3 Major systems involved in thrombosis and hemostasis


3 Major systems involved in thrombosis and hemostasis Vessel wall – Endothelium Platelets Coagulation cascade

Anti-platelet and Anti-coagulant Properties of the Endothelium – Covers highly thrombogenic basement membrane (collagen, TF). Uninjured endothelium does not bind platelets – NO from uninjured endothelium inhibit platelet aggregation and adhesion, PGI 2 (prostacyclin) inhibits platelet aggregation – TFPI – tissue factor pathway inhibitor – released from endothelial cells – Endothelial cells produce t- PA which activates fibrinolysis via plasminogen to plasmin

Platelet Adhesion Platelets are the first cells to adhere to injured vascular wall (subendothelium) Adhesion is mediated by v. WF Binding occurs only under high shear stress conditions !
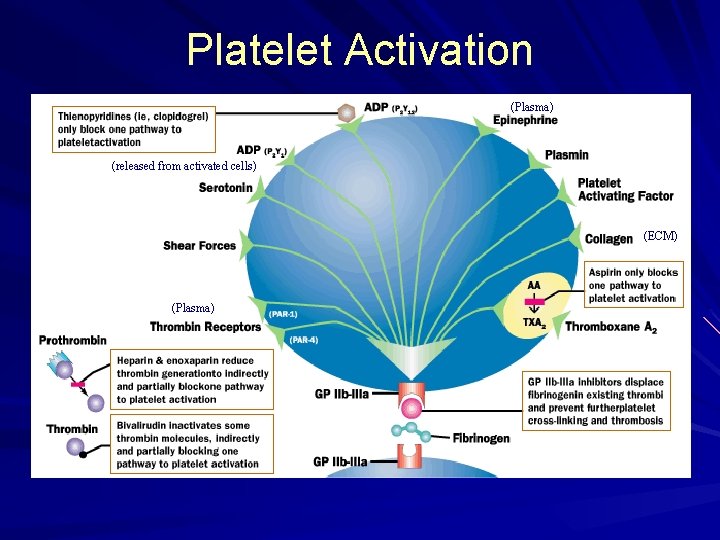
Platelet Activation (Plasma) (released from activated cells) (ECM) (Plasma)
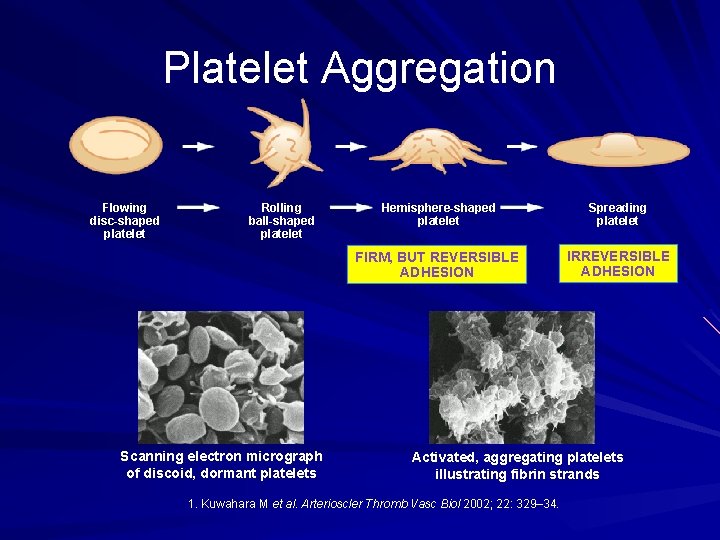
Platelet Aggregation Flowing disc-shaped platelet Rolling ball-shaped platelet Scanning electron micrograph of discoid, dormant platelets Hemisphere-shaped platelet Spreading platelet FIRM, BUT REVERSIBLE ADHESION IRREVERSIBLE ADHESION Activated, aggregating platelets illustrating fibrin strands 1. Kuwahara M et al. Arterioscler Thromb Vasc Biol 2002; 22: 329– 34.

3 Major systems involved Vessel wall – Endothelium Platelets Coagulation cascade

“Classic Coagulation Cascade” Intrinsic pathway XIIa Extrinsic Pathway XIa TF Prothrombin IXa VIIIa Fibrinogen VIIa Xa Va Thrombin Soft clot Fibrin XIIIa Hard clot Fibrin
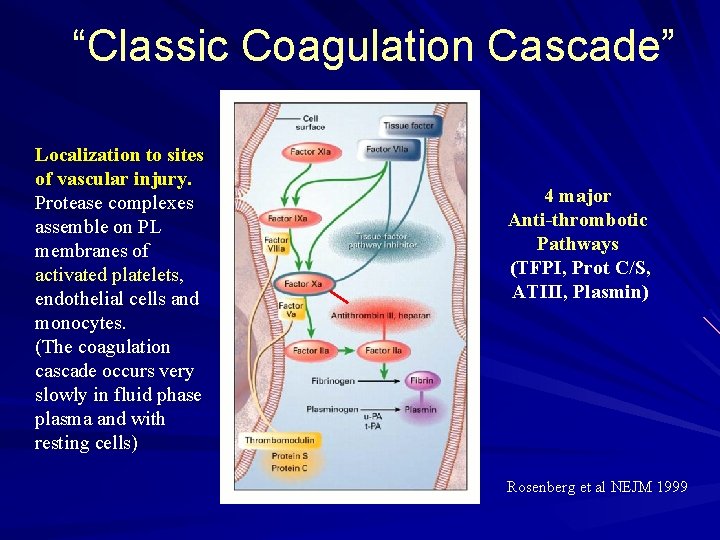
“Classic Coagulation Cascade” Localization to sites of vascular injury. Protease complexes assemble on PL membranes of activated platelets, endothelial cells and monocytes. (The coagulation cascade occurs very slowly in fluid phase plasma and with resting cells) 4 major Anti-thrombotic Pathways (TFPI, Prot C/S, ATIII, Plasmin) Rosenberg et al NEJM 1999

From “Classic” to Current View

Current View of the Coagulation System Initiation by vessel wall injury which exposes blood to cells with TF on their surface TF/FVIIa activates FX Xa + Va cleaves II small amounts of IIa (thrombin) Minute amounts of thrombin produced initially than lead to a marked increase in activation of FXI, FIX, FVIII, FV and marked generation of thrombin. Priming invloves adherence and activation of platelets. The small amounts of initial thrombin activates platelets release of FV + PL surface for protease activation Propagation – an explosive increase in thrombin generation mediated by the classic “intrinsic system” FXI, FIX Fxa/VIIIa/Va on activated platelets IIa + fibrin formation Schneider D et al, Circulation 2007

“The Great Balance” Thrombotic Complications Bleeding Complications
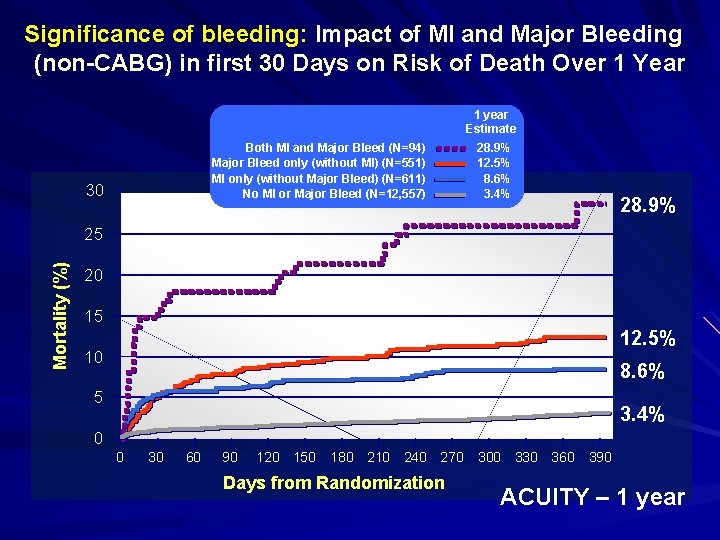
Significance of bleeding: Impact of MI and Major Bleeding (non-CABG) in first 30 Days on Risk of Death Over 1 Year 1 year Estimate Both MI and Major Bleed (N=94) Major Bleed only (without MI) (N=551) MI only (without Major Bleed) (N=611) No MI or Major Bleed (N=12, 557) 30 28. 9% 12. 5% 8. 6% 3. 4% 28. 9% Mortality (%) 25 20 15 12. 5% 10 8. 6% 5 3. 4% 0 0 30 60 90 120 150 180 210 240 270 Days from Randomization 300 330 360 390 ACUITY – 1 year

