33 B4 Applications Figure 33 11 illustrates typical

33 B-4 Applications Figure 33 -11 illustrates typical applications of bonded-phase partition chromatography for separating soft drink additives and organophosphate insecticides. Table 33 -2 further illustrates the variety of samples to which the technique is applicable.
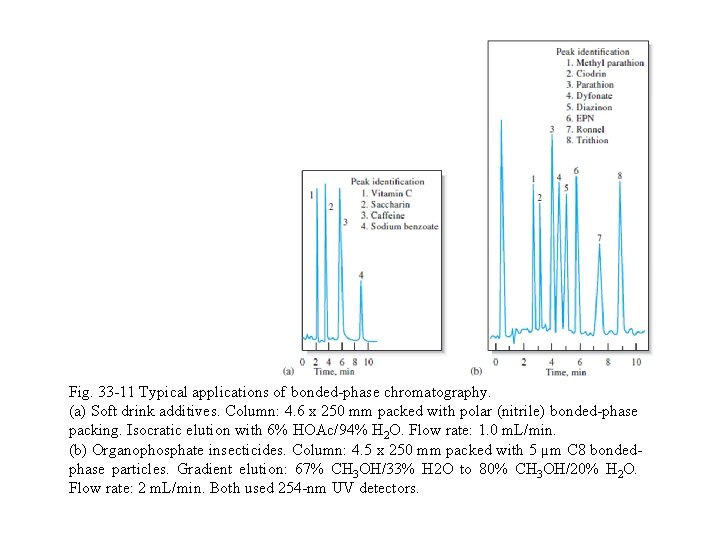
Fig. 33 -11 Typical applications of bonded-phase chromatography. (a) Soft drink additives. Column: 4. 6 x 250 mm packed with polar (nitrile) bonded-phase packing. Isocratic elution with 6% HOAc/94% H 2 O. Flow rate: 1. 0 m. L/min. (b) Organophosphate insecticides. Column: 4. 5 x 250 mm packed with 5 µm C 8 bondedphase particles. Gradient elution: 67% CH 3 OH/33% H 2 O to 80% CH 3 OH/20% H 2 O. Flow rate: 2 m. L/min. Both used 254 -nm UV detectors.


33 C Adsorption Chromatography Adsorption, or liquid-solid, chromatography is the classic form of liquid chromatography first introduced by Tswett at the beginning of the twentieth century. Because of the strong overlap between normal-phase partition chromatography and adsorption chromatography, many of the principles and techniques used for the former apply to adsorption chromatography. In fact, in many normal-phase separations, adsorption/displacement processes govern retention.

Finely divided silica and alumina are the only stationary phases that find use for adsorption chromatography. Silica is preferred for most applications because of its higher sample capacity. The adsorption characteristics of the two substances parallel one another. For both, retention times become longer as the polarity of the analyte increases.

Because of the versatility and ready availability of bonded stationary phases, traditional adsorption chromatography with solid stationary phases has seen decreasing use in recent years in favor of normal-phase chromatography

33 D Ion Chromatography Ion-exchange resin materials are useful as stationary phases for liquid chromatography where they are used to separate charged species. Ion chromatography as it is practiced today was first developed in the mid-1970 s when it was shown that anion or cation mixtures can be resolved on HPLC columns packed with anionexchange or cation-exchange resins.

At that time, detection was generally performed with conductivity measurements, which were not ideal because of high electrolyte concentrations in the mobile phase. The development of low-exchange capacity columns allowed the use of low-ionic-strength mobile phases that could be further deionized (ionization suppressed) to allow high sensitivity conductivity detection. Currently, several other detector types are available for ion chromatography, including spectrophotometric and electrochemical.

33 D-1 Ion Chromatography based on suppressors Conductivity detectors have many of the properties of the ideal detector. They can be highly sensitive, they are universal for charged species, and as a general rule, they respond in a predictable way to concentration changes. Furthermore, such detectors are simple to operate, inexpensive to construct and maintain, easy to miniaturize, and usually give prolonged, trouble-free service.

