3 Examples of Biological Polymers 1 Protein polypeptides
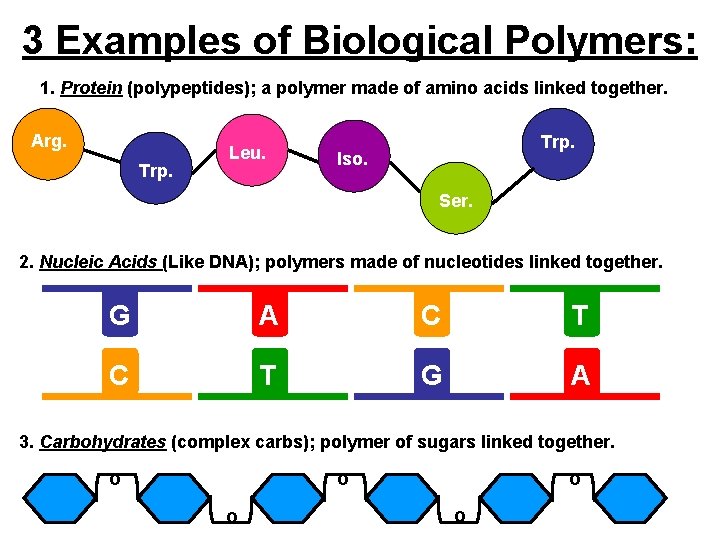
3 Examples of Biological Polymers: 1. Protein (polypeptides); a polymer made of amino acids linked together. Arg. Trp. Leu. Trp. Iso. Ser. 2. Nucleic Acids (Like DNA); polymers made of nucleotides linked together. G A C T G A 3. Carbohydrates (complex carbs); polymer of sugars linked together. o o o
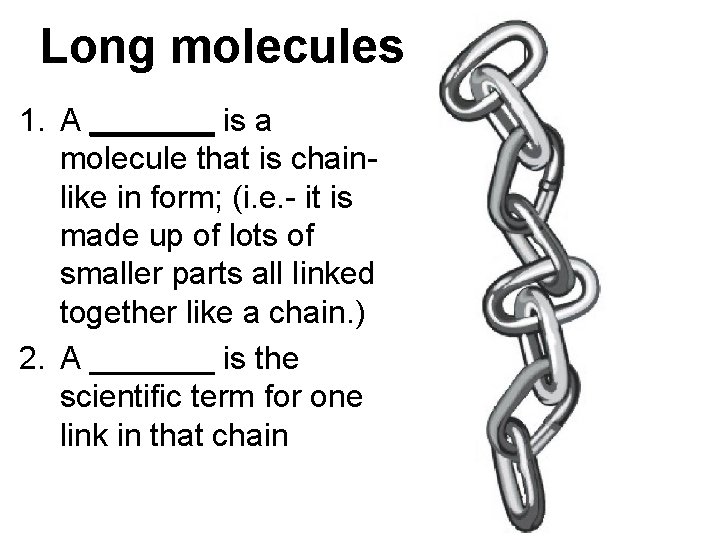
Long molecules 1. A _______ is a molecule that is chainlike in form; (i. e. - it is made up of lots of smaller parts all linked together like a chain. ) 2. A _______ is the scientific term for one link in that chain

Answers: 1. A polymer is a molecule that is chainlike in form; (i. e. - it is made up of lots of smaller parts all linked together like a chain. ) 2. A monomer is the scientific term for one link in that chain
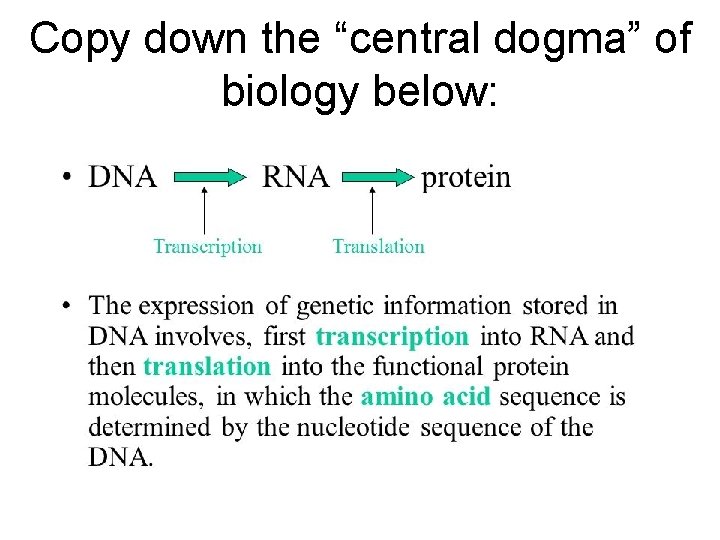
Copy down the “central dogma” of biology below:
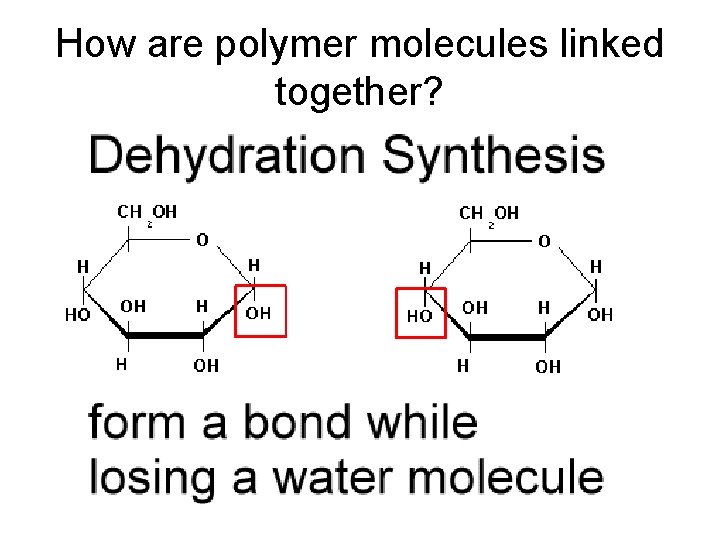
How are polymer molecules linked together?
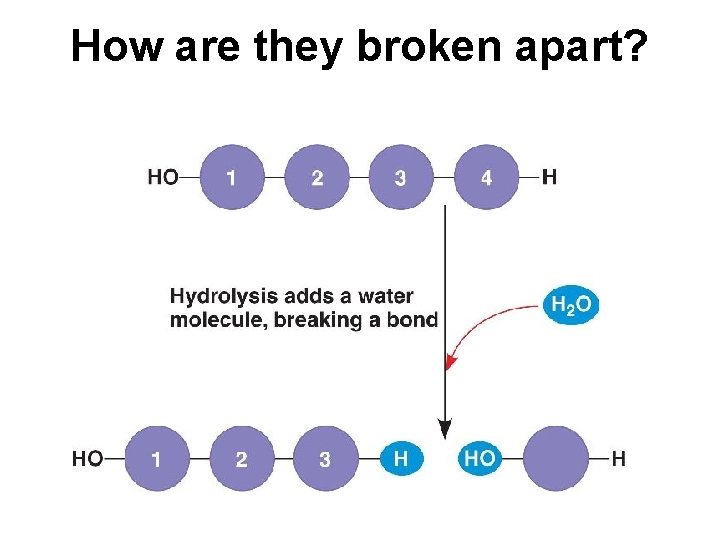
How are they broken apart?