Courtesy of Dr S. Steinhubl, U. Kentucky
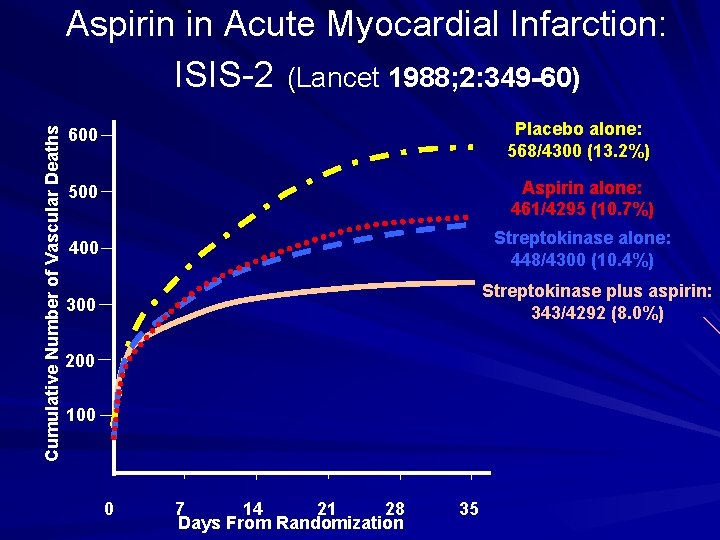
Cumulative Number of Vascular Deaths Aspirin in Acute Myocardial Infarction: ISIS-2 (Lancet 1988; 2: 349 -60) 600 Placebo alone: 568/4300 (13. 2%) 500 Aspirin alone: 461/4295 (10. 7%) 400 Streptokinase alone: 448/4300 (10. 4%) Streptokinase plus aspirin: 343/4292 (8. 0%) 300 200 100 0 7 14 21 28 Days From Randomization 35
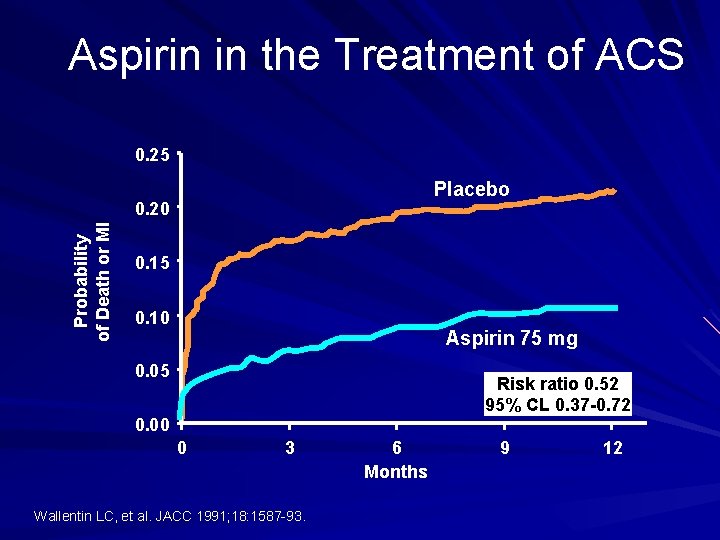
Aspirin in the Treatment of ACS 0. 25 Placebo Probability of Death or MI 0. 20 0. 15 0. 10 Aspirin 75 mg 0. 05 Risk ratio 0. 52 95% CL 0. 37 -0. 72 0. 00 0 3 Wallentin LC, et al. JACC 1991; 18: 1587 -93. 6 Months 9 12
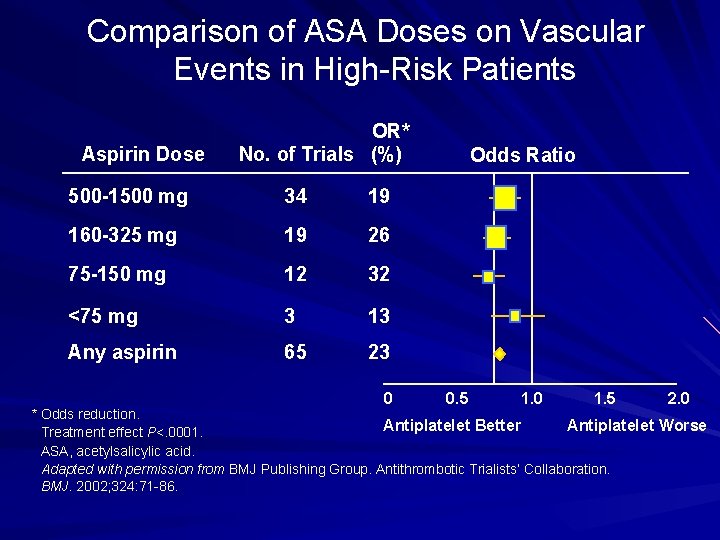
Comparison of ASA Doses on Vascular Events in High-Risk Patients Aspirin Dose OR* No. of Trials (%) 500 -1500 mg 34 19 160 -325 mg 19 26 75 -150 mg 12 32 <75 mg 3 13 Any aspirin 65 23 0 Odds Ratio 0. 5 1. 0 1. 5 2. 0 * Odds reduction. Antiplatelet Better Antiplatelet Worse Treatment effect P<. 0001. ASA, acetylsalicylic acid. Adapted with permission from BMJ Publishing Group. Antithrombotic Trialists’ Collaboration. BMJ. 2002; 324: 71 -86.
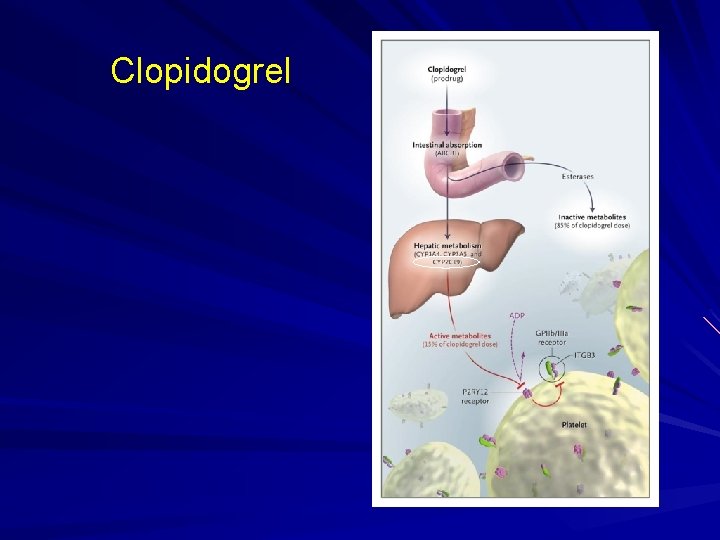
Clopidogrel
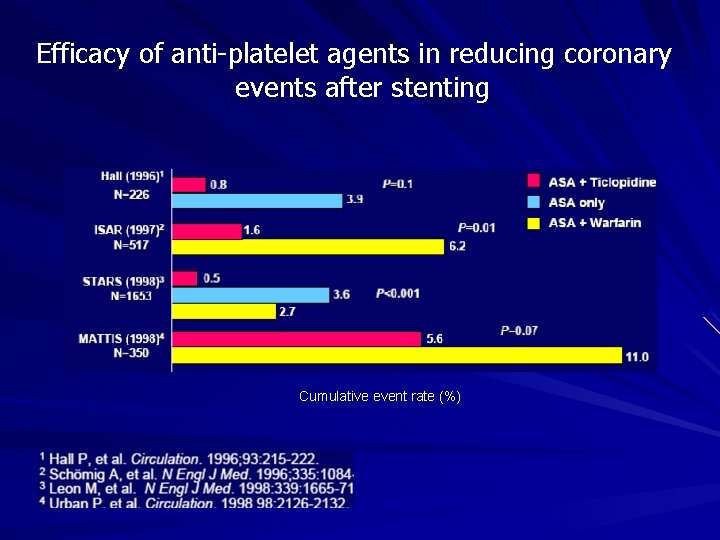
Efficacy of anti-platelet agents in reducing coronary events after stenting Cumulative event rate (%)
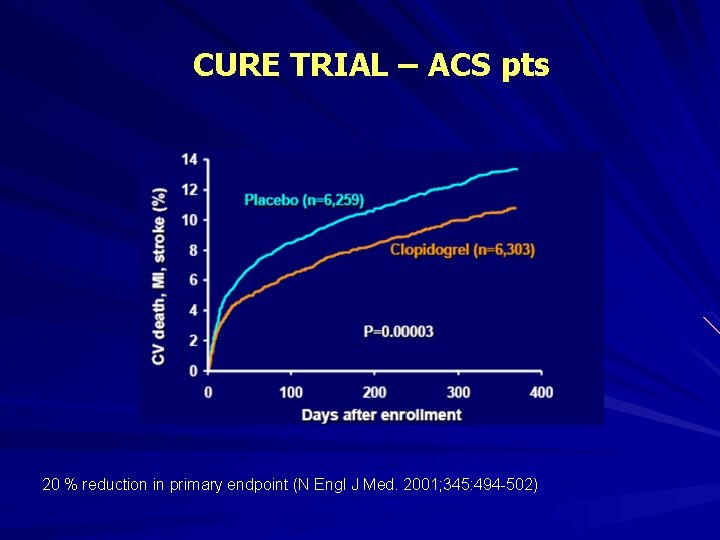
CURE TRIAL – ACS pts 20 % reduction in primary endpoint (N Engl J Med. 2001; 345: 494 -502)
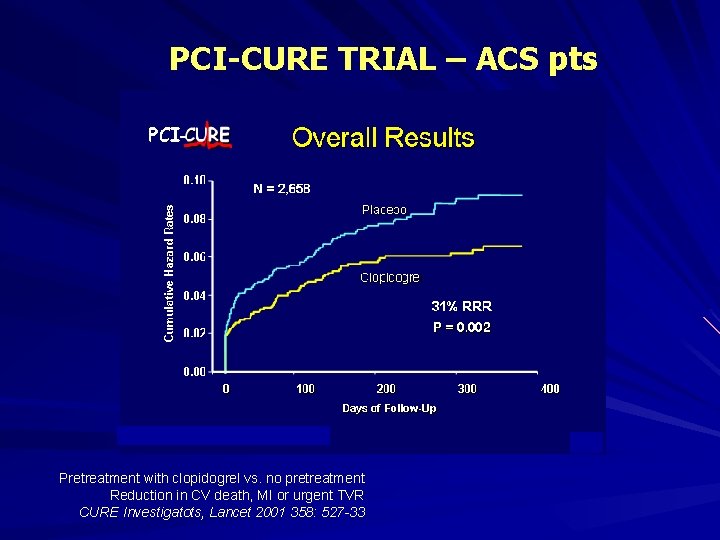
PCI-CURE TRIAL – ACS pts Pretreatment with clopidogrel vs. no pretreatment Reduction in CV death, MI or urgent TVR CURE Investigatots, Lancet 2001 358: 527 -33
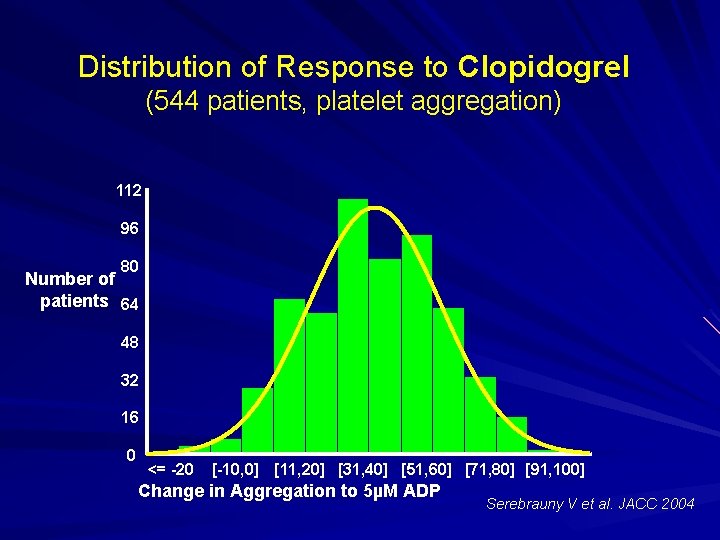
Distribution of Response to Clopidogrel (544 patients, platelet aggregation) 112 96 80 Number of patients 64 48 32 16 0 <= -20 [-10, 0] [11, 20] [31, 40] [51, 60] [71, 80] [91, 100] Change in Aggregation to 5µM ADP Serebrauny V et al. JACC 2004
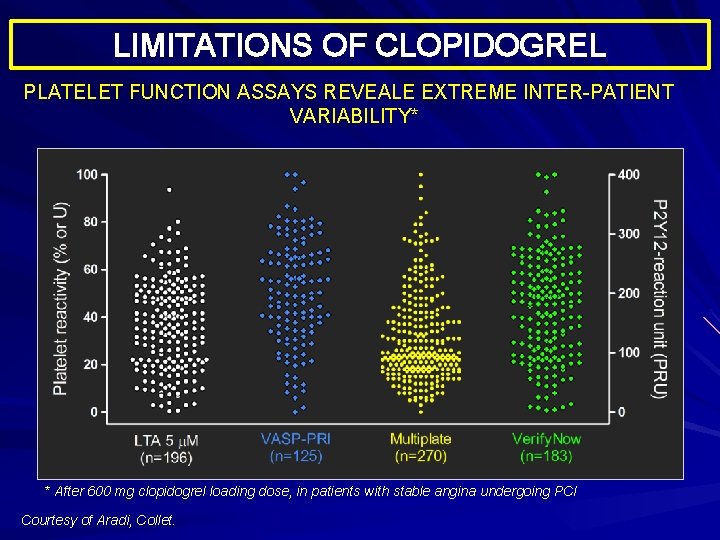
LIMITATIONS OF CLOPIDOGREL PLATELET FUNCTION ASSAYS REVEALE EXTREME INTER-PATIENT VARIABILITY* * After 600 mg clopidogrel loading dose, in patients with stable angina undergoing PCI Courtesy of Aradi, Collet.
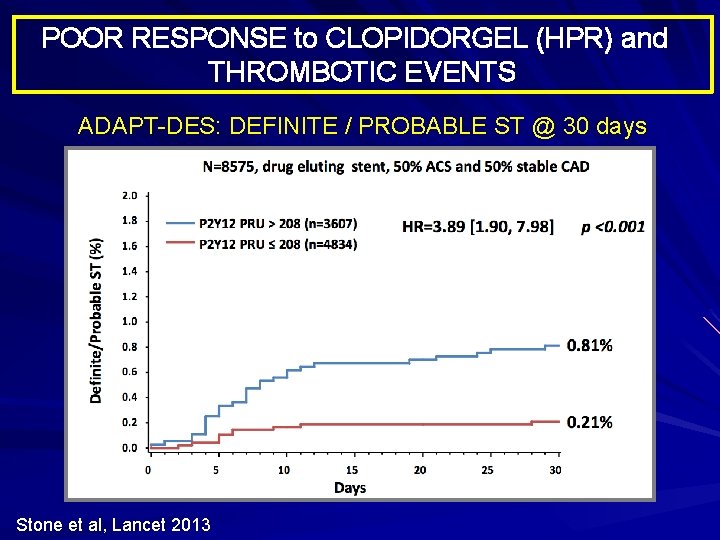
