3 rd Kitasato Harvard Symposium 2 October 2002

3 rd Kitasato Harvard Symposium 2 October 2002 Simultaneous/Worldwide Development Strategies : New Challenges New Approaches of Dose Range Finding Prof. Yusuke Tanigawara Keio University Hospital Tokyo, Japan 1

Drug Action E=f(C(t), S) > E=f(Dose, S) Y. Tanigawara 2002 2
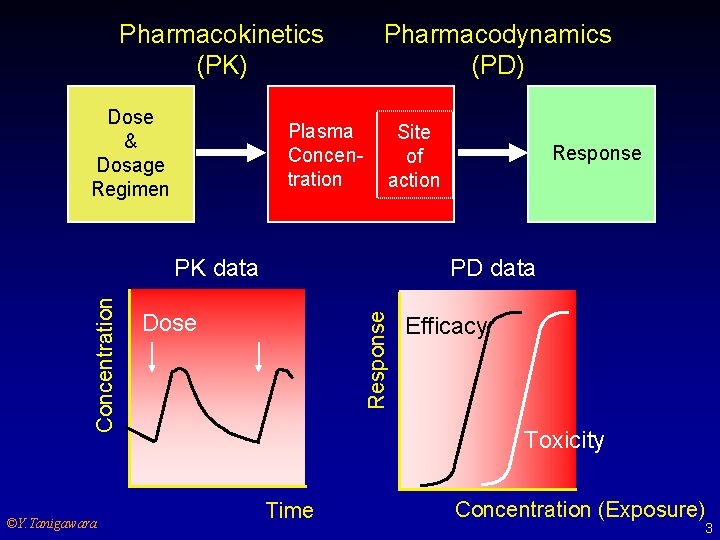
Pharmacokinetics (PK) Dose & Dosage Regimen Plasma Concentration Pharmacodynamics (PD) Site of action ©Y. Tanigawara PD data Dose Response Concentration PK data Response Efficacy Toxicity Time Concentration (Exposure) 3

Why PK/PD are needed. u u Human PK/PD, especially in patients, are important drug information. PK/PD provide a scientific framework for dose/dosage regimen vs concentrations vs response relationships. Factors affecting PK/PD are considered when dose is individualized for special populations such as geriatrics and organ dysfunction. PK/PD can be a “bridging” tool for introducing to new indications, new dosage forms, or new populations. Y. Tanigawara 2002 4
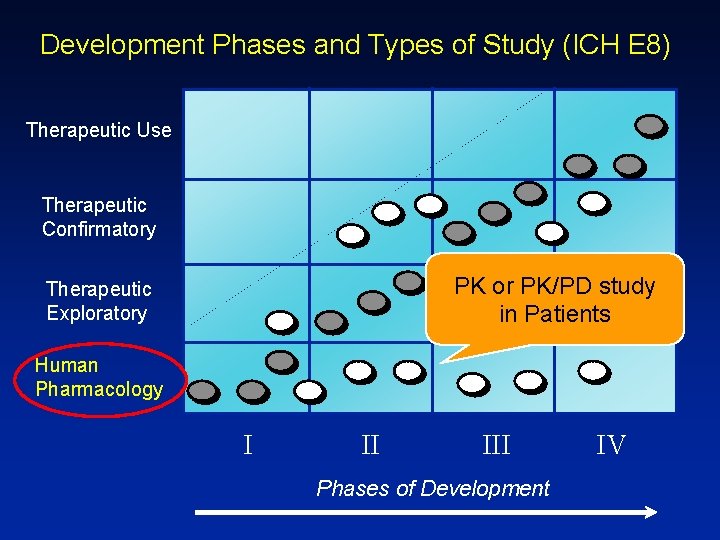
Development Phases and Types of Study (ICH E 8) Therapeutic Use Therapeutic Confirmatory PK or PK/PD study in Patients Therapeutic Exploratory Human Pharmacology I II III Phases of Development IV