BELLRINGER: Copy & answer the following: • Carbohydrates are compounds that contain the elements of Carbon, Hydrogen, and Oxygen in 1: 2: 1 ratios. Give an example by writing out the chemical formula for glucose.
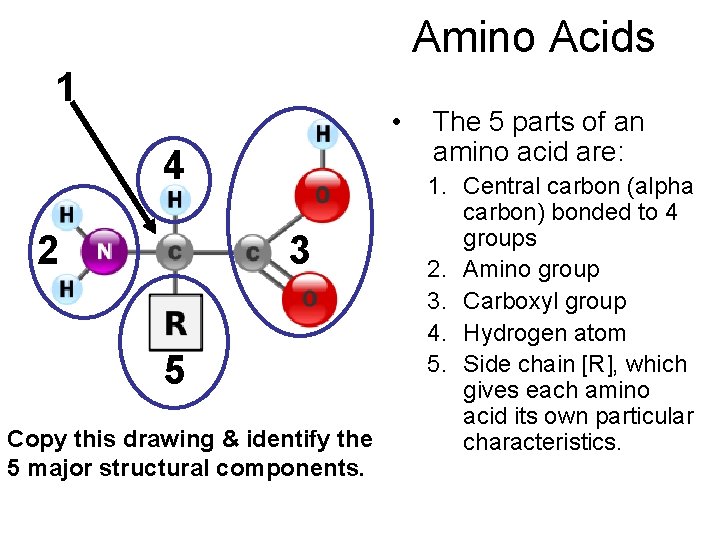
Amino Acids 1 • 4 2 3 5 Copy this drawing & identify the 5 major structural components. The 5 parts of an amino acid are: 1. Central carbon (alpha carbon) bonded to 4 groups 2. Amino group 3. Carboxyl group 4. Hydrogen atom 5. Side chain [R], which gives each amino acid its own particular characteristics.
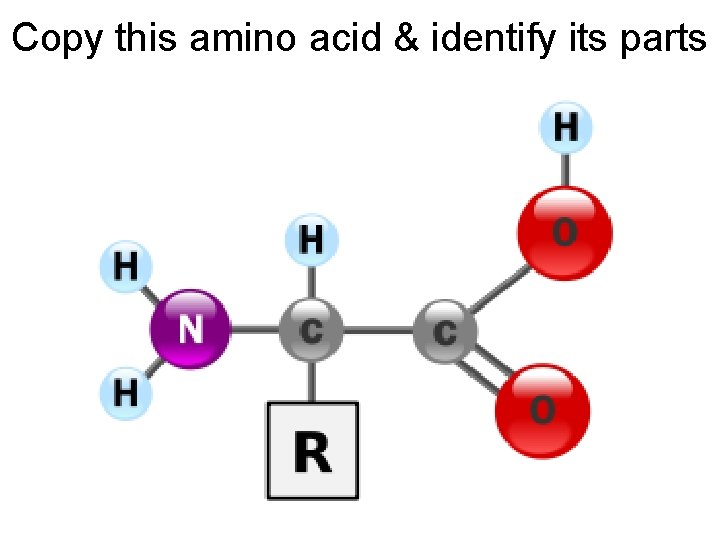
Copy this amino acid & identify its parts
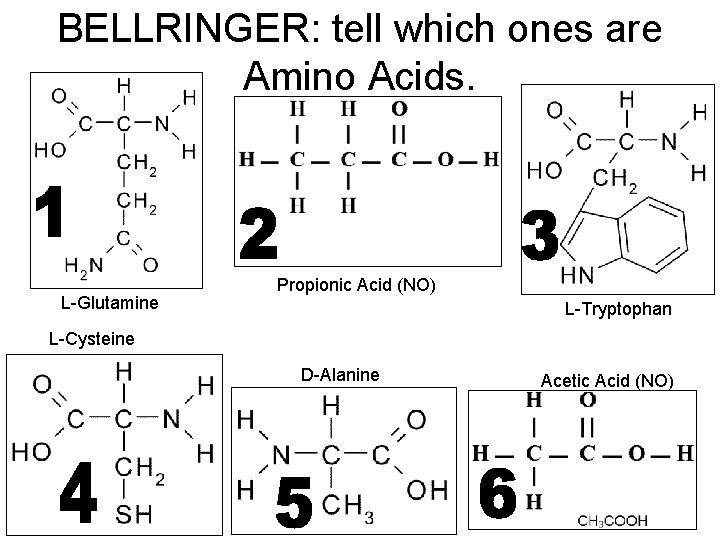
BELLRINGER: tell which ones are Amino Acids. L-Glutamine Propionic Acid (NO) L-Tryptophan L-Cysteine D-Alanine Acetic Acid (NO)
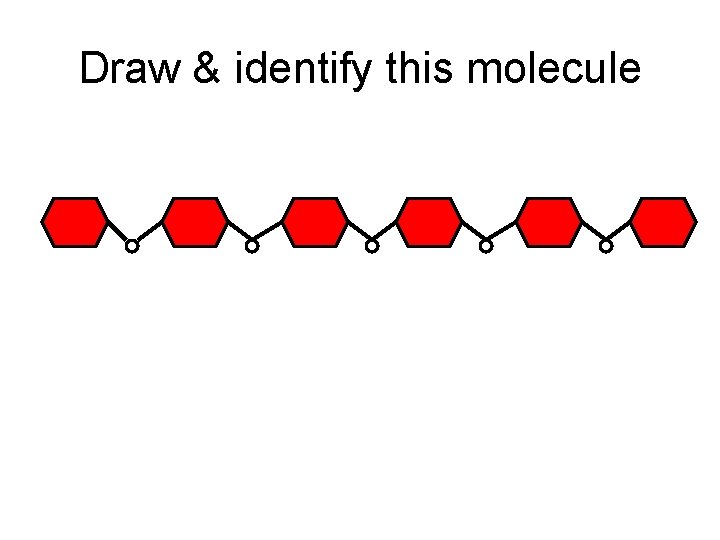
Draw & identify this molecule
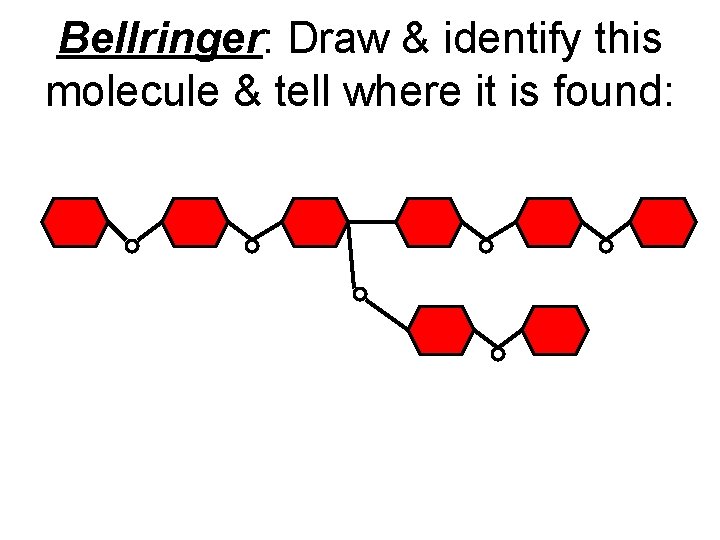
Bellringer: Draw & identify this molecule & tell where it is found:
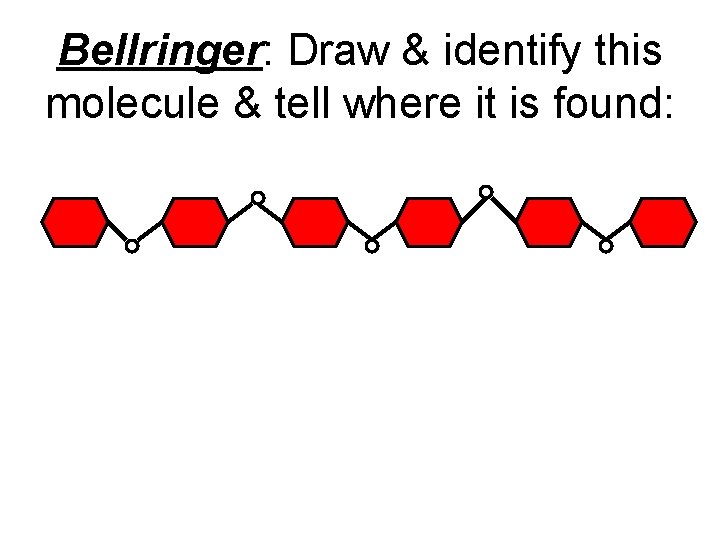
Bellringer: Draw & identify this molecule & tell where it is found:
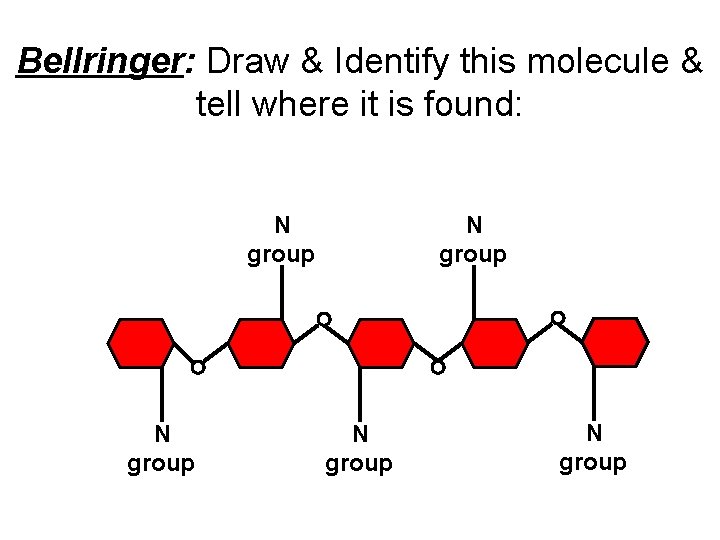
Bellringer: Draw & Identify this molecule & tell where it is found: N group N group
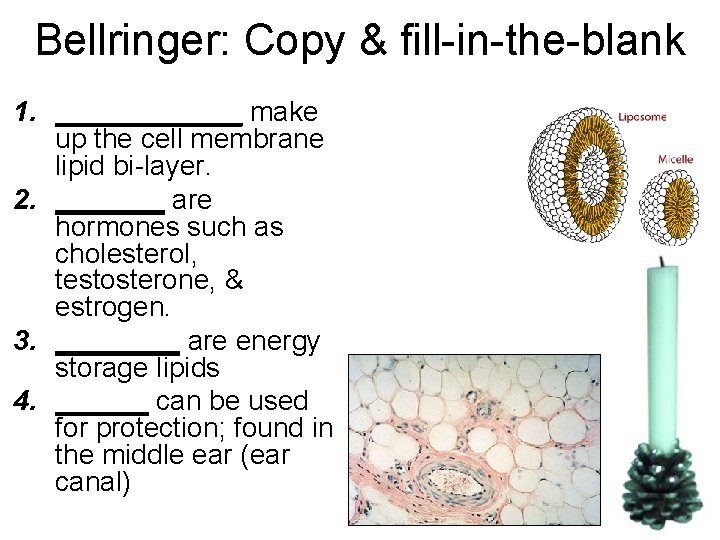
Bellringer: Copy & fill-in-the-blank 1. ______ make up the cell membrane lipid bi-layer. 2. _______ are hormones such as cholesterol, testosterone, & estrogen. 3. ____ are energy storage lipids 4. ______ can be used for protection; found in the middle ear (ear canal)
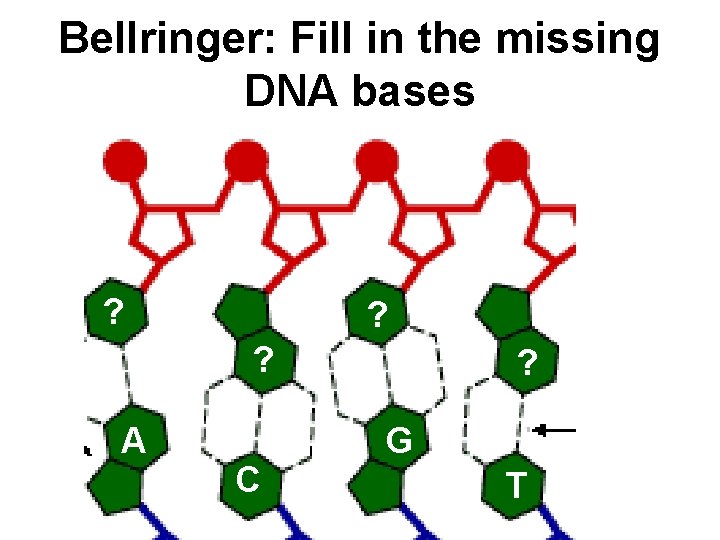
Bellringer: Fill in the missing DNA bases ? ? ? A C ? G T

Bellringer: Copy & Answer the following questions: 1. At room temperature are saturated fats solid or liquid? 2. ? At room temperature are unsaturated fats solid or liquid? 3. Why are trans fats so bad for you? C

Intro to Biochemistry: The. Chemistry of living things

Organic Chemistry The element carbon is a part of almost all biological molecules. -Carbon containing compounds are often called “organic” compounds. A trans-fatty acid. The green spheres in the middle represent carbon atoms.

Carbon: The backbone of life • Living organisms are made up mostly of molecules based on carbon. • Thus the term “carbonbased life forms”.