POOR RESPONSE to CLOPIDORGEL (HPR) and THROMBOTIC EVENTS ADAPT-DES: DEFINITE / PROBABLE ST @ 30 days Stone et al, Lancet 2013
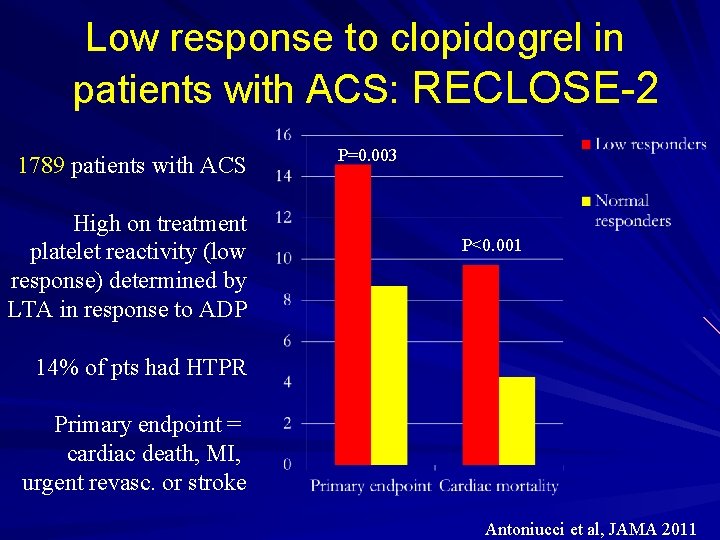
Low response to clopidogrel in patients with ACS: RECLOSE-2 1789 patients with ACS High on treatment platelet reactivity (low response) determined by LTA in response to ADP P=0. 003 P<0. 001 14% of pts had HTPR Primary endpoint = cardiac death, MI, urgent revasc. or stroke Antoniucci et al, JAMA 2011
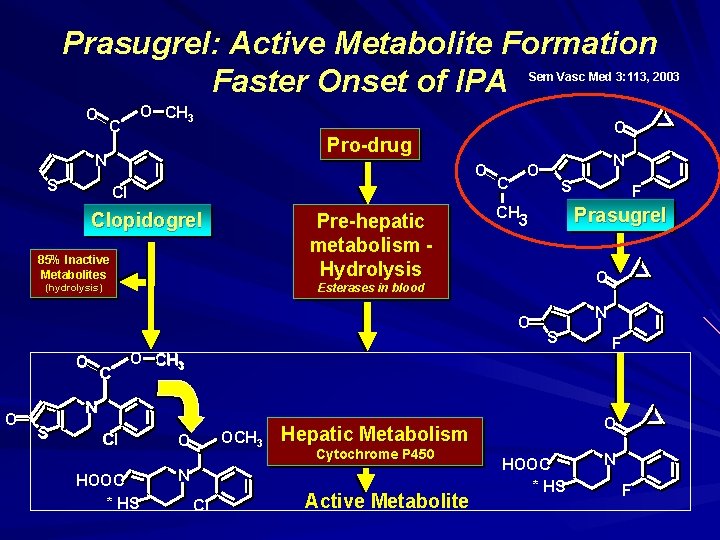
Prasugrel: Active Metabolite Formation Faster Onset of IPA Sem Vasc Med 3: 113, 2003 O O CH 3 C Pro-drug N S O O Cl Clopidogrel Pre-hepatic metabolism Hydrolysis 85% Inactive Metabolites C CH 3 O Prasugrel N S O CH 33 N S Cl HOOC * HS OCH 3 O N Cl Active Metabolite F O Hepatic Metabolism Cytochrome P 450 F O O C S Esterases in blood (hydrolysis) O N O HOOC * HS N F
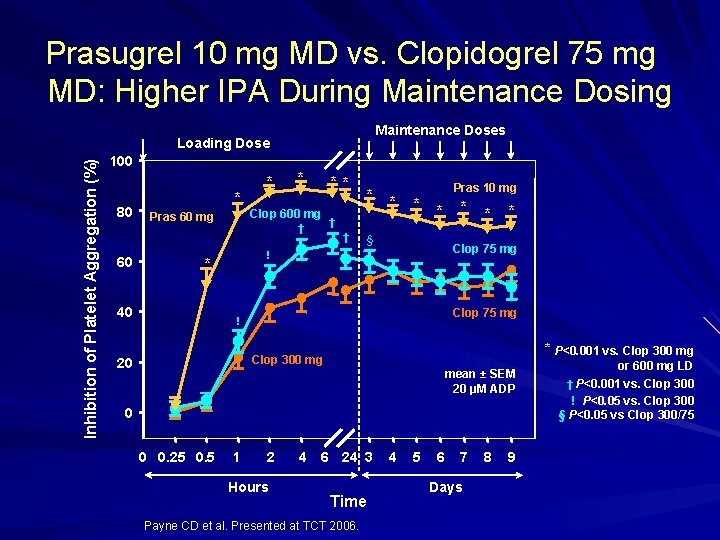
Prasugrel 10 mg MD vs. Clopidogrel 75 mg MD: Higher IPA During Maintenance Doses Inhibition of Platelet Aggregation (%) Loading Dose 100 80 60 * Pras 60 mg * Clop 600 mg † ** * * Pras 10 mg * † † * § * * * Clop 75 mg ! * 40 * Clop 75 mg ! * P<0. 001 vs. Clop 300 mg 20 mean ± SEM 20 μM ADP 0 0 0. 25 0. 5 1 2 Hours 4 6 24 3 Time Payne CD et al. Presented at TCT 2006. 4 5 6 7 Days 8 9 or 600 mg LD † P<0. 001 vs. Clop 300 ! P<0. 05 vs. Clop 300 § P<0. 05 vs Clop 300/75
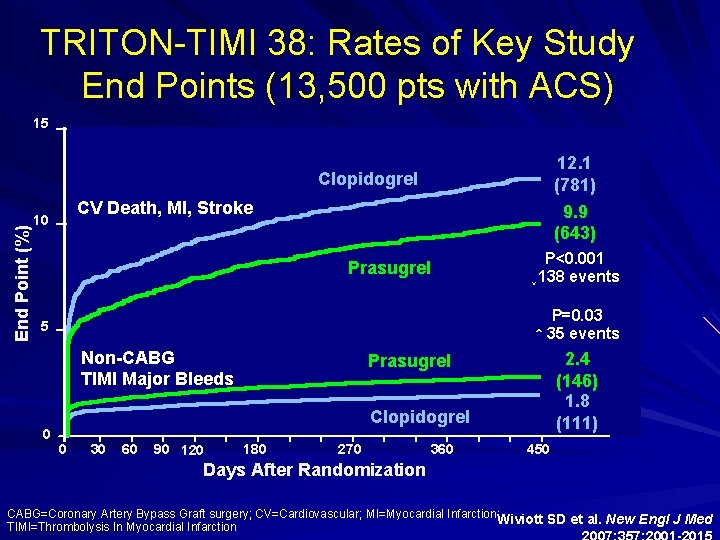
TRITON-TIMI 38: Rates of Key Study End Points (13, 500 pts with ACS) 15 12. 1 (781) 9. 9 (643) End Point (%) Clopidogrel CV Death, MI, Stroke 10 Prasugrel P=0. 03 ↑ 35 events 5 Non-CABG TIMI Major Bleeds 0 P<0. 001 ↓ 138 events 2. 4 (146) 1. 8 (111) Prasugrel Clopidogrel 0 30 60 90 120 180 270 360 450 Days After Randomization CABG=Coronary Artery Bypass Graft surgery; CV=Cardiovascular; MI=Myocardial Infarction; Wiviott SD et al. New Engl J Med TIMI=Thrombolysis In Myocardial Infarction
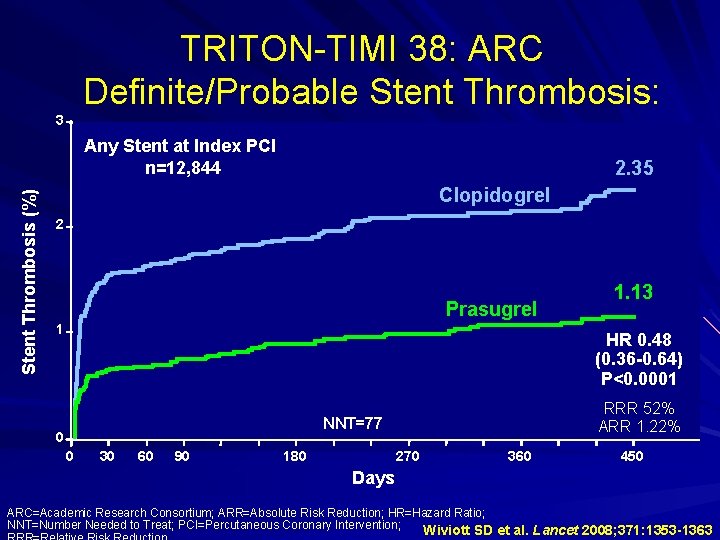
TRITON-TIMI 38: ARC Definite/Probable Stent Thrombosis: 3 Stent Thrombosis (%) Any Stent at Index PCI n=12, 844 2. 35 Clopidogrel 2 Prasugrel 1 1. 13 HR 0. 48 (0. 36 -0. 64) P<0. 0001 RRR 52% ARR 1. 22% NNT=77 0 0 30 60 90 180 270 360 450 Days ARC=Academic Research Consortium; ARR=Absolute Risk Reduction; HR=Hazard Ratio; NNT=Number Needed to Treat; PCI=Percutaneous Coronary Intervention; Wiviott SD et al. Lancet 2008; 371: 1353 -1363