The only limitation to the use of conductivity detectors, which delayed their general application to ion chromatography until the mid-1970 s, was due to the high electrolyte concentrations required to elute most analyte ions in a reasonable time. Such a problem solved by using ionic mobile phase suppresser by adding ions that form molecular products.

The conductivity detector is well suited for ion chromatography.

In suppressor-based ion chromatography, the ion-exchange column is followed by a suppressor column, or a suppressor membrane, that converts an ionic eluent into a nonionic species that does not interfere with the conductometric detection of analyte ions.

Figure 33 -12 shows two applications of ion chromatography based on a suppressor column and conductometric detection. In each, the ions were present in the parts-per-million range; the sample size was 50 m. L in one case and 100 m. L in the other. The method is particularly important for anion analysis because there is no other rapid and convenient method for handling mixtures of this type.
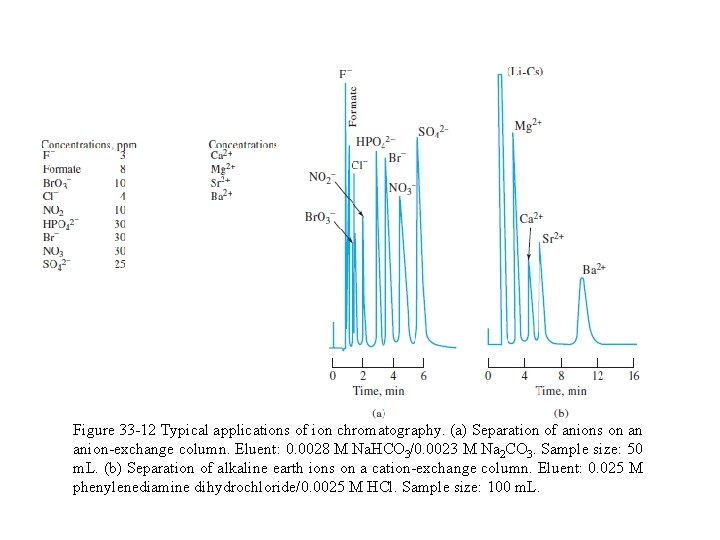
Figure 33 -12 Typical applications of ion chromatography. (a) Separation of anions on an anion-exchange column. Eluent: 0. 0028 M Na. HCO 3/0. 0023 M Na 2 CO 3. Sample size: 50 m. L. (b) Separation of alkaline earth ions on a cation-exchange column. Eluent: 0. 025 M phenylenediamine dihydrochloride/0. 0025 M HCl. Sample size: 100 m. L.

33 E Size-Exclusion Chromatography

In size-exclusion chromatography, fractionation is based on molecular size.

Size-exclusion, or gel chromatography, is a powerful technique that is articularly applicable to high-molecular-mass species. Packings for size-exclusion chromatography consist of small (~10 µm) silica or polymer particles containing a network of uniform pores into which solute and solvent molecules can diffuse. While in the pores, molecules are effectively trapped and removed from the flow of the mobile phase.

The average residence time of analyte molecules depends on their effective size. Molecules that are significantly larger than the average pore size of the packing are excluded and thus suffer no retention, that is, they travel through the column at the rate of the mobile phase. Molecules that are appreciably smaller than the pores can penetrate throughout the pore maze and are thus entrapped for the greatest time; they are last to elute.

Between these two extremes are intermediate-size molecules whose average penetration into the pores of the packing depends on their diameters. The fractionation that occurs within this group is directly related to molecular size and, to some extent, molecular shape. Note that size-exclusion separations differ from the other chromatographic procedures in the respect that there are no chemical or physical interactions between analytes and the stationary phase.

Indeed, such interactions are avoided because they lead to lower column efficiencies. Note also that, unlike other forms of chromatography, there is an upper limit to retention time because no analyte species is retained longer than those small molecules that totally permeate the stationary phase.
- Slides: 20