Carbon: The backbone of life • Americans, on average, consume 140 lbs of sugar per year, much of it in the form of fructose (High Fructose Corn Syrup) Fructose, a hexose sugar

Carbon: The backbone of life • Cellulose, found in plant cell walls, is the most abundant organic compound on Earth
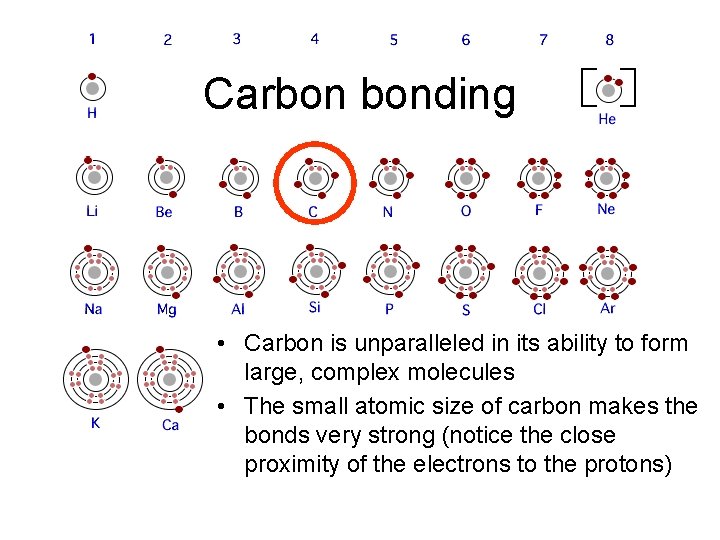
Carbon bonding • Carbon is unparalleled in its ability to form large, complex molecules • The small atomic size of carbon makes the bonds very strong (notice the close proximity of the electrons to the protons)

Macromolecules • Macromolecules are the large molecules used by living things. • There are 4 major kinds of biological macromolecules 1. 2. 3. 4. Proteins Carbohydrates Lipids Nucleic Acids

Proteins
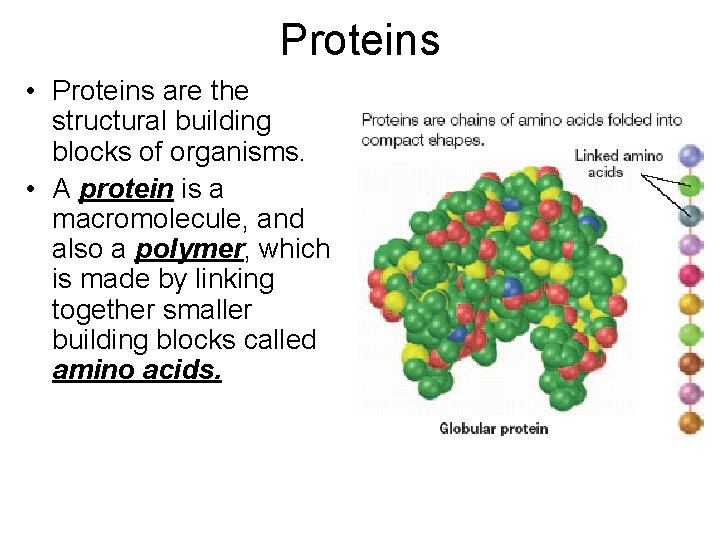
Proteins • Proteins are the structural building blocks of organisms. • A protein is a macromolecule, and also a polymer, which is made by linking together smaller building blocks called amino acids.
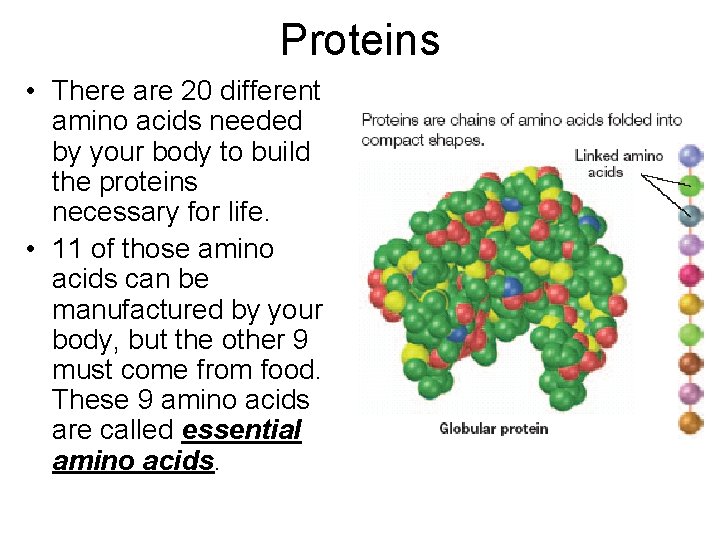
Proteins • There are 20 different amino acids needed by your body to build the proteins necessary for life. • 11 of those amino acids can be manufactured by your body, but the other 9 must come from food. These 9 amino acids are called essential amino acids.



Plant based proteins • Beans, Lentils, Seeds, and Nuts
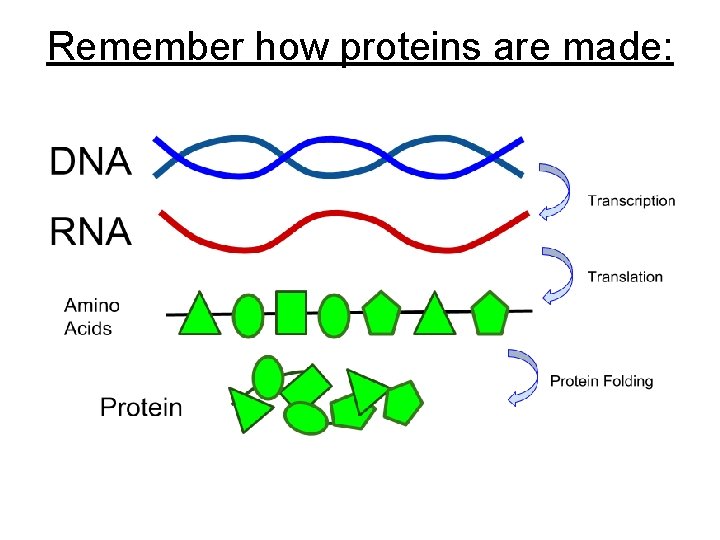
Remember how proteins are made:
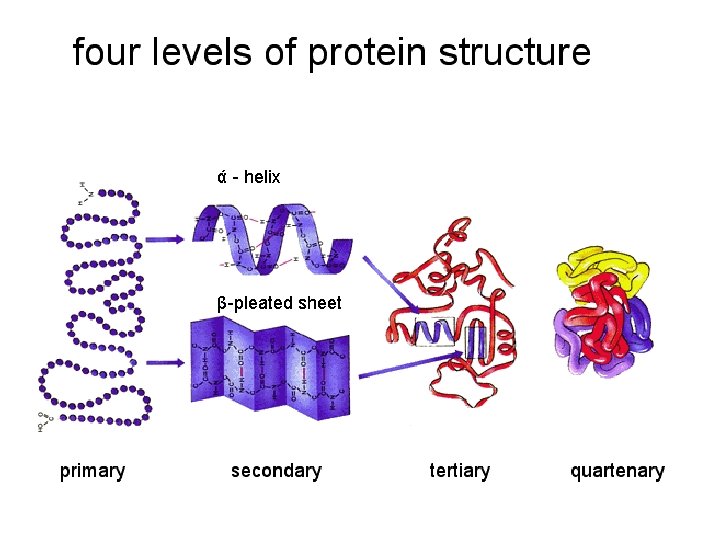
ά - helix β-pleated sheet
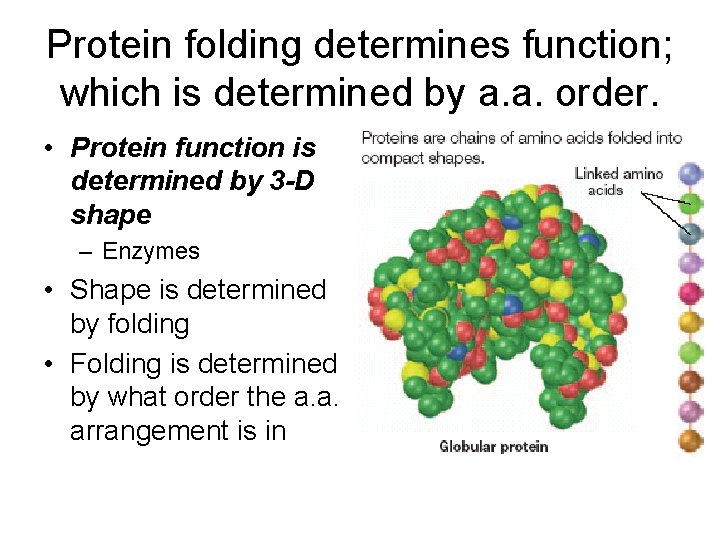
Protein folding determines function; which is determined by a. a. order. • Protein function is determined by 3 -D shape – Enzymes • Shape is determined by folding • Folding is determined by what order the a. a. arrangement is in

Proteins • Proteins are responsible for virtually all the structures of your body. – Hemoglobin carries oxygen in your blood. – Melanin shields your skin from devastating U. V. rays. – Myosin & Actin proteins make up your muscles. – Collagen allows your skin to be elastic. – Keratin helps keep your skin, hair, and nails waterproof.
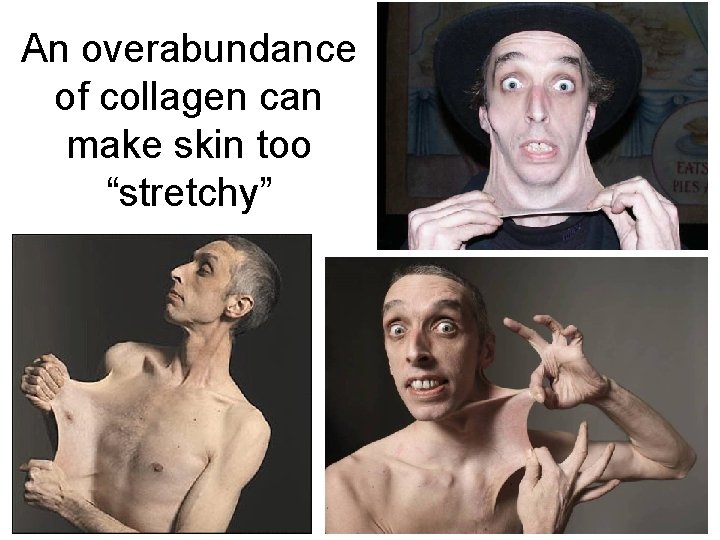
An overabundance of collagen can make skin too “stretchy”

Protein Denaturation • Sometimes a chemical can interact with a protein to break it down. • This is called protein “denaturation”

