive Order on Drug Pricing ● Transition of pharmacy benefit from Medi-Cal managed care to FFS does not require additional approval from the legislature or federal government ● “Medi-Cal Rx” carve-out includes the following when billed by a pharmacy on a pharmacy claim: ● Covered Outpatient Drugs, including Physician Administered Drugs, ● Medical Supplies, ● Enteral Nutritional Products

Governor Newsom’s Executive Order on Drug Pricing ● Medi-Cal Rx will not impact prescriptions billed on a medical or institutional claim by a pharmacy, or any other provider type. ● What will not change? ● The scope of the existing Medi-Cal pharmacy benefit ● Provision of pharmacy services in an inpatient or long-term care setting, regardless of delivery system ● Existing Medi-Cal managed care pharmacy carve-outs (e. g. , blood factor, HIV/AIDS drugs, antipsychotics, or drugs used to treat substance use disorder) ● The State Fair Hearing process

Governor Newsom’s Executive Order on Drug Pricing ● DHCS has released a Request for Proposal to contract with a vendor to oversee the Medi-Cal FFS pharmacy benefit. ● The PBM will be responsible for: – Claims and Utilization Management – Pharmacy Drug Rebate Administration – Provider and Beneficiary Support ● Proposals are due back to DHCS by October 1, 2019 ● Contract will be awarded November 5, 2019
Governor Newsom’s Executive Order on Drug Pricing

Governor Newsom’s Executive Order on Drug Pricing ● 2019/20 state budget directs the Department of Health Care Services to establish pharmacy advisory group ● Inform and receive feedback from stakeholders on changes for managed care plans and beneficiaries ● Update the advisory group on expected savings and changes to beneficiary access to medication ● Next stakeholder meeting is September 26 th at 2 pm in Sacramento ● WEBINAR INFORMATION: To join the webinar, participants can click on the following link, and follow the instructions. https: //dhcs. webex. com/dhcs/onstage/g. php? MTID=e 716 a 307 ab 683111 c 5 b 06 c 9 d 244705 bdf

Proposed Federal Changes

AHA Lawsuit Challenging CY 2018 and CY 2019 OPPS Payment Cuts to 340 B Hospitals • U. S. District Court ruled in favor of AHA and found the payment cuts to 340 B hospitals to be unlawful • Department of Health and Human Services appealed the ruling and it is with the D. C. Circuit Court • Hospitals are encouraged to issue appeals for highcost 340 B drugs to ensure your time limit for appeals is preserved • Once the appeal is denied, hospitals are encouraged to file a second level appeal

AHA Lawsuit Challenging CY 2018 and CY 2019 OPPS Payment Cuts to 340 B Hospitals • Following the Government's appeal to the appellate court in DC, the AHA requested an expedited briefing schedule • Court’s timeline is short—briefing schedule is by mid-Oct • The Government did meet their required date for filing their first brief on 9/3/2019

CMS Issues CY 2020 OPPS Proposed Rule • 340 b Drug Pricing Program: For CY 2020, CMS proposes to continue its current 340 B drug payment adjustment of average sales price minus 22. 5 percent. • CMS also acknowledges the ongoing litigation pertaining to the 340 B payment adjustment and seeks comment on alternative payment options for CY 2020 and potential remedies for CY 2018 and CY 2019 payments in the event of an adverse ruling on the 340 B payment policy by the United States Court of Appeals.

AHA Commitment to Good Stewardship Principles for the 340 B Program • By committing to Good Stewardship Principles, 340 B hospitals would: • Communicate the Value of the 340 B Program • Disclose Hospital’s 340 B Estimated Savings • Continue Rigorous Internal Oversight • Hospitals are encouraged to sign the pledge • 85% of 340 B Hospitals in CA signed on • List of signees will be made public in October • FAQs

Questions? Ryan Witz VP, Health Care Financing Initiatives California Hospital Association rwitz@calhospital. org 916 -552 -7642
- Slides: 26