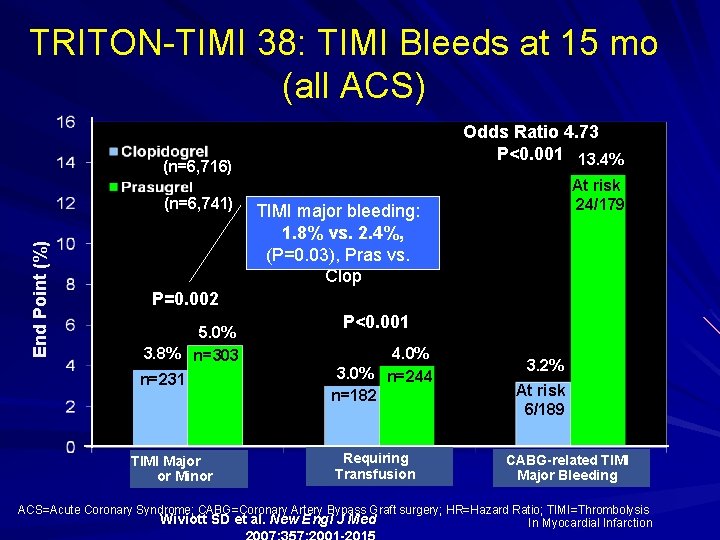
End Point (%) TRITON-TIMI 38: TIMI Bleeds at 15 mo (all ACS) (n=6, 716) Odds Ratio 4. 73 P<0. 001 13. 4% (n=6, 741) At risk 24/179 TIMI major bleeding: 1. 8% vs. 2. 4%, (P=0. 03), Pras vs. Clop P=0. 002 5. 0% 3. 8% n=303 n=231 TIMI Major or Minor P<0. 001 4. 0% 3. 0% n=244 n=182 Requiring Transfusion 3. 2% At risk 6/189 CABG-related TIMI Major Bleeding ACS=Acute Coronary Syndrome; CABG=Coronary Artery Bypass Graft surgery; HR=Hazard Ratio; TIMI=Thrombolysis Wiviott SD et al. New Engl J Med In Myocardial Infarction
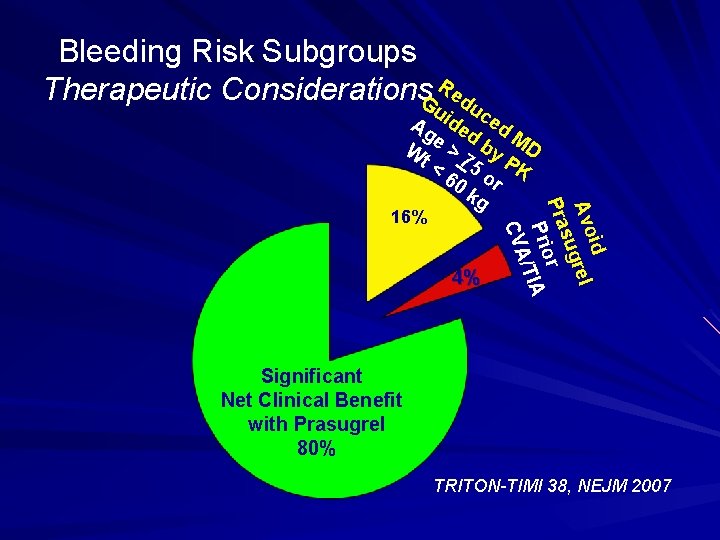
Bleeding Risk Subgroups Therapeutic Considerations. Gu. Redu 16% 4% id Avo rel sug Pra r Prio TIA / CVA Ag ide ced d M e W > by D t < 75 PK 60 or kg Significant Net Clinical Benefit with Prasugrel 80% TRITON-TIMI 38, NEJM 2007
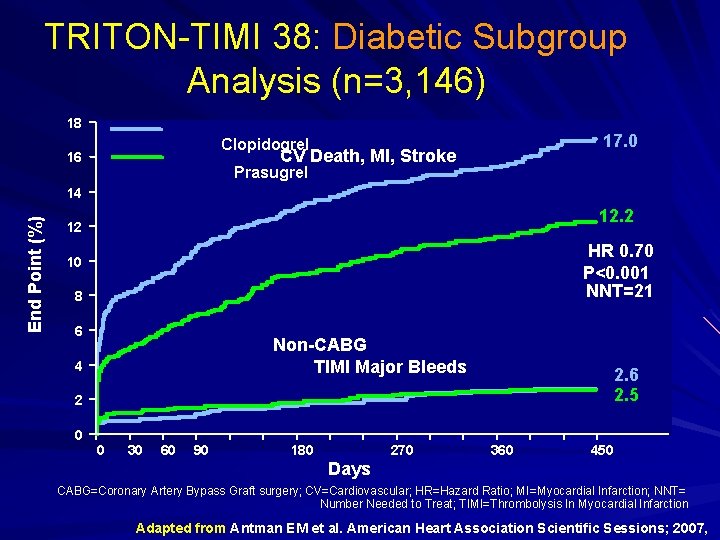
TRITON-TIMI 38: Diabetic Subgroup Analysis (n=3, 146) 18 17. 0 Clopidogrel CV Death, MI, Stroke 16 Prasugrel End Point (%) 14 12. 2 12 HR 0. 70 P<0. 001 NNT=21 10 8 6 Non-CABG TIMI Major Bleeds 4 2. 6 2. 5 2 0 0 30 60 90 180 270 360 450 Days CABG=Coronary Artery Bypass Graft surgery; CV=Cardiovascular; HR=Hazard Ratio; MI=Myocardial Infarction; NNT= Number Needed to Treat; TIMI=Thrombolysis In Myocardial Infarction Adapted from Antman EM et al. American Heart Association Scientific Sessions; 2007,
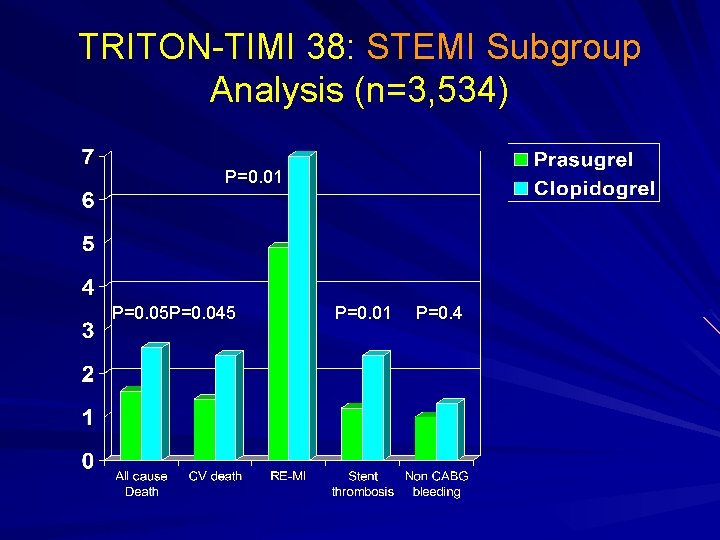
TRITON-TIMI 38: STEMI Subgroup Analysis (n=3, 534) P=0. 01 P=0. 05 P=0. 045 P=0. 01 P=0. 4
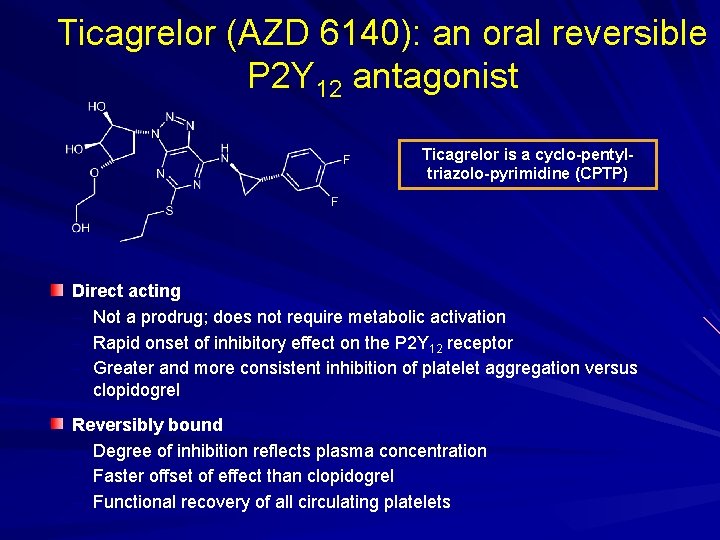
Ticagrelor (AZD 6140): an oral reversible P 2 Y 12 antagonist Ticagrelor is a cyclo-pentyltriazolo-pyrimidine (CPTP) Direct acting – Not a prodrug; does not require metabolic activation – Rapid onset of inhibitory effect on the P 2 Y 12 receptor – Greater and more consistent inhibition of platelet aggregation versus clopidogrel Reversibly bound – Degree of inhibition reflects plasma concentration – Faster offset of effect than clopidogrel – Functional recovery of all circulating platelets
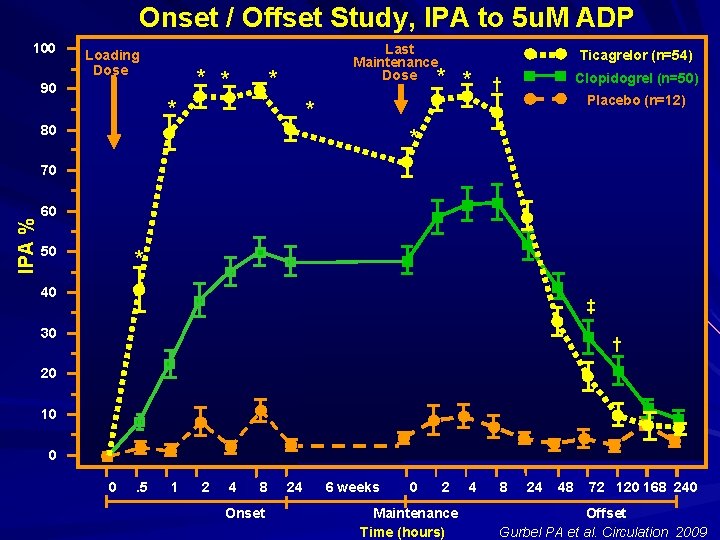
