3 Szerves vegyletek Alknok s cikloalknok Based on

3. Szerves vegyületek: Alkánok és cikloalkánok Based on Mc. Murry’s Organic Chemistry, 6 th edition, Chapter 3 © 2003 Ronald Kluger Department of Chemistry University of Toronto

A szerves vegyületek csoportjai n A szerves vegyületeket csoportokba lehet sorolni közös szerkezeti jellemzőik alapján n Ezen kurzus keretében az egyes vegyületek sajátságait ismerjük meg, úgy, hogy a vegyületcsoportok sajátságait tárgyaljuk n Ez a fejezet az alkánokkal, olyan vegyületekkel foglalkozik, amelyek csak szén- és hidrogénatomokat tartalmaznak, amelyek mindegyike kizárólag egyszeres kötéssel kapcsolódik össze Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 2
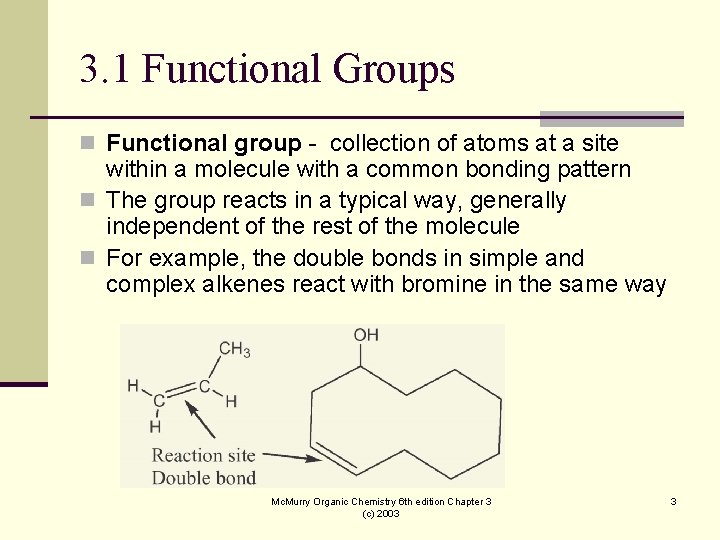
3. 1 Functional Groups n Functional group - collection of atoms at a site within a molecule with a common bonding pattern n The group reacts in a typical way, generally independent of the rest of the molecule n For example, the double bonds in simple and complex alkenes react with bromine in the same way Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 3

Survey of Functional Groups n Table 3. 1 lists a wide variety of functional groups that you should recognize n As you learn about them in each chapter it will be easier to recognize them n The functional groups affect the reactions, structure, and physical properties of every compound in which they occur Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 4

Types of Functional Groups: Multiple Carbon–Carbon Bonds n Alkenes have a C-C double bond n Alkynes have a C-C triple bond n Arenes have special bonds that are represented as alternating single and double C-C bonds in a sixmembered ring Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 5

Functional Groups with Carbon Singly Bonded to an Electronegative Atom n Alkyl halide: C bonded to halogen (C-X) n Alcohol: C bonded O of a hydroxyl group (C-OH) n Ether: Two C’s bonded to the same O (C-O-C) n Amine: C bonded to N (C-N) n Thiol: C bonded to SH group (C-SH) n Sulfide: Two C’s bonded to same S (C-S-C) n Bonds are polar, with partial positive charge on C ( +) and partial negative charge ( ) on electronegative atom Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 6

Groups with a Carbon–Oxygen Double Bond (Carbonyl Groups) n Aldehyde: one hydrogen bonded to C=O n Ketone: two C’s bonded to the C=O n Carboxylic acid: OH bonded to the C=O n Ester: C-O bonded to the C=O n Amide: C-N bonded to the C=O n Acid chloride: Cl bonded to the C=O n Carbonyl C has partial positive charge ( +) n Carbonyl O has partial negative charge ( -). Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 7

3. 2 Alkanes and Alkane Isomers n Alkanes: Compounds with C-C single bonds and C-H n n bonds only (no functional groups) Connecting carbons can lead to large or small molecules The formula for an alkane with no rings in it must be Cn. H 2 n+2 where the number of C’s is n Alkanes are saturated with hydrogen (no more can be added They are also called aliphatic compounds Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 8