Fetal Alcohol Syndrome (FAS)
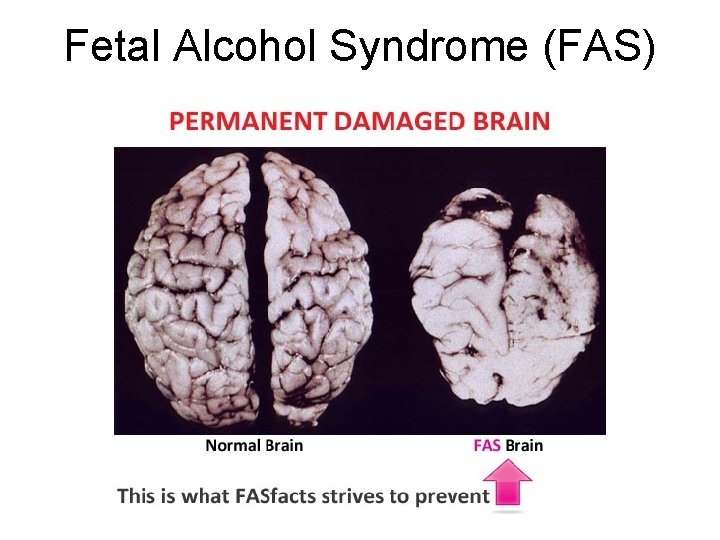
Fetal Alcohol Syndrome (FAS)


Carbohydrates
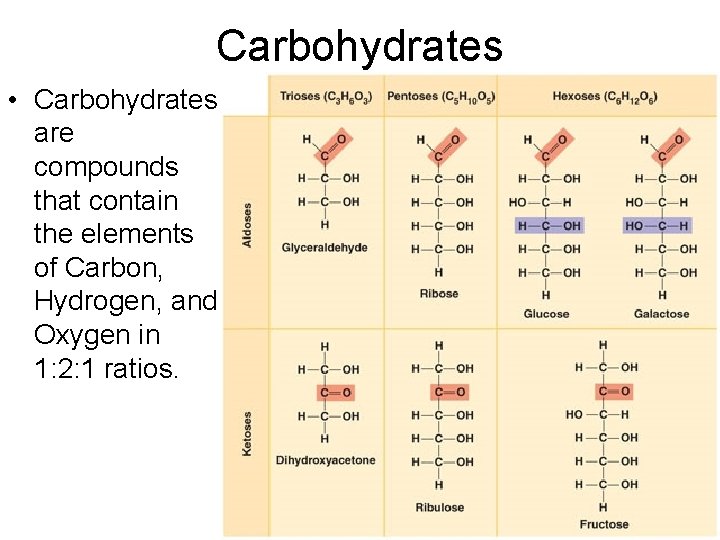
Carbohydrates • Carbohydrates are compounds that contain the elements of Carbon, Hydrogen, and Oxygen in 1: 2: 1 ratios.
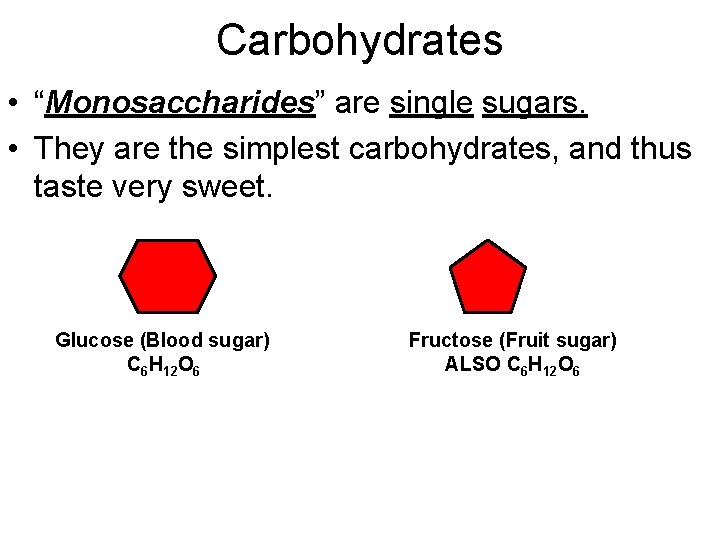
Carbohydrates • “Monosaccharides” are single sugars. • They are the simplest carbohydrates, and thus taste very sweet. Glucose (Blood sugar) C 6 H 12 O 6 Fructose (Fruit sugar) ALSO C 6 H 12 O 6
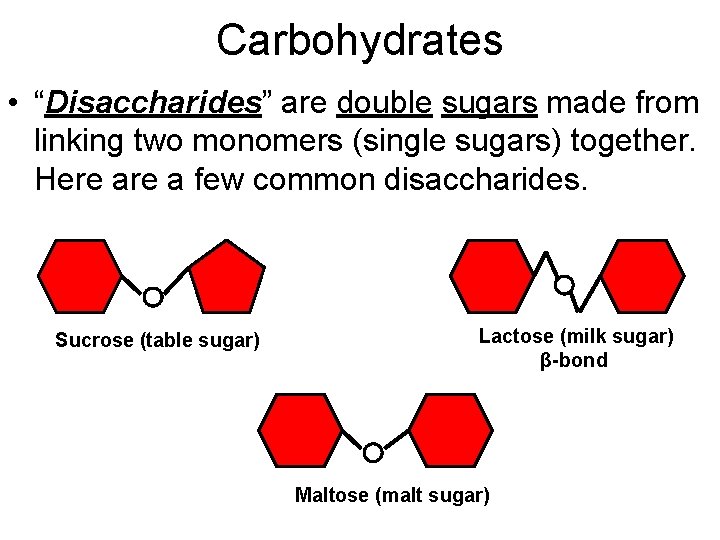
Carbohydrates • “Disaccharides” are double sugars made from linking two monomers (single sugars) together. Here a few common disaccharides. Sucrose (table sugar) Lactose (milk sugar) β-bond Maltose (malt sugar)
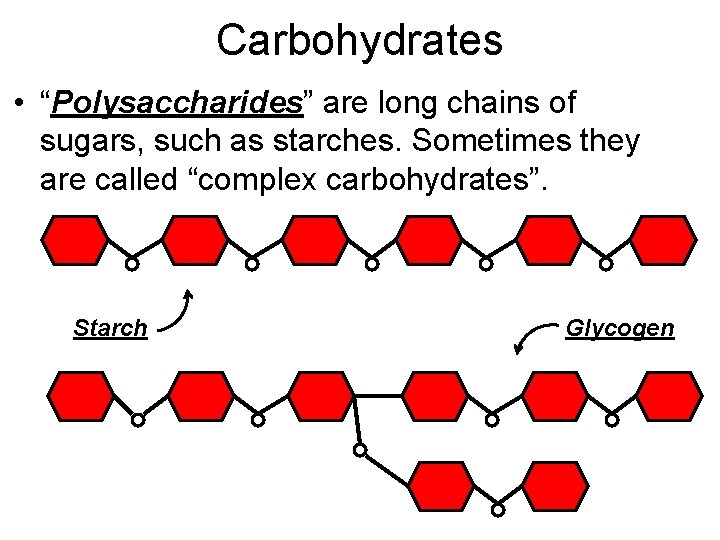
Carbohydrates • “Polysaccharides” are long chains of sugars, such as starches. Sometimes they are called “complex carbohydrates”. Starch Glycogen
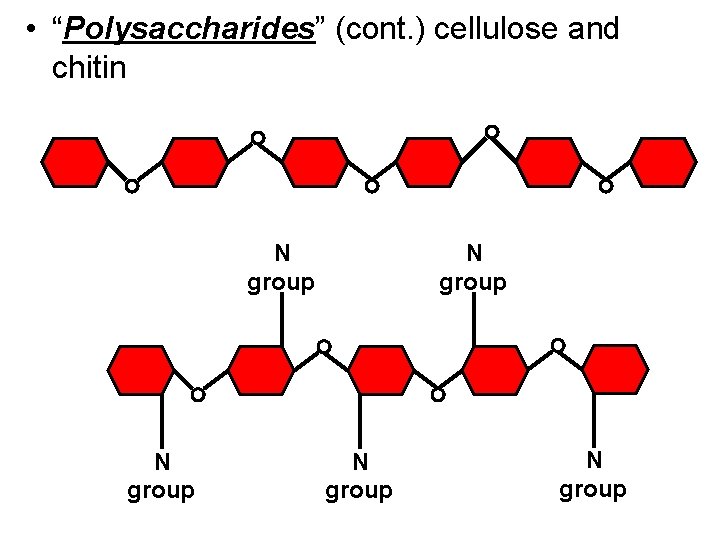
• “Polysaccharides” (cont. ) cellulose and chitin N group N group
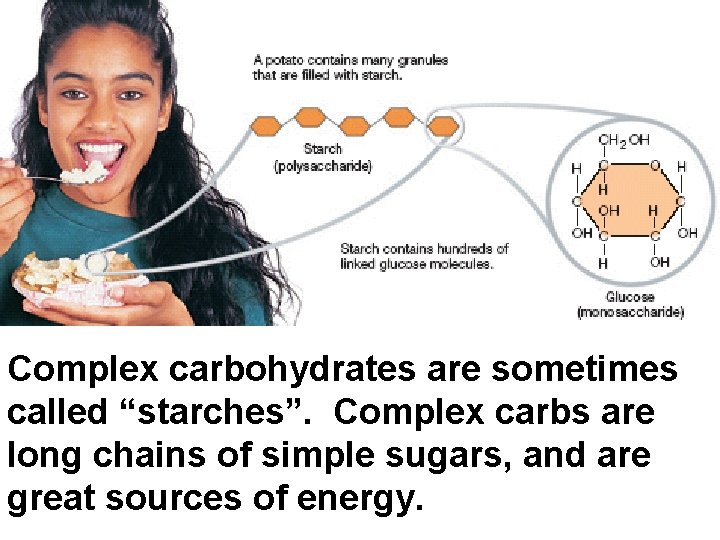
Complex carbohydrates are sometimes called “starches”. Complex carbs are long chains of simple sugars, and are great sources of energy.
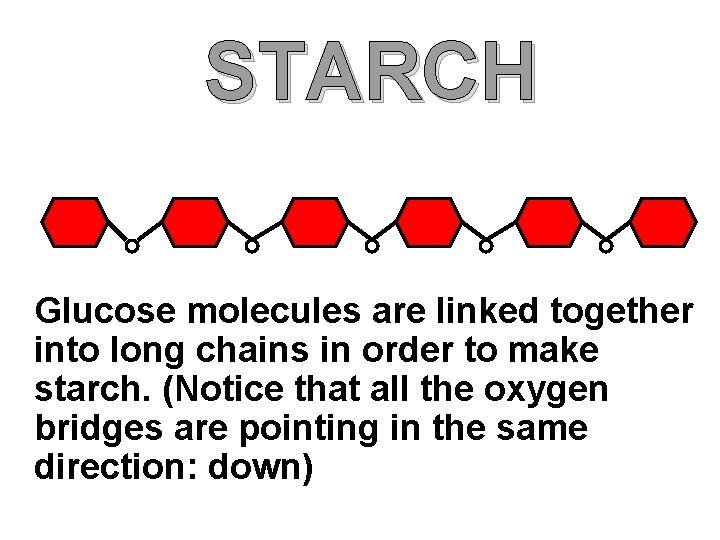
STARCH Glucose molecules are linked together into long chains in order to make starch. (Notice that all the oxygen bridges are pointing in the same direction: down)
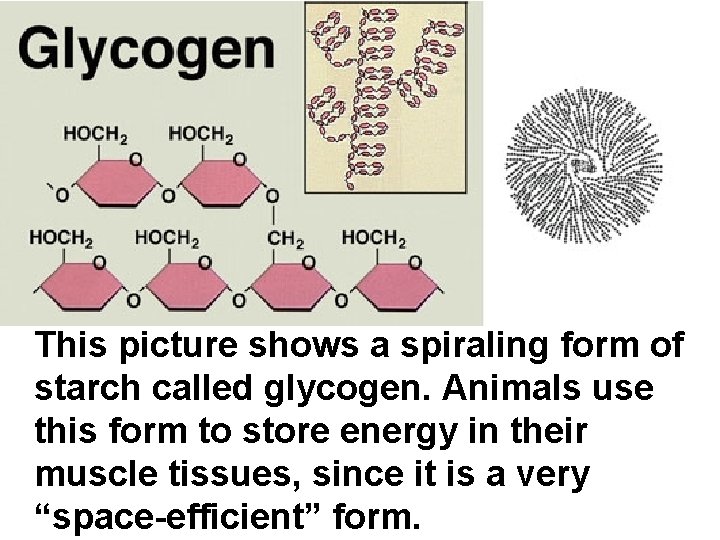
This picture shows a spiraling form of starch called glycogen. Animals use this form to store energy in their muscle tissues, since it is a very “space-efficient” form.
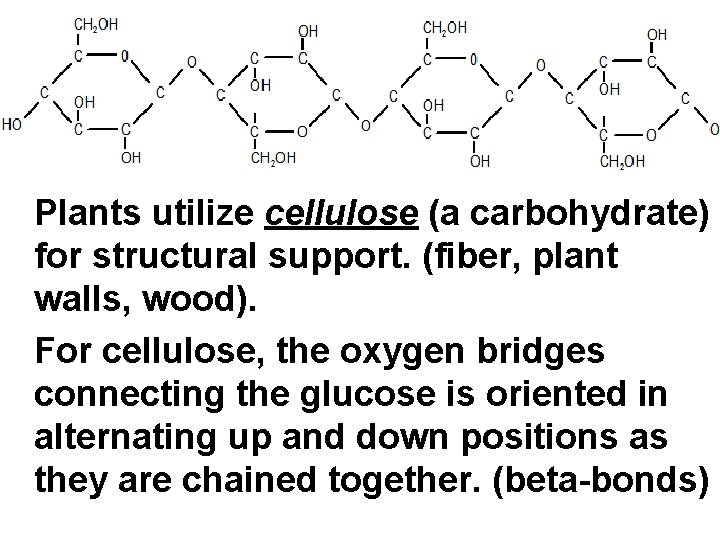
Plants utilize cellulose (a carbohydrate) for structural support. (fiber, plant walls, wood). For cellulose, the oxygen bridges connecting the glucose is oriented in alternating up and down positions as they are chained together. (beta-bonds)
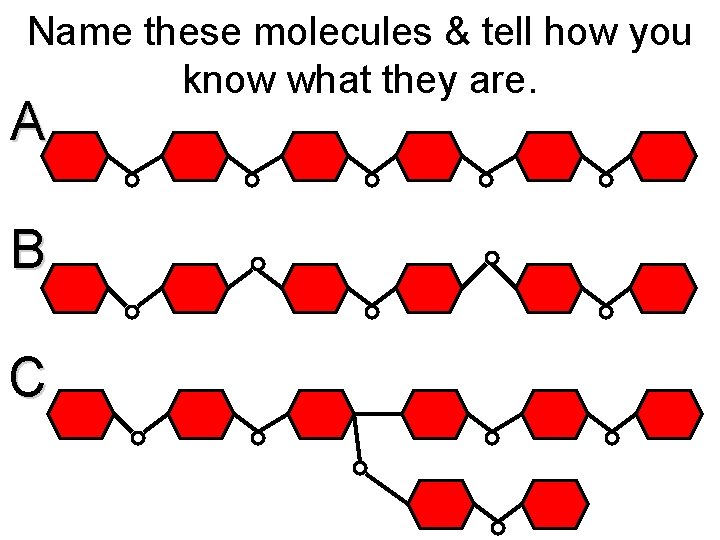
Name these molecules & tell how you know what they are. A B C
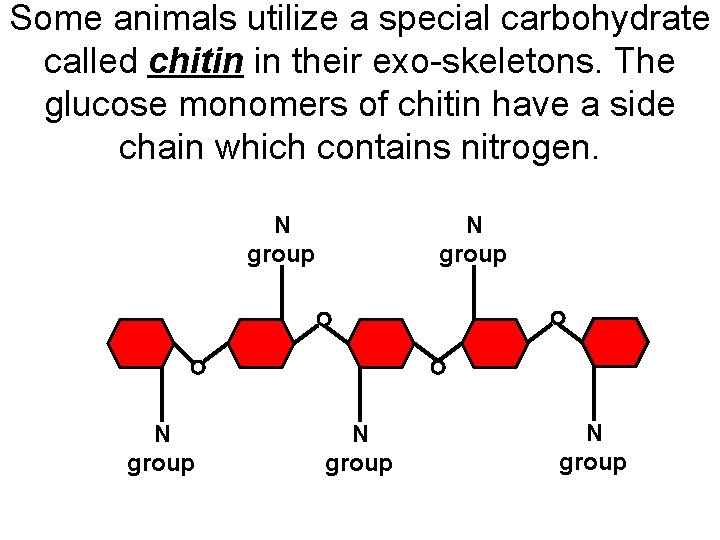
Some animals utilize a special carbohydrate called chitin in their exo-skeletons. The glucose monomers of chitin have a side chain which contains nitrogen. N group N group
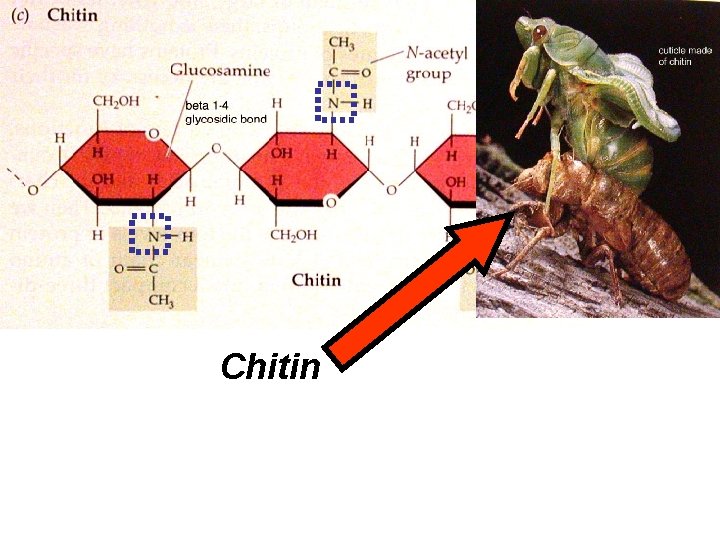
Chitin