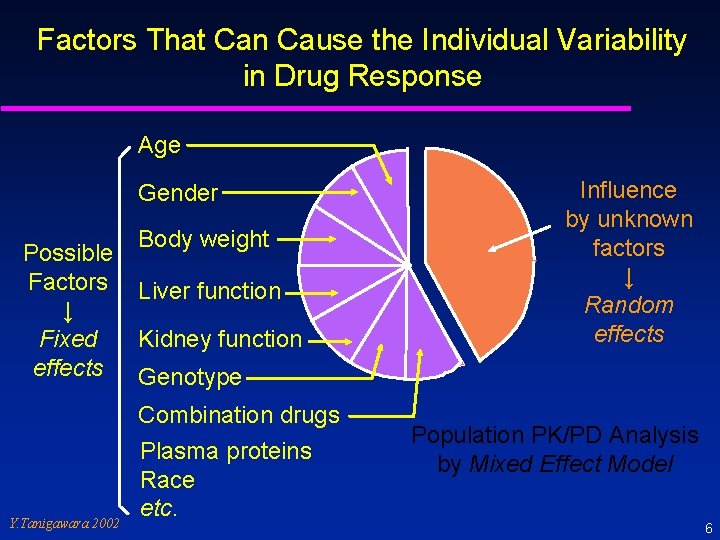
Factors That Can Cause the Individual Variability in Drug Response Age Gender Possible Factors ↓ Fixed effects Body weight Liver function Kidney function Genotype Combination drugs Y. Tanigawara 2002 Influence by unknown factors ↓ Random effects Plasma proteins Race etc. Population PK/PD Analysis by Mixed Effect Model 6

Population Pharmacokinetics (PK) and Pharmacodynamics (PD) ü ü ü describe typical profiles of PK/PD in a target population (patients that a drug is applied). describe magnitudes of inter- and intra-individual variability. describe factors that can affect the PK/PD of a drug (genetic, physiological, pathological, environmental). provide dosing guidance for special populations such as geriatrics, pediatrics, organ dysfunction, drug interactions, genetic deficiency of a particular enzyme. . . etc. provide a scientific basis for individualization of dosage regimen. can be studied based upon sparsely sampled data. → Feasible method to obtain patient data. Y. Tanigawara 2002 7
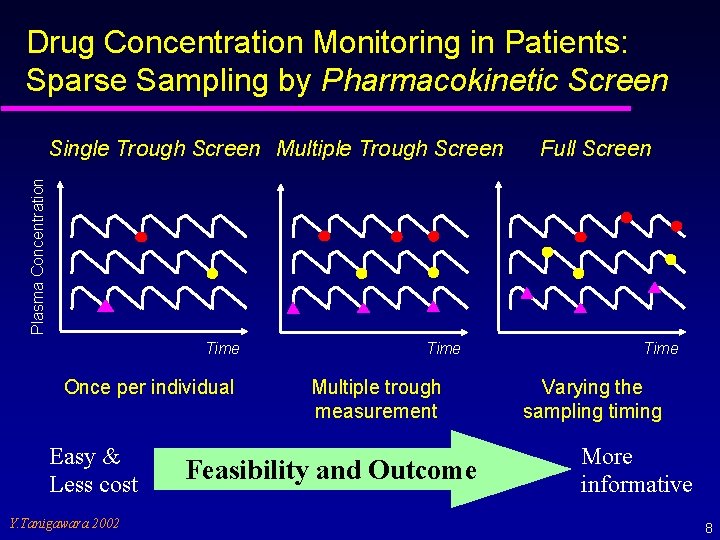