Onset / Offset Study, IPA to 5 u. M ADP 100 Loading Dose * * 90 Last Maintenance Dose * * * Ticagrelor (n=54) * Clopidogrel (n=50) † Placebo (n=12) * 80 * IPA % 70 60 50 * 40 ‡ 30 † 20 10 0 0 . 5 1 2 4 8 Onset 24 6 weeks 0 2 Maintenance Time (hours) 4 8 24 48 72 120 168 240 Offset Gurbel PA et al. Circulation 2009
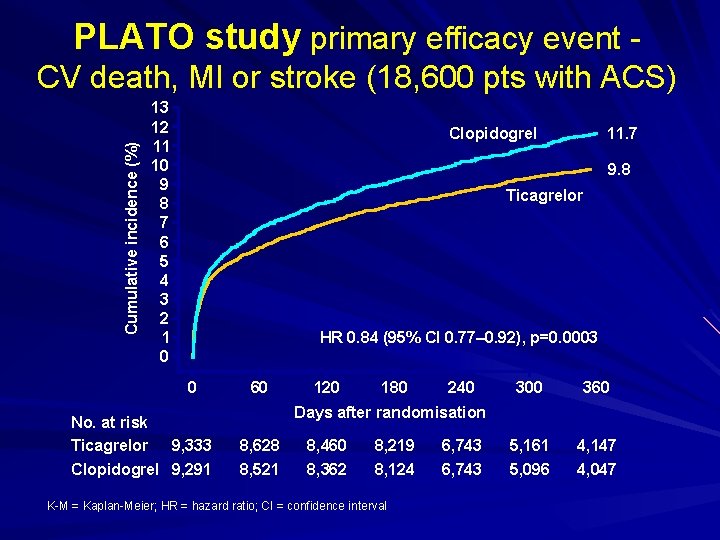
PLATO study primary efficacy event Cumulative incidence (%) CV death, MI or stroke (18, 600 pts with ACS) 13 12 11 10 9 8 7 6 5 4 3 2 1 0 11. 7 Clopidogrel 9. 8 Ticagrelor HR 0. 84 (95% CI 0. 77– 0. 92), p=0. 0003 0 No. at risk Ticagrelor 9, 333 Clopidogrel 9, 291 60 120 180 240 300 360 5, 161 5, 096 4, 147 4, 047 Days after randomisation 8, 628 8, 521 8, 460 8, 362 8, 219 8, 124 K-M = Kaplan-Meier; HR = hazard ratio; CI = confidence interval 6, 743
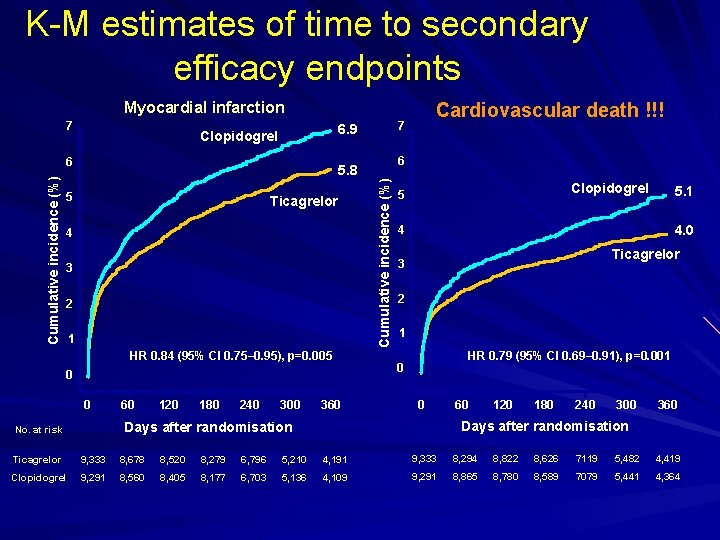
K-M estimates of time to secondary efficacy endpoints Myocardial infarction 7 6. 9 Clopidogrel 6 5. 8 5 Ticagrelor 4 3 2 1 HR 0. 84 (95% CI 0. 75– 0. 95), p=0. 005 0 0 60 120 180 240 300 360 Cumulative incidence (%) 6 Cardiovascular death !!! 7 Clopidogrel 5 4. 0 4 Ticagrelor 3 2 1 HR 0. 79 (95% CI 0. 69– 0. 91), p=0. 001 0 0 60 120 180 240 300 360 Days after randomisation No. at risk 5. 1 Ticagrelor 9, 333 8, 678 8, 520 8, 279 6, 796 5, 210 4, 191 9, 333 8, 294 8, 822 8, 626 7119 5, 482 4, 419 Clopidogrel 9, 291 8, 560 8, 405 8, 177 6, 703 5, 136 4, 109 9, 291 8, 865 8, 780 8, 589 7079 5, 441 4, 364
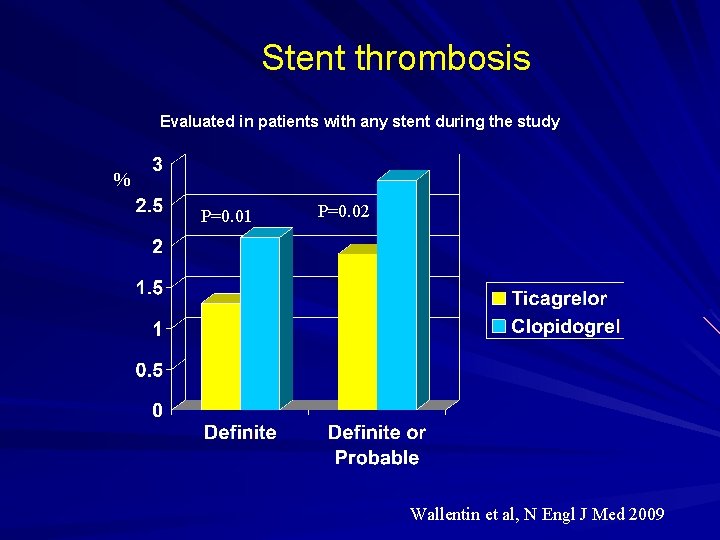
Stent thrombosis Evaluated in patients with any stent during the study % P=0. 01 P=0. 02 Wallentin et al, N Engl J Med 2009
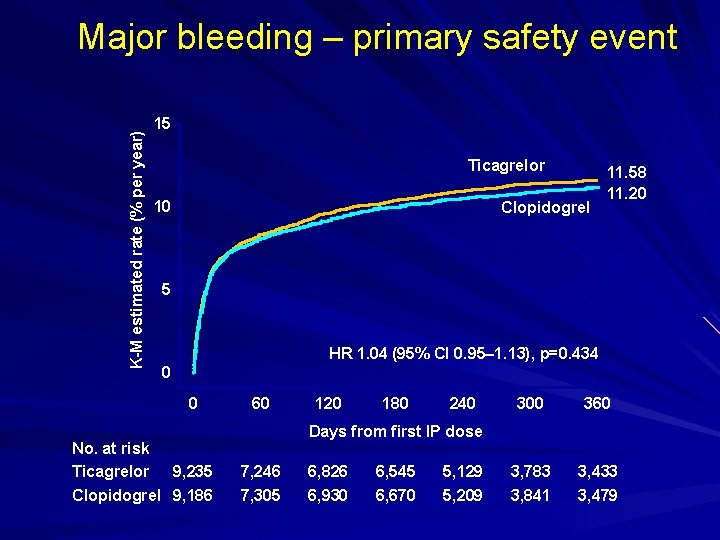
K-M estimated rate (% per year) Major bleeding – primary safety event 15 Ticagrelor 10 Clopidogrel 11. 58 11. 20 5 HR 1. 04 (95% CI 0. 95– 1. 13), p=0. 434 0 0 No. at risk Ticagrelor 9, 235 Clopidogrel 9, 186 60 120 180 240 300 360 3, 783 3, 841 3, 433 3, 479 Days from first IP dose 7, 246 7, 305 6, 826 6, 930 6, 545 6, 670 5, 129 5, 209
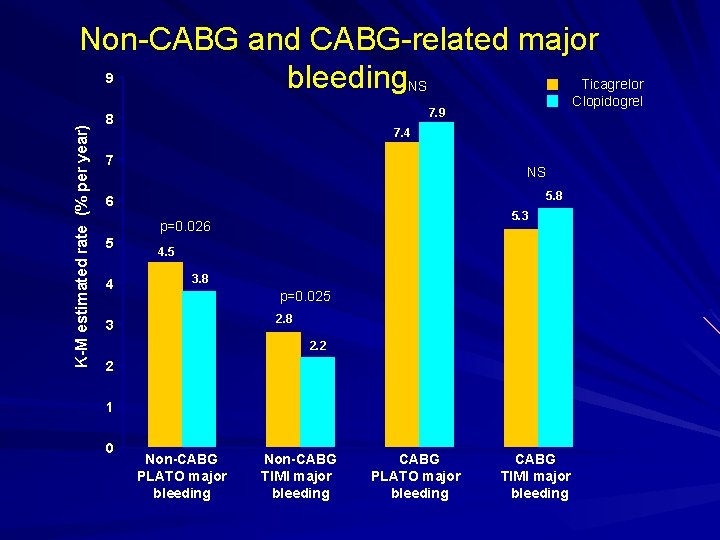
K-M estimated rate (% per year) Non-CABG and CABG-related major 9 bleeding. NS Ticagrelor 8 7. 4 7 NS 5. 8 6 5. 3 p=0. 026 5 4 Clopidogrel 7. 9 4. 5 3. 8 p=0. 025 2. 8 3 2. 2 2 1 0 Non-CABG PLATO major bleeding Non-CABG TIMI major bleeding CABG PLATO major bleeding CABG TIMI major bleeding