Crossword #1 / Quiz #1 Over Proteins and Carbohydrates

Lipids
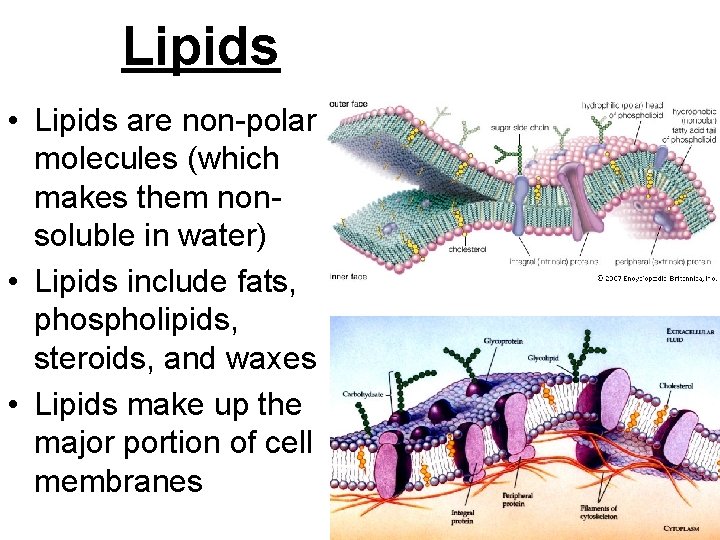
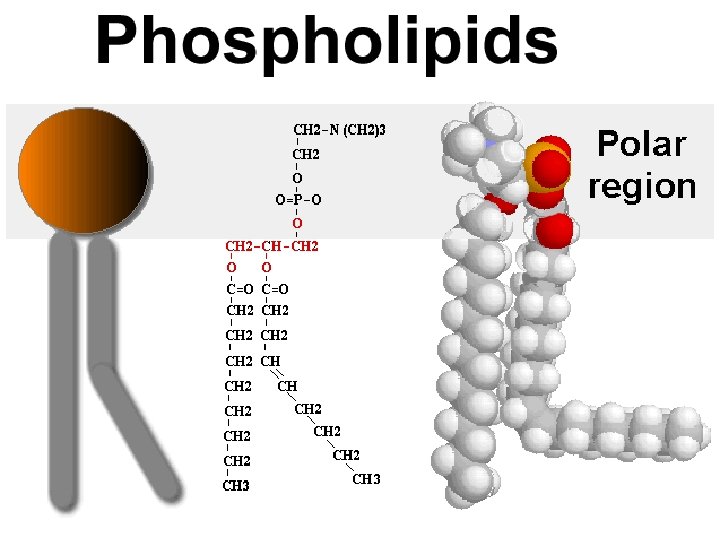
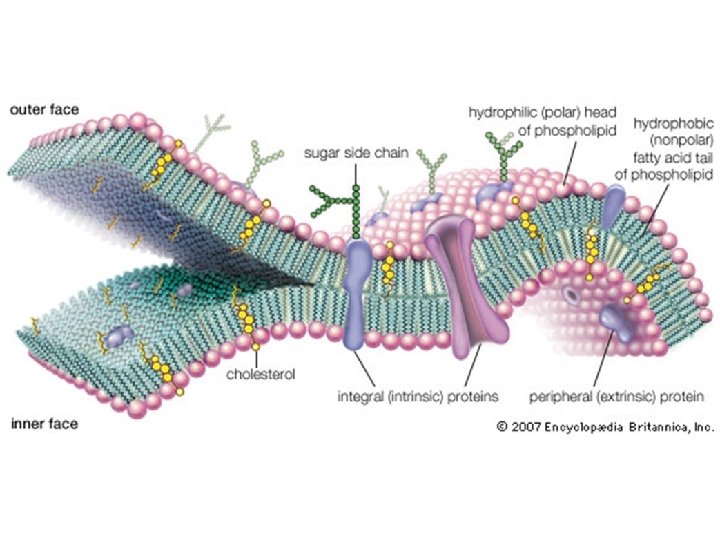
Lipids • Lipids are non-polar molecules (which makes them nonsoluble in water) • Lipids include fats, phospholipids, steroids, and waxes • Lipids make up the major portion of cell membranes