Alkane Isomers n CH 4 = methane, C 2 H 6 = ethane, C 3 H 8= propane n The molecular formula of an alkane with more than three carbons can give more than one structure n n C 4 (butane) = butane and isobutane C 5 (pentane) = pentane, 2 -methylbutane, and 2, 2 dimethylpropane n Alkanes with C’s connected to no more than 2 other C’s are straight-chain or normal alkanes n Alkanes with one or more C’s connected to 3 or 4 C’s are branched-chain alkanes Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 9

Constitutional Isomers n Isomers that differ in how their atoms are arranged in chains are called constitutional isomers n Compounds other than alkanes can be constitutional isomers of one another n They must have the same molecular formula to be isomers Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 10
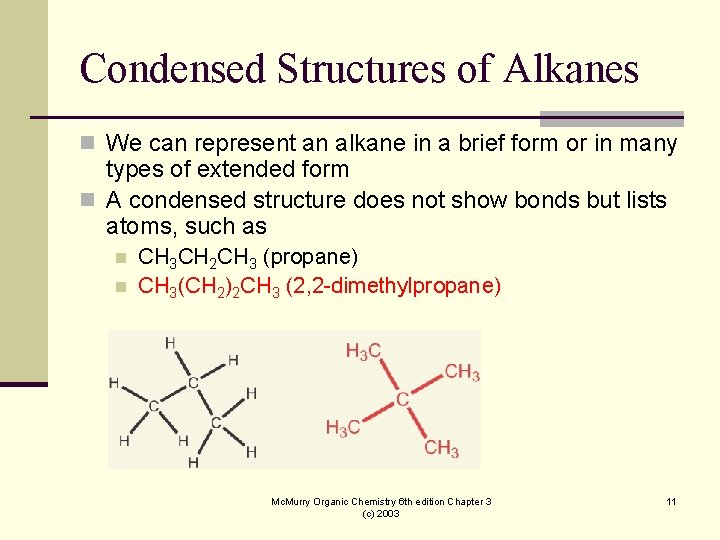
Condensed Structures of Alkanes n We can represent an alkane in a brief form or in many types of extended form n A condensed structure does not show bonds but lists atoms, such as n n CH 3 CH 2 CH 3 (propane) CH 3(CH 2)2 CH 3 (2, 2 -dimethylpropane) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 11
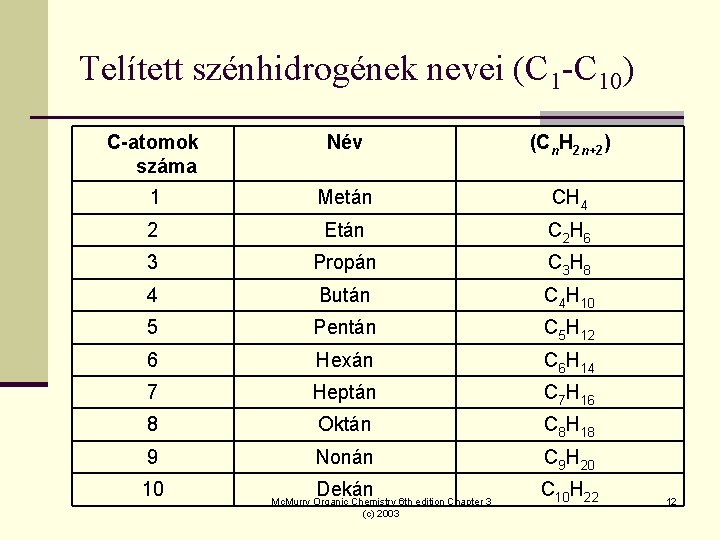
Telített szénhidrogének nevei (C 1 -C 10) C-atomok száma Név (Cn. H 2 n+2) 1 Metán CH 4 2 Etán C 2 H 6 3 Propán C 3 H 8 4 Bután C 4 H 10 5 Pentán C 5 H 12 6 Hexán C 6 H 14 7 Heptán C 7 H 16 8 Oktán C 8 H 18 9 Nonán C 9 H 20 Dekán C 10 H 22 10 Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 12

Telített szénhidrogének nevei (C 11 -C 20) C-atomok száma Név (Cn. H 2 n+2) 11 Undekán C 11 H 24 12 Dodekán C 12 H 26 13 Tridekán C 13 H 28 14 Tetradekán C 14 H 30 15 Pentadekán C 15 H 32 16 Hexadekán C 16 H 34 17 Heptadekán C 17 H 36 18 Oktadekán C 18 H 38 19 Nonadekán C 19 H 40 20 Eikozán C 20 H 42 Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 13