Dyspnoea with Ticagrelor All patients Ticagrelor Clopidogrel (n=9, 235) (n=9, 186) p value* Dyspnoea, % Any 13. 8 7. 8 <0. 001 Requiring discontinuation of study Rx 0. 9 0. 1 <0. 001 “Ticagrelor-related dyspnoea is usually mild or moderate in intensity, selflimiting and does not appear to be associated with differences in any efficacy or safety outcomes with ticagrelor compared with clopidogrel therapy in ACS pts. ” Storey et al, Eur Heart J 2011
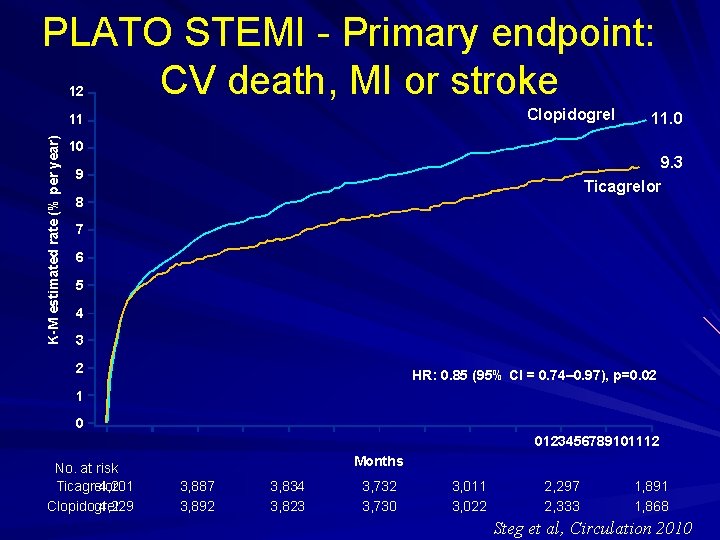
PLATO STEMI - Primary endpoint: CV death, MI or stroke 12 Clopidogrel K-M estimated rate (% per year) 11 11. 0 10 9. 3 9 Ticagrelor 8 7 6 5 4 3 2 HR: 0. 85 (95% CI = 0. 74– 0. 97), p=0. 02 1 0 0123456789101112 No. at risk Ticagrelor 4, 201 Clopidogrel 4, 229 Months 3, 887 3, 892 3, 834 3, 823 3, 732 3, 730 3, 011 3, 022 2, 297 2, 333 1, 891 1, 868 Steg et al, Circulation 2010
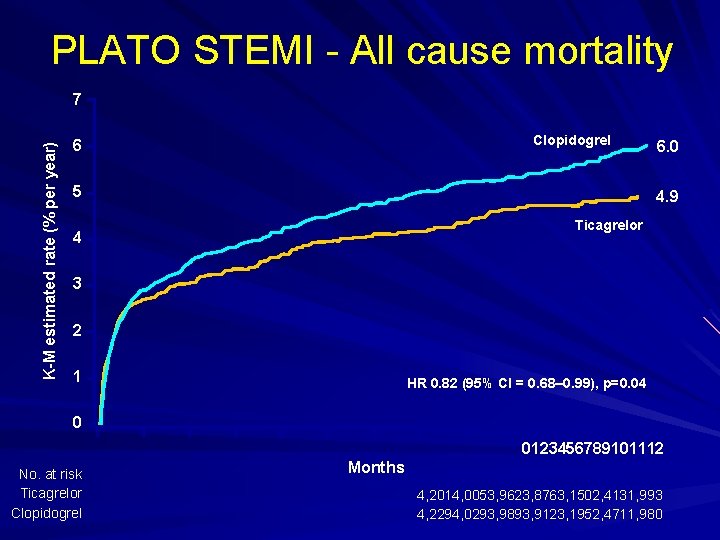
PLATO STEMI - All cause mortality K-M estimated rate (% per year) 7 Clopidogrel 6 5 6. 0 4. 9 Ticagrelor 4 3 2 1 HR 0. 82 (95% CI = 0. 68– 0. 99), p=0. 04 0 0123456789101112 No. at risk Ticagrelor Clopidogrel Months 4, 2014, 0053, 9623, 8763, 1502, 4131, 993 4, 2294, 0293, 9893, 9123, 1952, 4711, 980
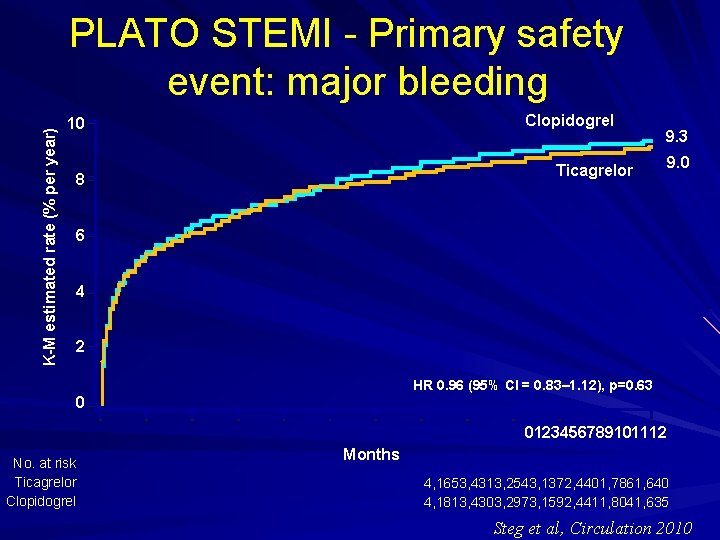
K-M estimated rate (% per year) PLATO STEMI - Primary safety event: major bleeding Clopidogrel 10 9. 3 Ticagrelor 8 9. 0 6 4 2 HR 0. 96 (95% CI = 0. 83– 1. 12), p=0. 63 0 0123456789101112 No. at risk Ticagrelor Clopidogrel Months 4, 1653, 4313, 2543, 1372, 4401, 7861, 640 4, 1813, 4303, 2973, 1592, 4411, 8041, 635 Steg et al, Circulation 2010
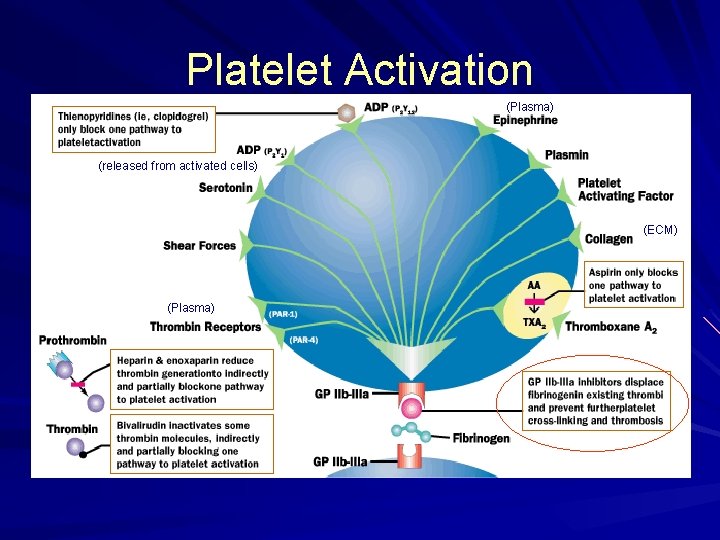
Platelet Activation (Plasma) (released from activated cells) (ECM) (Plasma)