Lipids: • Phospholipids make up the cell membrane lipid bi-layer. • Steroids are hormones such as cholesterol, testosterone, & estrogen. • Fats are energy storage lipids • Waxes can be used for protection; found in the middle ear (ear canal)
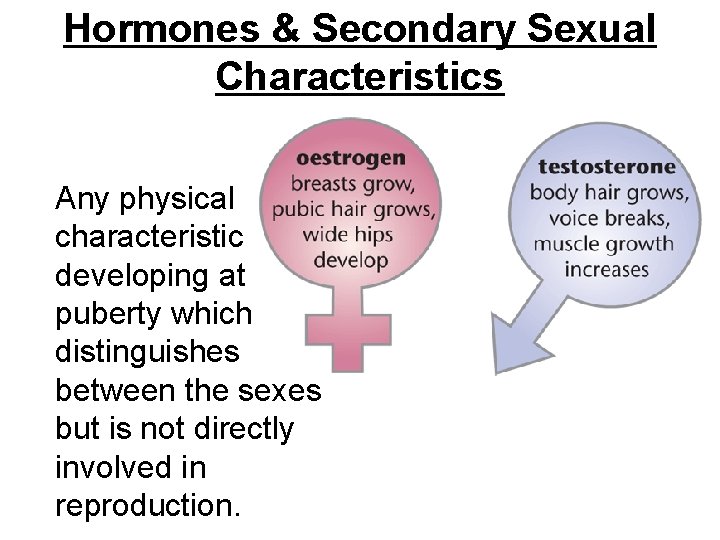
Hormones & Secondary Sexual Characteristics Any physical characteristic developing at puberty which distinguishes between the sexes but is not directly involved in reproduction.
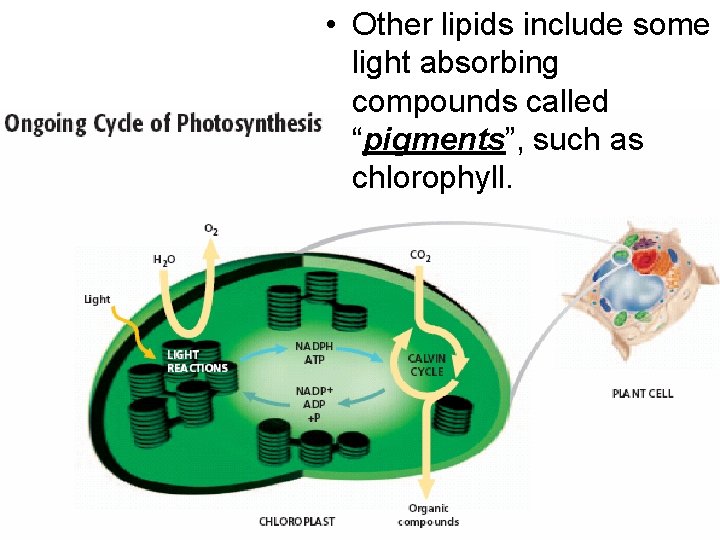
• Other lipids include some light absorbing compounds called “pigments”, such as chlorophyll.
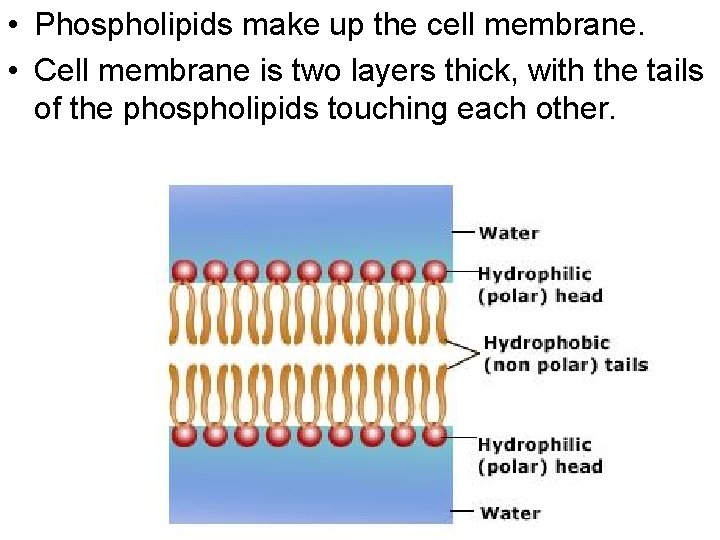
• Phospholipids make up the cell membrane. • Cell membrane is two layers thick, with the tails of the phospholipids touching each other.
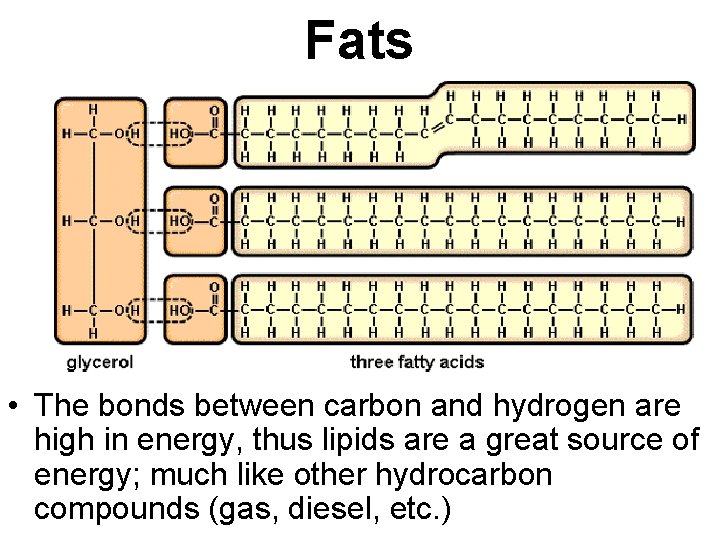
Fats • The bonds between carbon and hydrogen are high in energy, thus lipids are a great source of energy; much like other hydrocarbon compounds (gas, diesel, etc. )
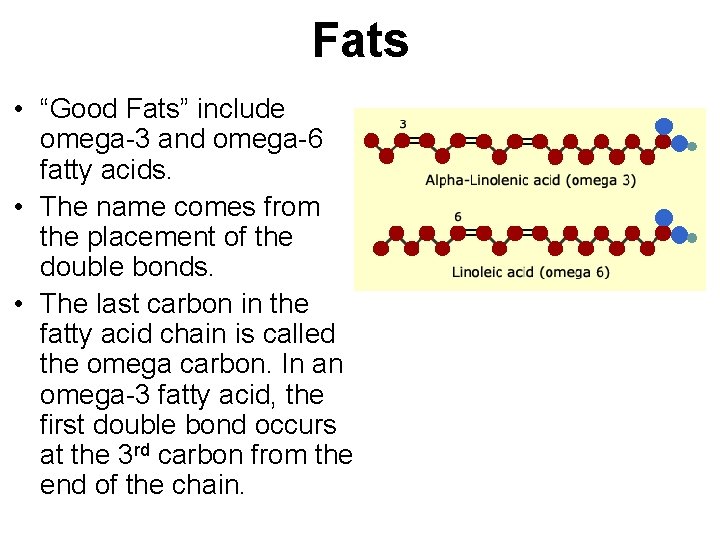
Fats • “Good Fats” include omega-3 and omega-6 fatty acids. • The name comes from the placement of the double bonds. • The last carbon in the fatty acid chain is called the omega carbon. In an omega-3 fatty acid, the first double bond occurs at the 3 rd carbon from the end of the chain.
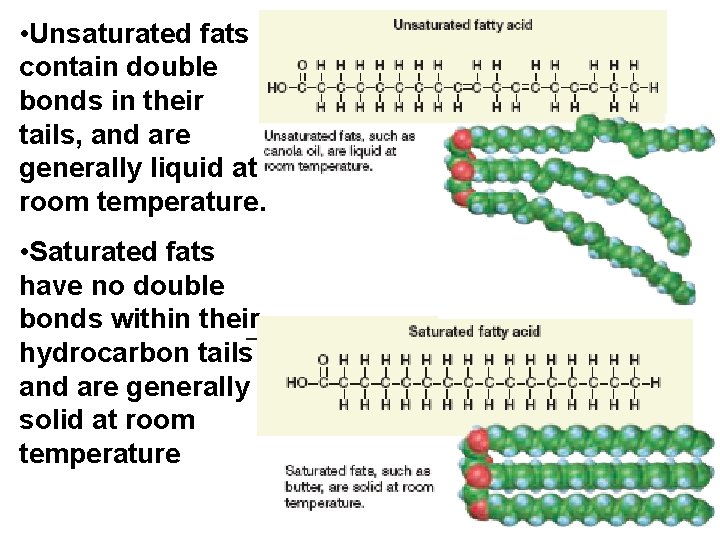
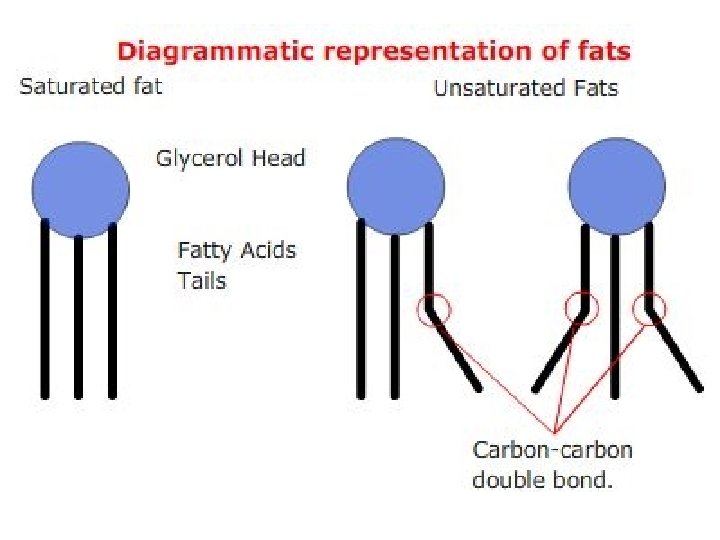
• Unsaturated fats contain double bonds in their tails, and are generally liquid at room temperature. • Saturated fats have no double bonds within their hydrocarbon tails and are generally solid at room temperature