Drug Concentration Monitoring in Patients: Sparse Sampling by Pharmacokinetic Screen Full Screen Plasma Concentration Single Trough Screen Multiple Trough Screen Time Once per individual Easy & Less cost Y. Tanigawara 2002 Time Multiple trough measurement Feasibility and Outcome Time Varying the sampling timing More informative 8
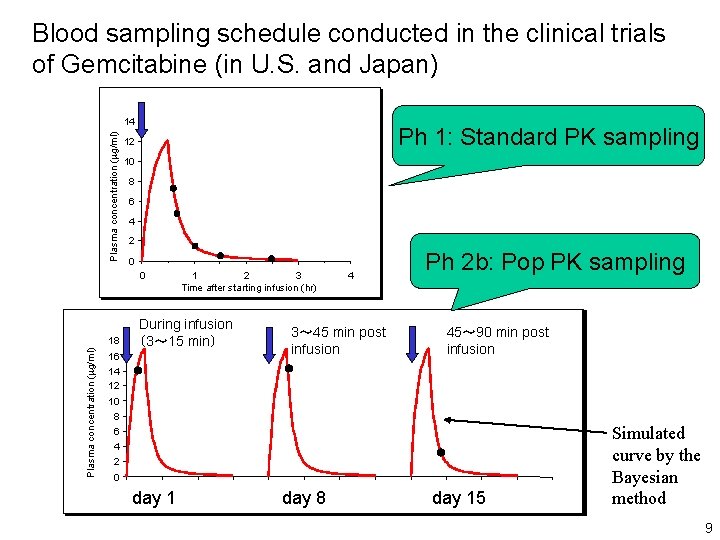
Blood sampling schedule conducted in the clinical trials of Gemcitabine (in U. S. and Japan) Plasma concentration (mg/ml) 14 Ph 1: Standard PK sampling 12 10 8 6 4 2 0 0 Plasma concentration (mg/ml) 18 During infusion (3~ 15 min) 16 14 12 1 2 3 Time after starting infusion (hr) ● 4 Ph 2 b: Pop PK sampling 3~ 45 min post infusion 45~ 90 min post infusion ● 10 8 6 4 ● 2 0 day 1 day 8 day 15 Simulated curve by the Bayesian method 9
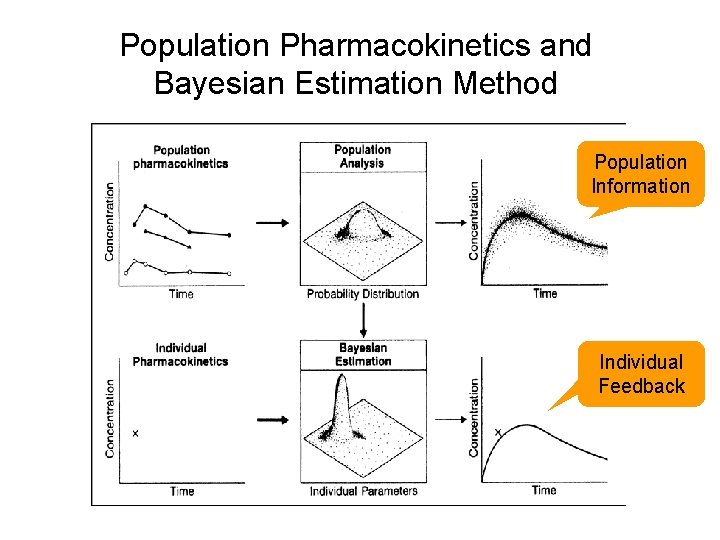
Population Pharmacokinetics and Bayesian Estimation Method Population Information Individual Feedback 10

PK/PD Relationship of Antibacterial Agents Plasma Concentration Efficacy of Aminoglycosides Peak AUC / MIC AUC Efficacy of New Quinolones Trough MIC Time above MIC 0 4 8 Safety of Aminoglycosides 12 16 Time (hr) Efficacy of b-lactam, macrolides, glycopeptides Y. Tanigawara 2002 11