GP IIb/IIIA Inhibitors Abciximab (Reo. Pro ) – the first inhibitor developed and approved for clinical use. Chimeric monoclonal antibody – 7 E 3, the murine constant region was replaced by its human counterpart Eptifibatide (Integrilin ) – synthetic cyclic hepta -peptide derived from a sequence found in the venom of the southeastern pygmy rattlesnake Tirofiban (Aggrastat ) – synthetic small molecule with structure similar to that of the RGD sequence of the snake venom echistatin

Glycoprotein IIb/IIIa Receptor Antagonists Abciximab Pharma Fab portion of chimeric monoclonal antibody Plasma ½ life 30 minutes Specificity Not specific Dose 0. 25 mcg/kg bolus followed by 0. 125 mcg/kg/min drip (max 10 mcg/min) for 12 -24 hours Tirofiban Eptifibatide Synthetic non-peptide Cyclic heptapeptide 1. 8 hours 2. 5 hours Highly specific 0. 4 mcg/kg/min for 30 minutes followed by 0. 1 mcg/kg/min drip for 48 -96 hours 180 mcg/kg bolus (x 2) followed by 2. 0 mcg/kg/min drip for 18 -24 hours
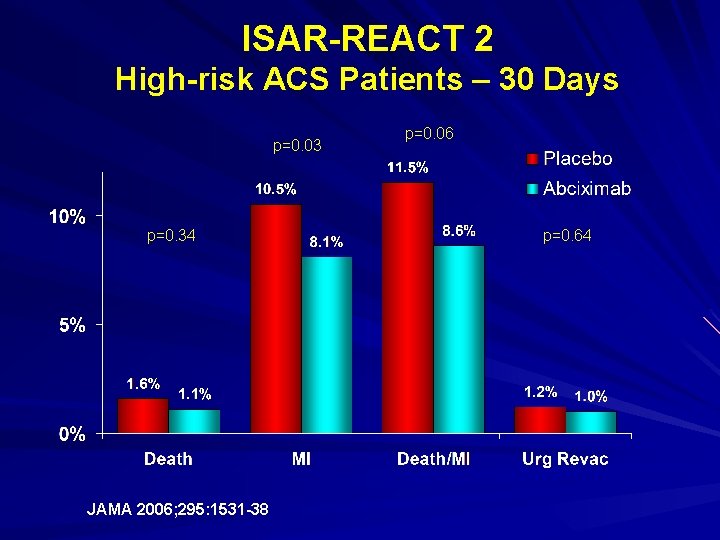
ISAR-REACT 2 High-risk ACS Patients – 30 Days p=0. 03 p=0. 34 JAMA 2006; 295: 1531 -38 p=0. 06 p=0. 64
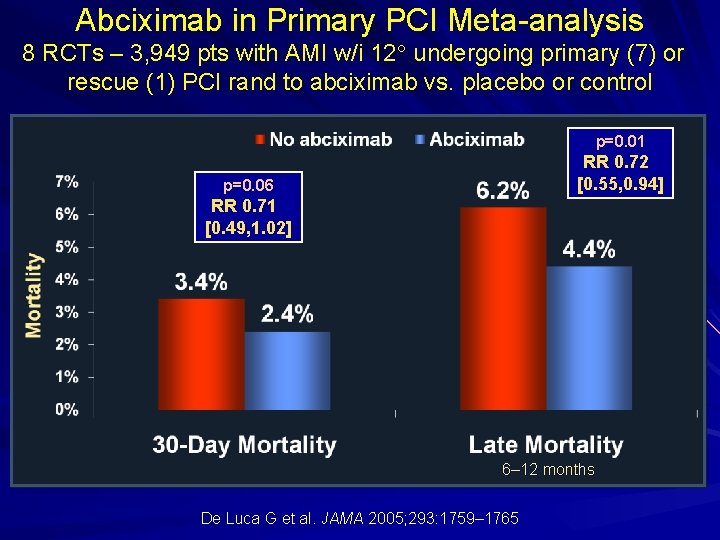
Abciximab in Primary PCI Meta-analysis 8 RCTs – 3, 949 pts with AMI w/i 12 undergoing primary (7) or rescue (1) PCI rand to abciximab vs. placebo or control p=0. 01 RR 0. 72 [0. 55, 0. 94] p=0. 06 RR 0. 71 [0. 49, 1. 02] 6– 12 months De Luca G et al. JAMA 2005; 293: 1759– 1765
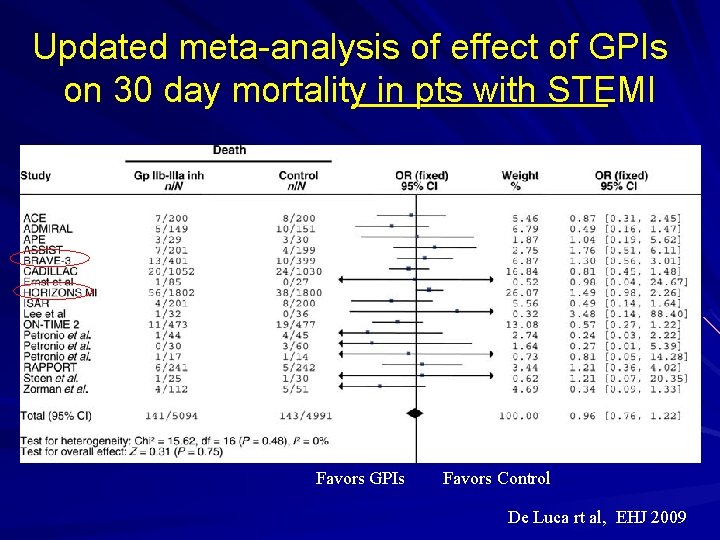
Updated meta-analysis of effect of GPIs on 30 day mortality in pts with STEMI Favors GPIs Favors Control De Luca rt al, EHJ 2009
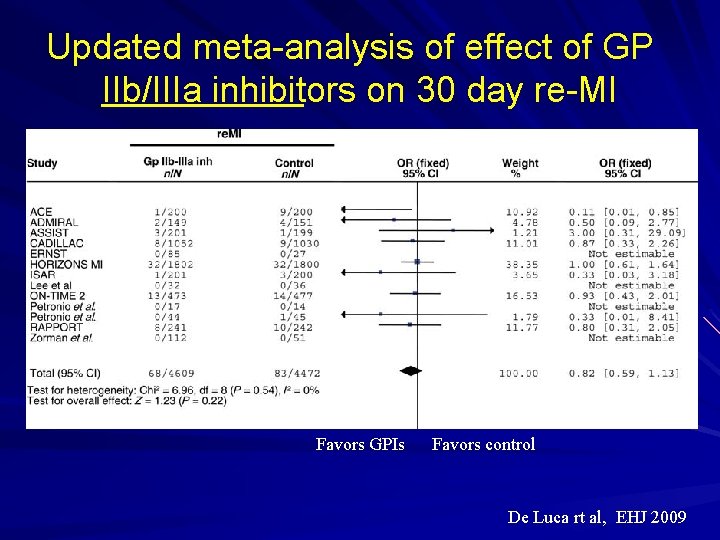
Updated meta-analysis of effect of GP IIb/IIIa inhibitors on 30 day re-MI Favors GPIs Favors control De Luca rt al, EHJ 2009
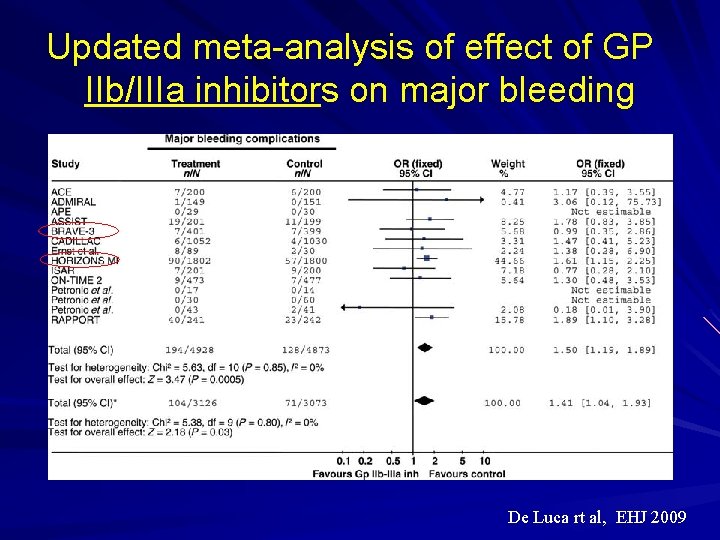
Updated meta-analysis of effect of GP IIb/IIIa inhibitors on major bleeding De Luca rt al, EHJ 2009
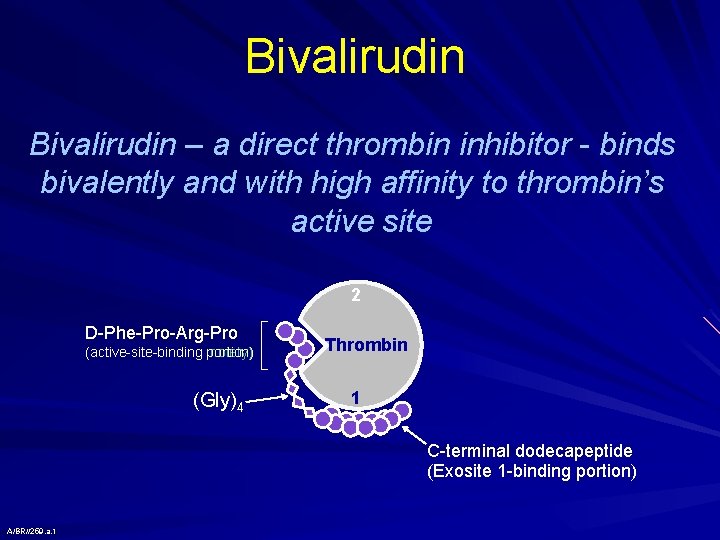
Bivalirudin – a direct thrombin inhibitor - binds bivalently and with high affinity to thrombin’s active site 2 D-Phe-Pro-Arg-Pro (active-site-binding portion) moiety) (Gly)4 Thrombin 1 C-terminal dodecapeptide (Exosite 1 -binding portion) A/BR//259. a. 1
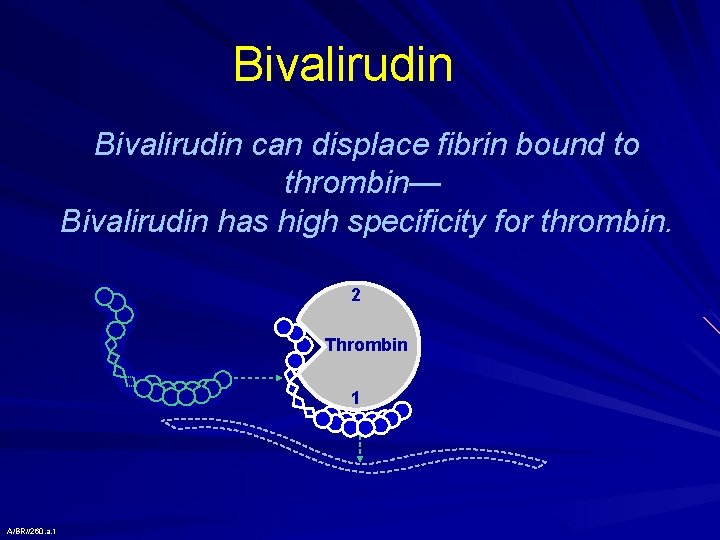
Bivalirudin can displace fibrin bound to thrombin— Bivalirudin has high specificity for thrombin. 2 Thrombin 1 A/BR//260. a. 1