3. 3 Alkilcsoportok n Alkilcsoport – eltávolítunk egy hidrogént az alkánból (a szerkezet része) n Rövidítés: “R” (Radical) n Nem valós részecskék n Név: az alkánok -án végződését –il-re cseréljük -CH 3: “metil” (a metánból) n -CH 2 CH 3: “etil” (az etánból) n Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 14

Types of Alkyl groups n Classified by the connection site (See Figure 3. 3) n a carbon at the end of a chain (primary alkyl group) n a carbon in the middle of a chain (secondary alkyl group) n a carbon with three carbons attached to it (tertiary alkyl group) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 15

3. 4 A szerves vegyületek nevezéktana n Triviális nevek n Genfi nómenklatúra (1892) n Szisztematikus nómenklatúra n Részletesen ezt tanulmányozzuk n IUPAC (International Union of Pure and Applied Chemistry) n Továbbiak n Csoportfunkciós (pl. metil-klorid, CH 3 Cl) n Helyettesítéses (pl. oxaciklohexán) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 16

Blue book BLUE BOOK Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013, IUPAC Blue book, prepared for publication by Henri A Favre and Warren H Powell, by RSC Publishing, 2014 [ISBN 978 -0 -85404 -182 -4]; https: //doi. org/10. 1039/9781849733069 Online release Contents list and errata (www prepared by G. P. Moss) An HTML production of the IUPAC Nomenclature of Organic Chemistry has been prepared by Advanced Chemistry Development, Inc and is searchable on the ACD/Labs website <https: //www. acdlabs. com/iupac/nomenclature>. https: //www. qmul. ac. uk/sbcs/iupac/bibliog/BBcontents. html Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 17

1998 Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 18

3. 4 Az alkánok nevezéktana n A vegyületeknek a következő általános szerkezetű szisztematikus nevet adjuk: n Előtag(ok) – alapszénhidrogén szótöve – végződés(ek) n Szabályok n Kiválasztjuk a leghosszabb szénláncot n A szénláncban megszámozzuk a szénatomokat n A szubsztituensek helyzetét az a szénatom adja meg, amelyikhez kapcsolódnak n A vegyületnév egy szó n Az összetett szubsztituenseket úgy nevezzük el, mintha önálló vegyületek lennének (-il végződéssel) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 19

Kémiai rajzolóprogramok Chem. Draw, Chem 3 D Chem. Sketch (ACDLabs) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 20