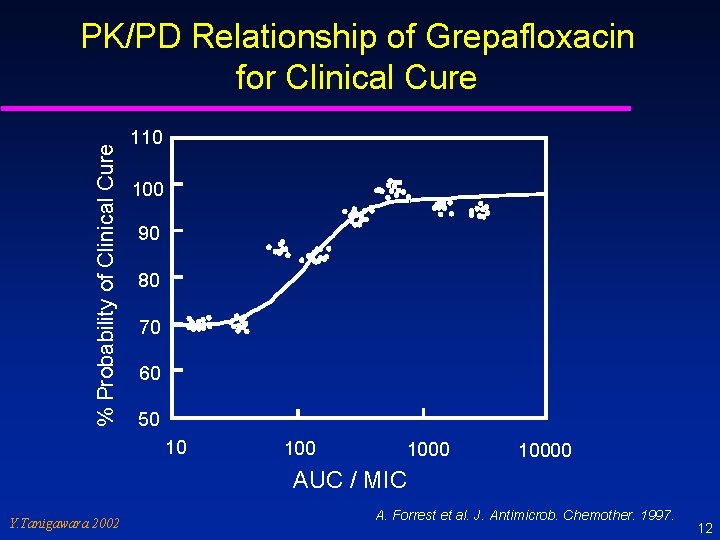
% Probability of Clinical Cure PK/PD Relationship of Grepafloxacin for Clinical Cure 110 100 90 80 70 60 50 10 10000 AUC / MIC Y. Tanigawara 2002 A. Forrest et al. J. Antimicrob. Chemother. 1997. 12

Bridging Study - ICH E 5 - A supplemental study performed in the new region to provide pharmacodynamic or clinical data on efficacy, safety, dosage and dose regimen in the new region that will allow extrapolation of the foreign clinical data package to the new region. Such studies could include further pharmacokinetic information. Definition of not only PK but also PD and doseresponse early in the development program may facilitate the determination of the need for, and nature of, any requisite bridging data. Y. Tanigawara 2002 13
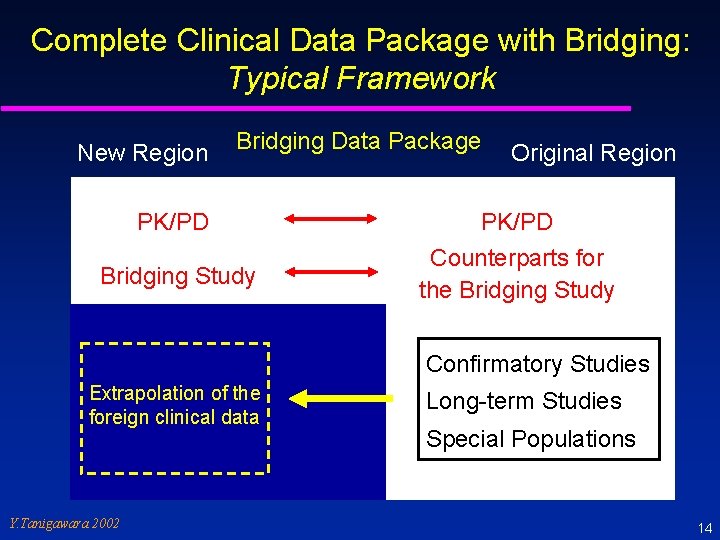
Complete Clinical Data Package with Bridging: Typical Framework New Region Bridging Data Package PK/PD Bridging Study Original Region PK/PD Counterparts for the Bridging Study Confirmatory Studies Extrapolation of the foreign clinical data Y. Tanigawara 2002 Long-term Studies Special Populations 14
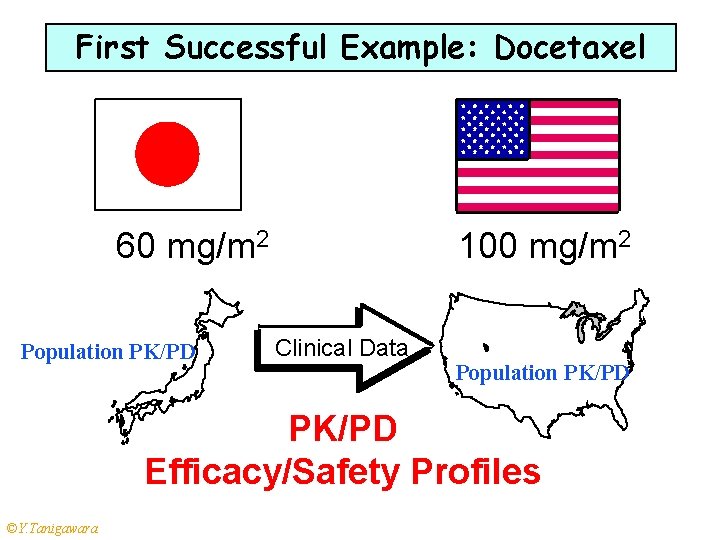