Bivalirudin is cleared from plasma by combination of renal mecahnisms and proteolytic cleavage Plasma half life = 25 minutes (norm renal funct) Mod. renal impairment, half life = 34 minutes (dose reduced) Almost immediate prolongation of ACT. a. PTT Coagulation times return to normal after about 1 hour following drug d/c No HIT !!
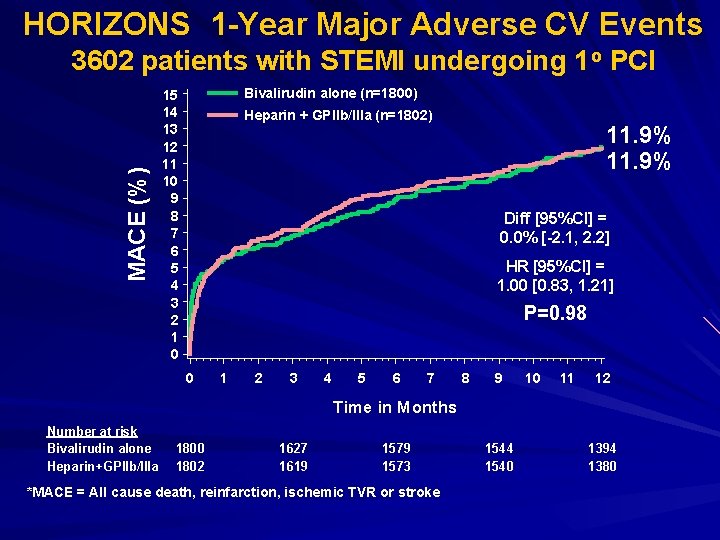
HORIZONS 1 -Year Major Adverse CV Events MACE (%) 3602 patients with STEMI undergoing 1 o PCI Bivalirudin alone (n=1800) 15 14 13 12 11 10 9 8 7 6 5 4 3 2 1 0 Heparin + GPIIb/IIIa (n=1802) 11. 9% Diff [95%CI] = 0. 0% [-2. 1, 2. 2] HR [95%CI] = 1. 00 [0. 83, 1. 21] P=0. 98 0 1 2 3 4 5 6 7 8 9 10 11 12 Time in Months Number at risk Bivalirudin alone Heparin+GPIIb/IIIa 1800 1802 1627 1619 1573 *MACE = All cause death, reinfarction, ischemic TVR or stroke 1544 1540 1394 1380
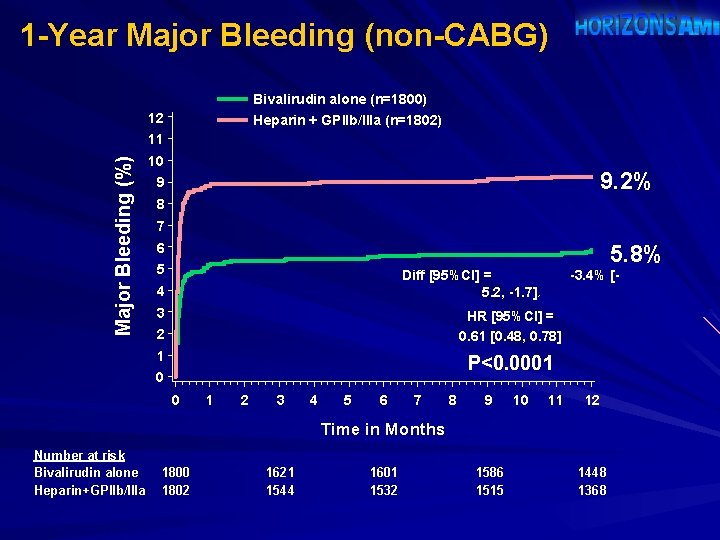
1 -Year Major Bleeding (non-CABG) Bivalirudin alone (n=1800) 12 Heparin + GPIIb/IIIa (n=1802) Major Bleeding (%) 11 10 9. 2% 9 8 7 6 5 5. 8% Diff [95%CI] = 5. 2, -1. 7] 4 -3. 4% [- 2 3 HR [95%CI] = 0. 61 [0. 48, 0. 78] 2 1 P<0. 0001 0 0 1 2 3 4 5 6 7 8 9 10 11 12 Time in Months Number at risk Bivalirudin alone Heparin+GPIIb/IIIa 1800 1802 1621 1544 1601 1532 1586 1515 1448 1368
![1 -Year Mortality: Cardiac and Non Cardiac Bivalirudin alone (n=1800) 5 HR [95%CI] = 1 -Year Mortality: Cardiac and Non Cardiac Bivalirudin alone (n=1800) 5 HR [95%CI] =](http://slidetodoc.com/presentation_image_h2/7fbe2cf6f95888e1b23b59bff0e4d8dd/image-58.jpg)
1 -Year Mortality: Cardiac and Non Cardiac Bivalirudin alone (n=1800) 5 HR [95%CI] = 0. 57 [0. 38, 0. 84] Mortality (%) Heparin + GPIIb/IIIa (n=1802) P=0. 005 4 3 3. 8% Δ = 1. 7% Cardiac 2. 9% 2. 1% 2 1. 8% Δ = 1. 1% P=0. 03 1 Non Cardiac 1. 3% 1. 1% 0 0 1 2 3 4 5 6 7 8 9 10 11 12 Time in Months Number at risk Bivalirudin alone Heparin+GPIIb/IIIa 1800 1802 1705 1678 1684 1663 1669 1646 1520 1486

NCDR PCI registry including >1, 500, 000 pts undergoing PCI in 955 US hospitals 23% of the patients treated with bivalirudin 50% elective, 36% urgent, 14% emergent PCI Bivalirudin associated with OR 0. 67 (0. 63 -0. 70) for bleeding events !! (Bleeding defined as requiring blood transfusion, prolonged hospital stay, or ↓ 3 g/d. L in Hg)

THANK YOU
- Slides: 60