3. 5 Properties of Alkanes n Called paraffins (low affinity compounds) because they do not react as most chemicals n They will burn in a flame, producing carbon dioxide, water, and heat n They react with Cl 2 in the presence of light to replace H’s with Cl’s (not controlled) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 21
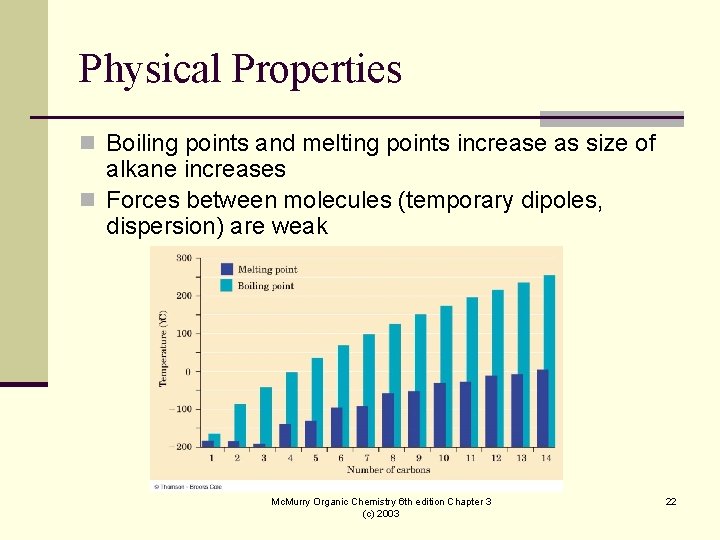
Physical Properties n Boiling points and melting points increase as size of alkane increases n Forces between molecules (temporary dipoles, dispersion) are weak Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 22
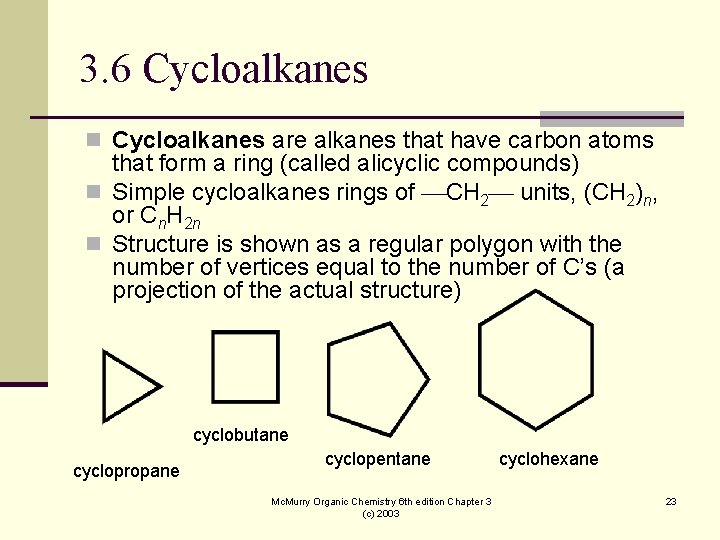
3. 6 Cycloalkanes n Cycloalkanes are alkanes that have carbon atoms that form a ring (called alicyclic compounds) n Simple cycloalkanes rings of CH 2 units, (CH 2)n, or Cn. H 2 n n Structure is shown as a regular polygon with the number of vertices equal to the number of C’s (a projection of the actual structure) cyclobutane cyclopropane cyclopentane Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 cyclohexane 23
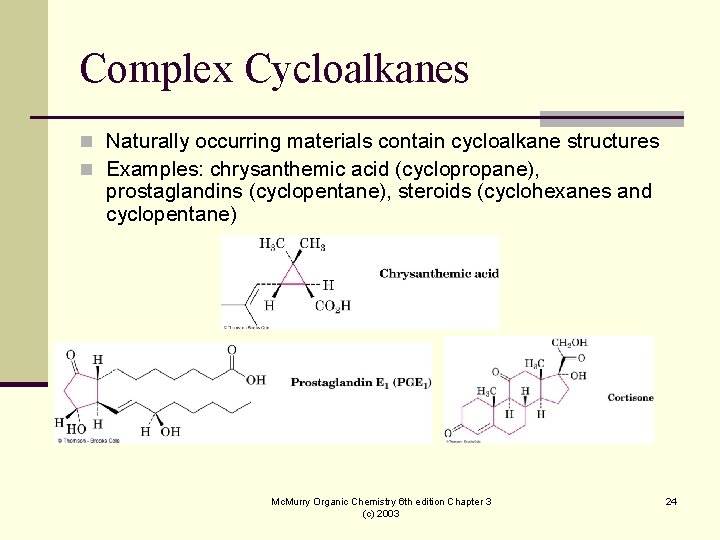
Complex Cycloalkanes n Naturally occurring materials contain cycloalkane structures n Examples: chrysanthemic acid (cyclopropane), prostaglandins (cyclopentane), steroids (cyclohexanes and cyclopentane) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 24
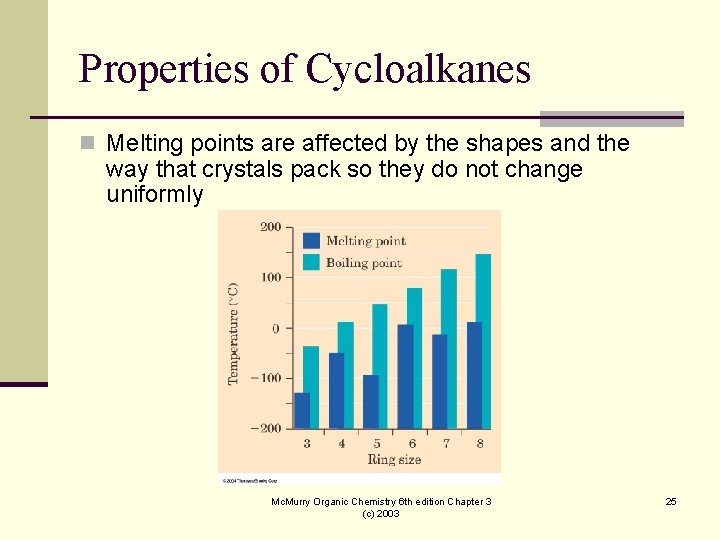
Properties of Cycloalkanes n Melting points are affected by the shapes and the way that crystals pack so they do not change uniformly Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 25