First Successful Example: Docetaxel 60 mg/m 2 Population PK/PD 100 mg/m 2 Clinical Data Population PK/PD Efficacy/Safety Profiles ©Y. Tanigawara 15

Interpretation of PK Data (1) u Clinical PK data in Japanese are essential for the complete clinical data package. u If there is a difference… u Conduct a population PK analysis to gain an insight into the observed ethnic difference. u It might be caused due to different body sizes between Japanese and Caucasian. u It might be caused by different enzymatic activity. u It might be a consequence of different food conditions (low fat, high fat, fasted, non-fasted). u Explainable … ? Y. Tanigawara 2002 16
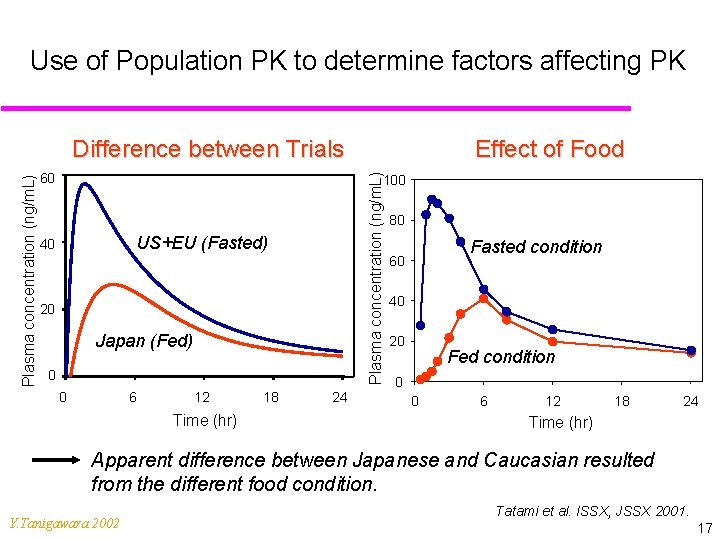
Use of Population PK to determine factors affecting PK 60 Effect of Food Plasma concentration (ng/m. L) Difference between Trials 100 US+EU (Fasted) 40 20 Japan (Fed) 0 0 6 12 Time (hr) 18 24 80 Fasted condition 60 40 20 Fed condition 0 0 6 12 18 24 Time (hr) Apparent difference between Japanese and Caucasian resulted from the different food condition. Y. Tanigawara 2002 Tatami et al. ISSX, JSSX 2001. 17