• Plant fats are generally unsaturated (such as sunflower oil), while fats derived from animal sources are generally saturated (such as lard).
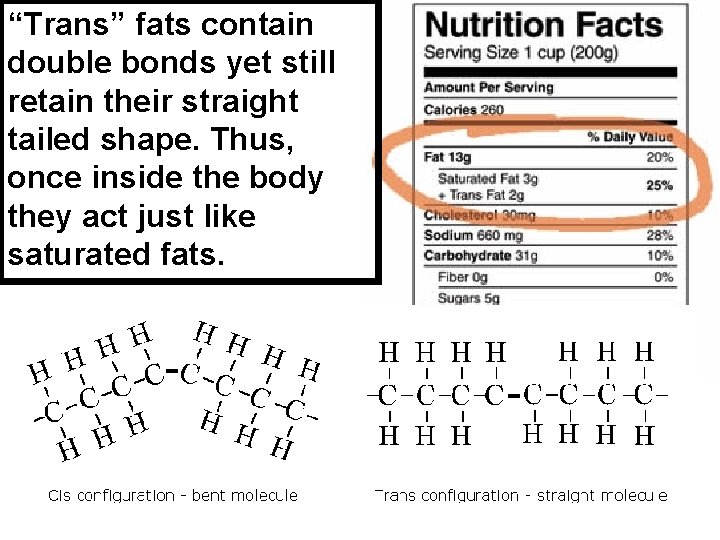
“Trans” fats contain double bonds yet still retain their straight tailed shape. Thus, once inside the body they act just like saturated fats.
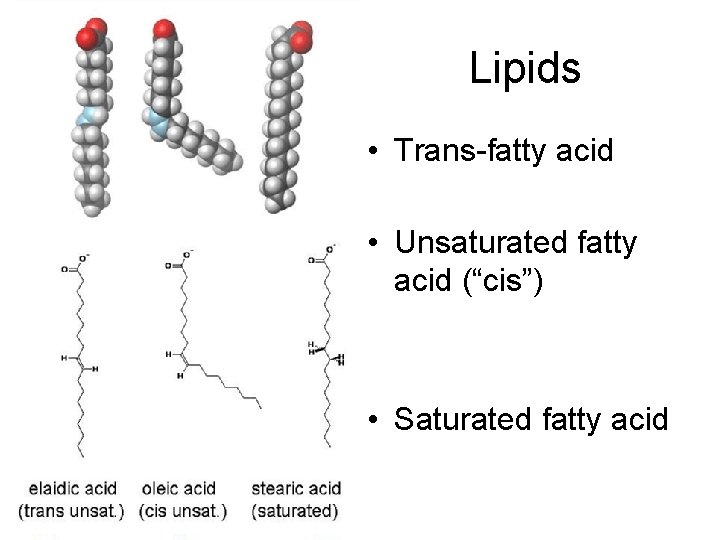
Lipids • Trans-fatty acid • Unsaturated fatty acid (“cis”) • Saturated fatty acid
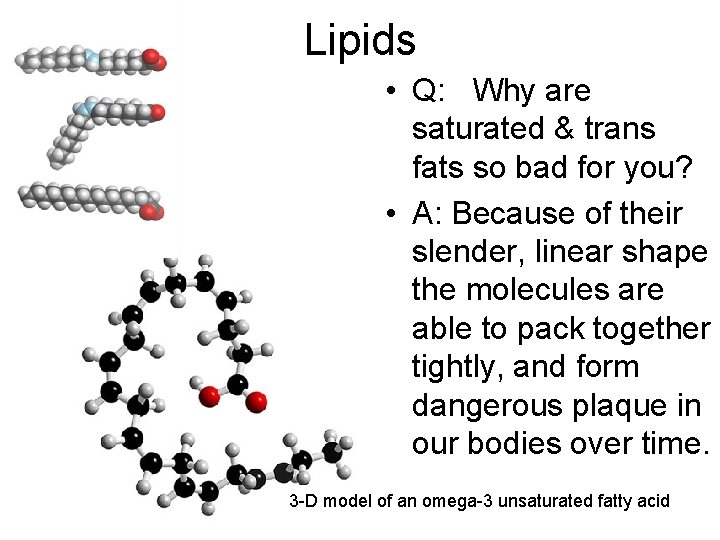
Lipids • Q: Why are saturated & trans fats so bad for you? • A: Because of their slender, linear shape the molecules are able to pack together tightly, and form dangerous plaque in our bodies over time. 3 -D model of an omega-3 unsaturated fatty acid

Nucleic Acids
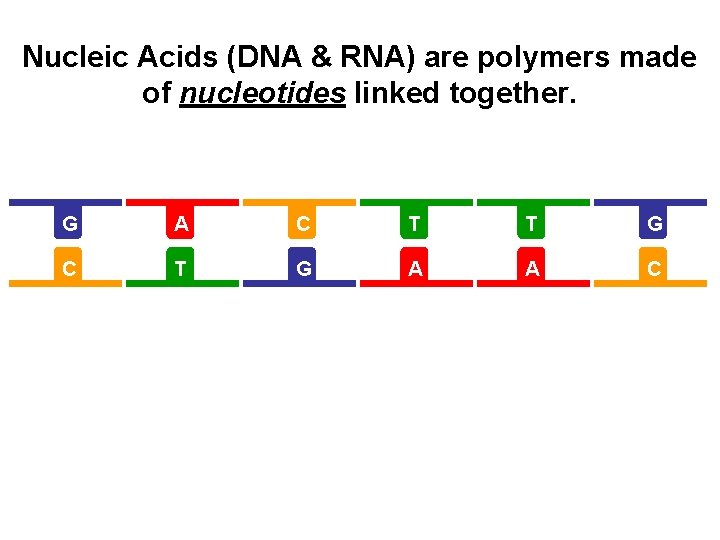
Nucleic Acids (DNA & RNA) are polymers made of nucleotides linked together. G A C T T G C T G A A C
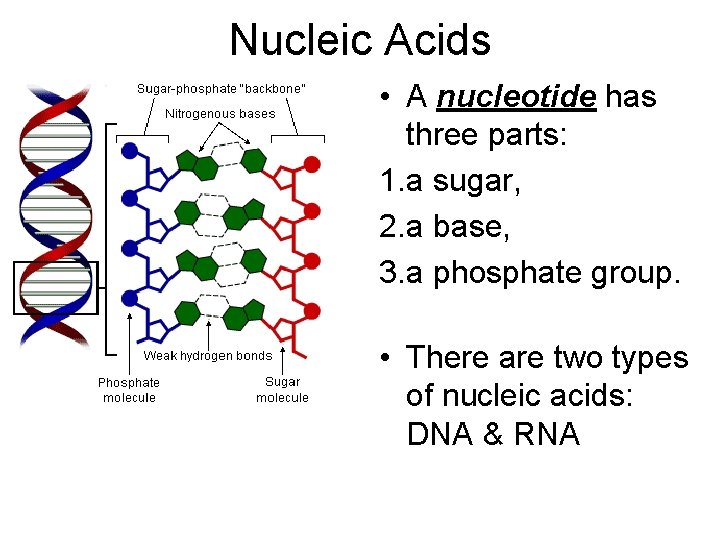
Nucleic Acids • A nucleotide has three parts: 1. a sugar, 2. a base, 3. a phosphate group. • There are two types of nucleic acids: DNA & RNA
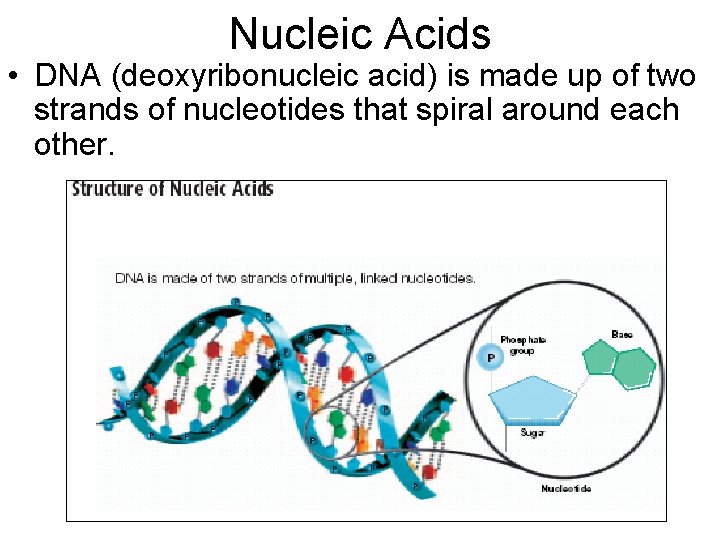
Nucleic Acids • DNA (deoxyribonucleic acid) is made up of two strands of nucleotides that spiral around each other.
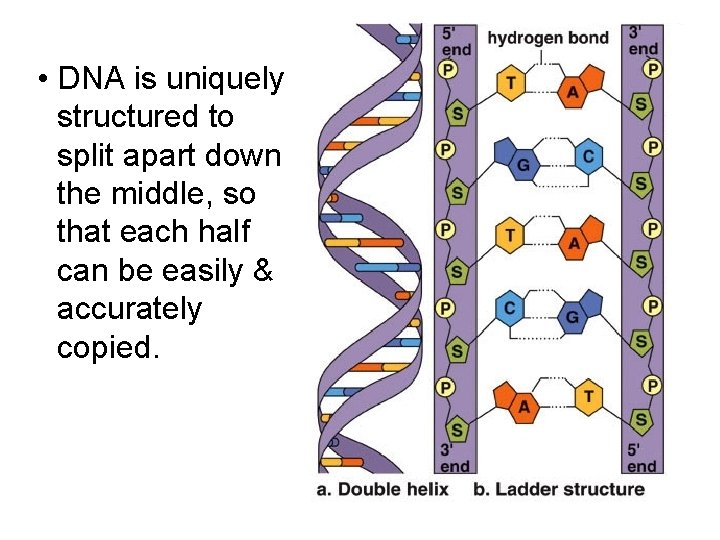
• DNA is uniquely structured to split apart down the middle, so that each half can be easily & accurately copied.
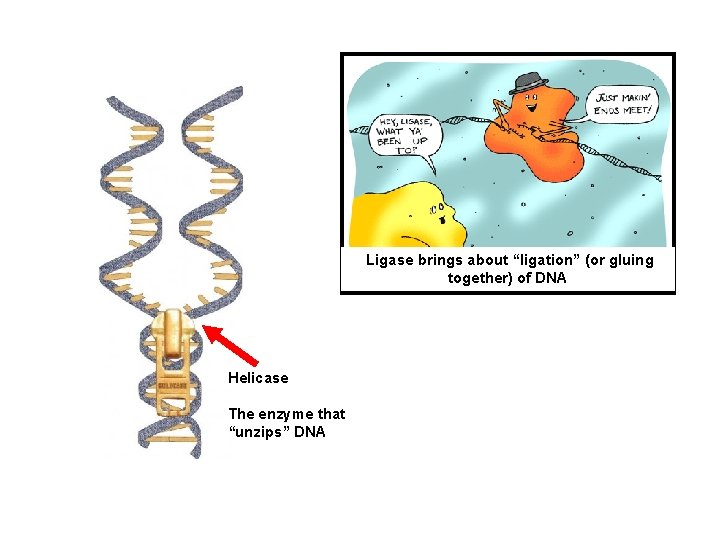
Ligase brings about “ligation” (or gluing together) of DNA Helicase The enzyme that “unzips” DNA