3. 7 Naming Cycloalkanes n Count the number of carbon atoms in the ring and the number in the largest substituent chain. If the number of carbon atoms in the ring is equal to or greater than the number in the substituent, the compound is named as an alkyl-substituted cycloalkane n For an alkyl- or halo-substituted cycloalkane, start at a point of attachment as C 1 and number the substituents on the ring so that the second substituent has as low a number as possible. n Number the substituents and write the name n See text for more details and examples Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 26
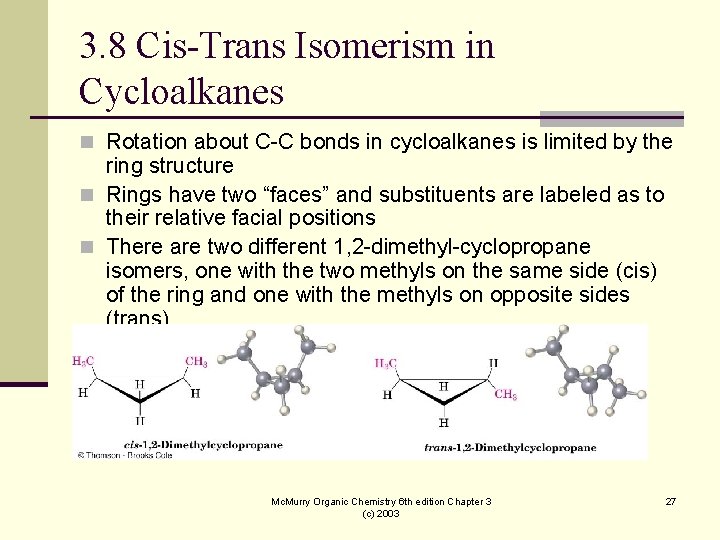
3. 8 Cis-Trans Isomerism in Cycloalkanes n Rotation about C-C bonds in cycloalkanes is limited by the ring structure n Rings have two “faces” and substituents are labeled as to their relative facial positions n There are two different 1, 2 -dimethyl-cyclopropane isomers, one with the two methyls on the same side (cis) of the ring and one with the methyls on opposite sides (trans) Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 27
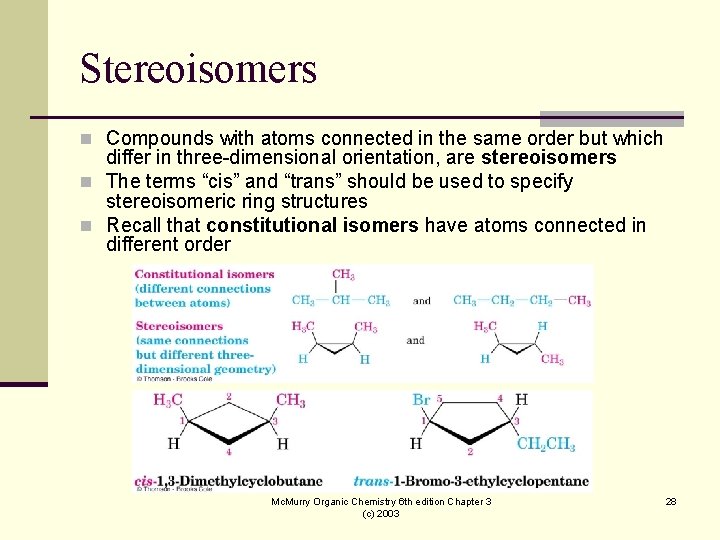
Stereoisomers n Compounds with atoms connected in the same order but which differ in three-dimensional orientation, are stereoisomers n The terms “cis” and “trans” should be used to specify stereoisomeric ring structures n Recall that constitutional isomers have atoms connected in different order Mc. Murry Organic Chemistry 6 th edition Chapter 3 (c) 2003 28
- Slides: 28