Interpretation of PK Data (2) u Clinical PK data in Japanese are essential for the complete clinical data package. u If there is a PK difference… u How much PK difference impacts on clinical efficacy and safety. Need to modify dose? u Require a PK/PD or dose-response relationship to consider the influence of PK difference. u Secondary use of a BE criteria when PK/PD data are absent. Y. Tanigawara 2002 18
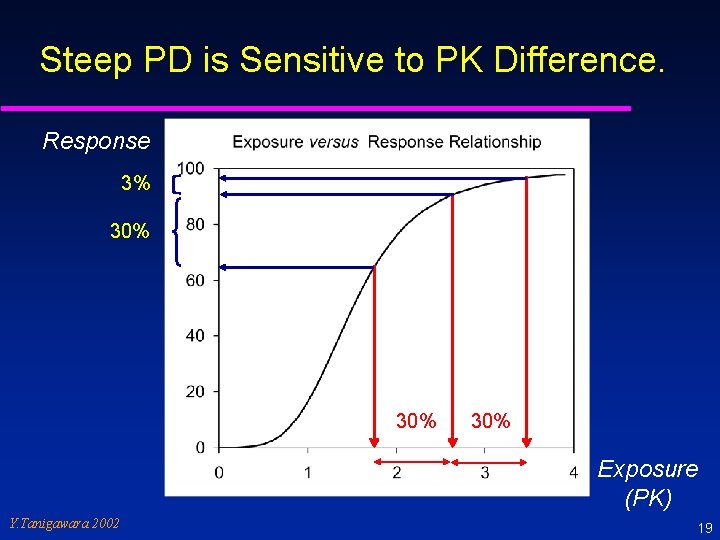
Steep PD is Sensitive to PK Difference. Response 3% 30% 30% Exposure (PK) Y. Tanigawara 2002 19

Pharmacogenomics Genetic polymorphisms of drug metabolizing enzymes Altered PK Altered PD Altered Efficacy/Safety profiles Influences of genotype are attributed to individual variability, rather than racial difference. Y. Tanigawara 2002 20
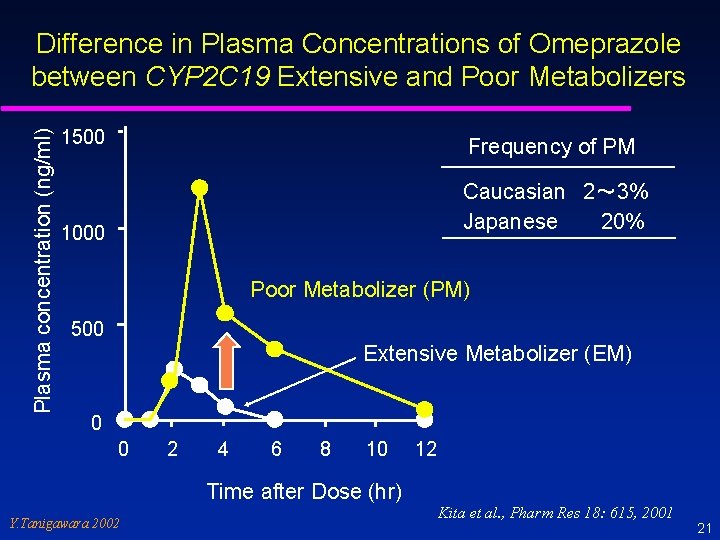
Plasma concentration (ng/ml) Difference in Plasma Concentrations of Omeprazole between CYP 2 C 19 Extensive and Poor Metabolizers 1500 Frequency of PM Caucasian 2~ 3% Japanese 20% 1000 Poor Metabolizer (PM) 500 Extensive Metabolizer (EM) 0 0 2 4 6 8 10 12 Time after Dose (hr) Y. Tanigawara 2002 Kita et al. , Pharm Res 18: 615, 2001 21
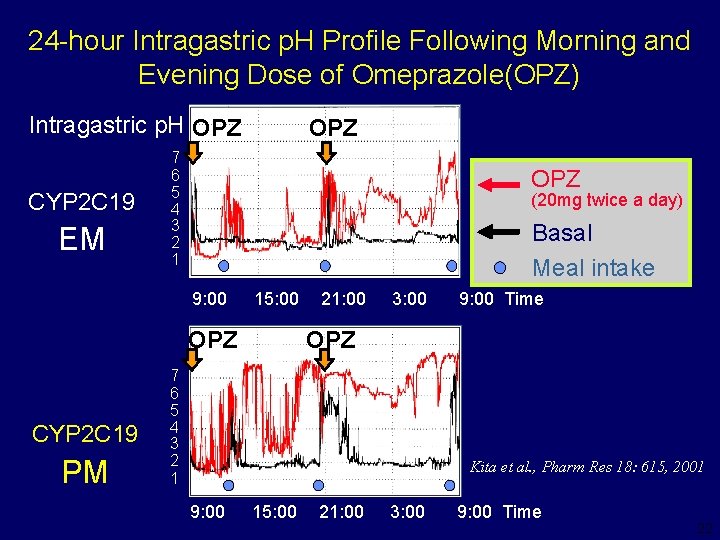