Mutations There are three main types of point mutations: 1. Substitution – one nucleotide is substituted for another. 2. Deletion - a nucleotide is removed from the DNA strand not replaced. 3. Insertion – an extra nucleotide is inserted into the DNA A C A Substitution A T G A C G A
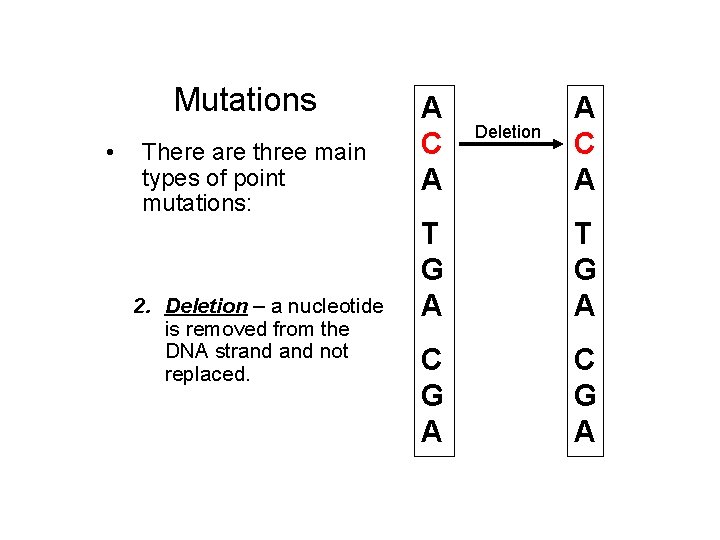
Mutations • There are three main types of point mutations: 1. Substitution – one nucleotide is substituted for another. 2. Deletion – a nucleotide is removed from the DNA strand not replaced. 3. Insertion – An extra nucleotide is inserted into the strand. A C A Deletion A C A A T T G A T G G A A C C G A C G G A A
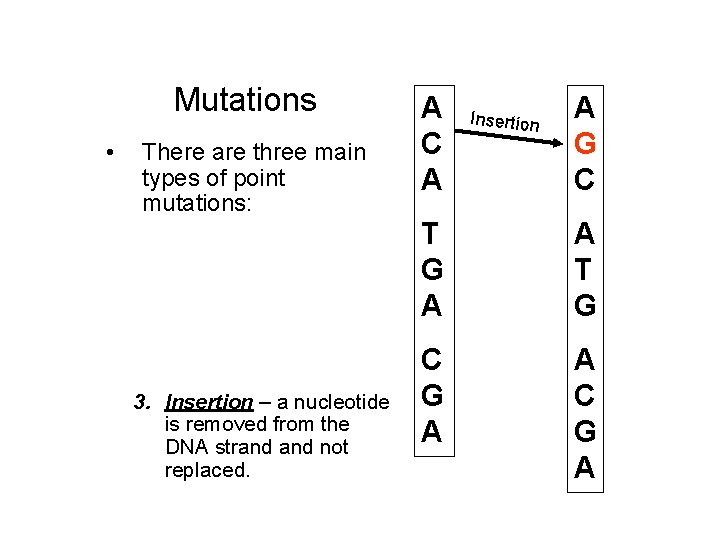
Mutations • There are three main types of point mutations: 1. Substitution – one nucleotide is substituted for another. 2. Deletion – a nucleotide is removed from the DNA strand not replaced. 3. Insertion – a nucleotide is removed from the DNA strand not replaced. A C A Insertion A G C T G A A T G C G A A C G A

Mutations • Mutations change the message that is contained in our DNA! Consider the following sentence: HOW FAR DID THE DOG RUN Point mutation (substitution) HOW FAT DID THE DOG RUN The sentence meaning has changed a little, but it is still readable.

Mutations • A frameshift mutation is much more destructive to the genetic code. Lets take the same sentence, but let’s insert a Z into the third word: HOW FAR DID THE DOG RUN Insertion (“Z”) One “reading frame” HOW FAT RDI DTH EDO GRU N The insertion of just one letter has caused all the other letters to shift over one place. This kind of a mutation is called a frameshift.
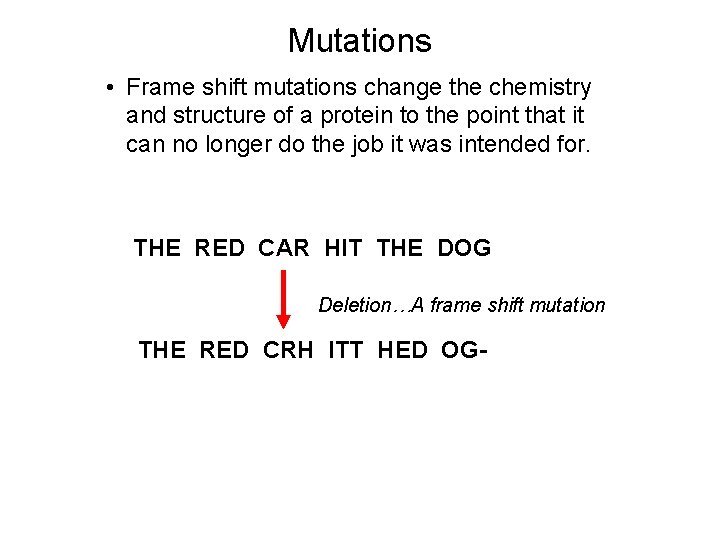
Mutations • Frame shift mutations change the chemistry and structure of a protein to the point that it can no longer do the job it was intended for. THE RED CAR HIT THE DOG Deletion…A frame shift mutation THE RED CRH ITT HED OG-
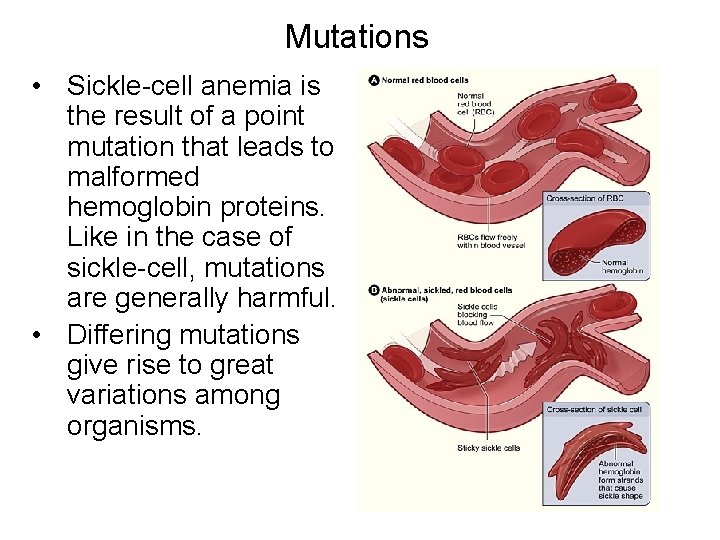
Mutations • Sickle-cell anemia is the result of a point mutation that leads to malformed hemoglobin proteins. Like in the case of sickle-cell, mutations are generally harmful. • Differing mutations give rise to great variations among organisms.
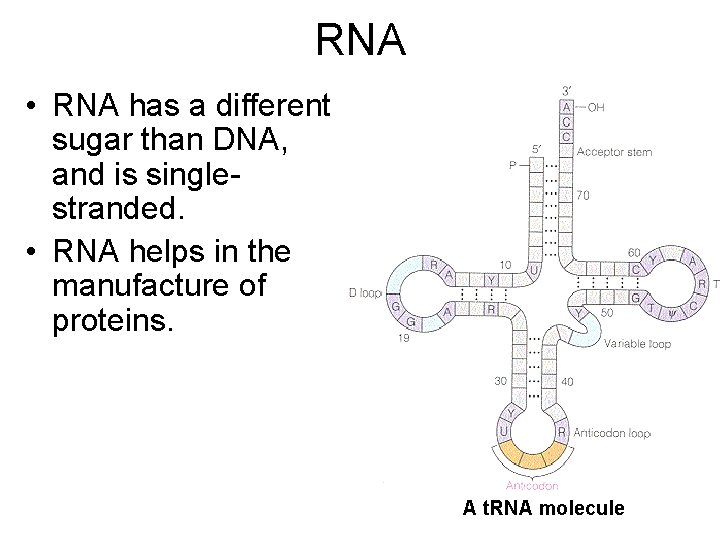
RNA • RNA has a different sugar than DNA, and is singlestranded. • RNA helps in the manufacture of proteins. A t. RNA molecule
- Slides: 83