24 -hour Intragastric p. H Profile Following Morning and Evening Dose of Omeprazole(OPZ) Intragastric p. H OPZ CYP 2 C 19 EM OPZ 7 6 5 4 3 2 1 OPZ (20 mg twice a day) Basal Meal intake 9: 00 15: 00 OPZ CYP 2 C 19 PM 21: 00 3: 00 9: 00 Time OPZ 7 6 5 4 3 2 1 Kita et al. , Pharm Res 18: 615, 2001 9: 00 15: 00 21: 00 3: 00 9: 00 Time 22
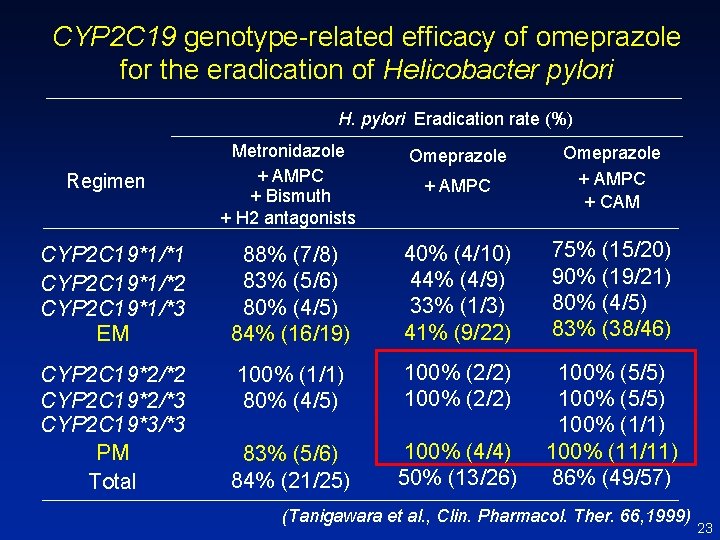
CYP 2 C 19 genotype-related efficacy of omeprazole for the eradication of Helicobacter pylori H. pylori Eradication rate (%) Metronidazole + AMPC + Bismuth + H 2 antagonists Omeprazole + AMPC + CAM CYP 2 C 19*1/*1 CYP 2 C 19*1/*2 CYP 2 C 19*1/*3 EM 88% (7/8) 83% (5/6) 80% (4/5) 84% (16/19) 40% (4/10) 44% (4/9) 33% (1/3) 41% (9/22) 75% (15/20) 90% (19/21) 80% (4/5) 83% (38/46) CYP 2 C 19*2/*2 CYP 2 C 19*2/*3 CYP 2 C 19*3/*3 PM Total 100% (1/1) 80% (4/5) 100% (2/2) 83% (5/6) 84% (21/25) 100% (4/4) 50% (13/26) 100% (5/5) 100% (1/1) 100% (11/11) 86% (49/57) Regimen (Tanigawara et al. , Clin. Pharmacol. Ther. 66, 1999) 23

Summary u PK/PD provides a scientific basis for dose range finding. u Population PK/PD analysis coupled with the sparse blood sampling is an important strategy for drug development. u Pharmacogenomics will be a useful approach for targeting patient population. Y. Tanigawara 2002 24
- Slides: 24