3 D BIOCHEMICAL MODELING USING NUCLEAR MAGNETIC RESONANCE

3 D BIOCHEMICAL MODELING USING NUCLEAR MAGNETIC RESONANCE MICROSCOPY BME 281 Presentation 2 – Ron Greene

THE IMPORTANCE OF VISUALIZATION “The few wonders of the world only exist while there are those with the sight to see them. ” – Charles de Lint (Celtic folk musician )
A BRIEF HISTORY OF 3 D MOLECULAR MODELING 1609 - Galileo Galilei develops compound microscope 1951 - Erwin Müller invents field ion microscope; sees first atoms 1964 - Aaron Klug shows Xray diffraction could be used to develop crystallographic electron microscopy 1971 - Protein Data Bank established as a repository for 3 D structural data of proteins and nucleic acids 1978 - Kurth Würthrich used NMR to determine protein structures 1981 - Don Wiley determined the structure of the hemagglutinin protein from the surface of the influenza virus 1998 - Rod Mac. Kinnon publishes first high-resolution structure of an ion channel 2007 - Brian Kobilka solves first structure of a human G protein coupled receptor
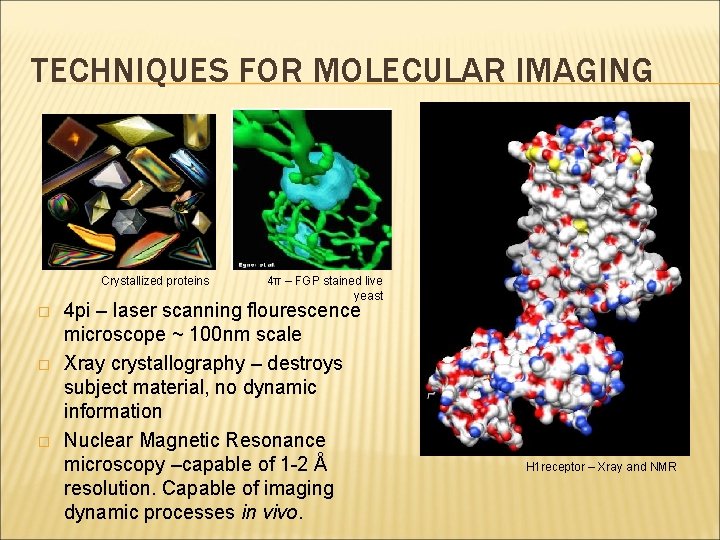
TECHNIQUES FOR MOLECULAR IMAGING Crystallized proteins � � � 4π – FGP stained live yeast 4 pi – laser scanning flourescence microscope ~ 100 nm scale Xray crystallography – destroys subject material, no dynamic information Nuclear Magnetic Resonance microscopy –capable of 1 -2 Å resolution. Capable of imaging dynamic processes in vivo. H 1 receptor – Xray and NMR

WHAT IS NUCLEAR MAGNETIC RESONANCE? � � � Spin – fundamental physical property of protons, electrons and neutrons Nuclear Magnetic Resonance – a phenomenon which occurs in certain atoms when exposed to magnetic field Elements with enough net spin resonate with detectable magnitudes

HOW DOES ONE MAKE A NUCLEUS RESONATE? When placed in a magnetic field of strength B, a particle with a net spin can absorb a photon, of frequency ω. Chemical shift affects the Larmor frequency at which a given atom resonates Chemical shift is a precise metric of the chemical environment of an atom Chemical shift After hitting with pulse to elevate energy states, NMR is measured by induction as nuclei relax to lower state Gyroscopic ratio Larmor frequency
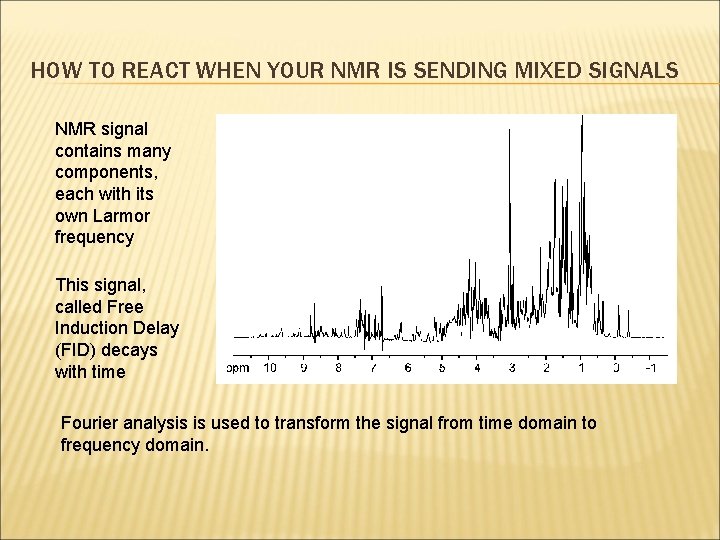
HOW TO REACT WHEN YOUR NMR IS SENDING MIXED SIGNALS NMR signal contains many components, each with its own Larmor frequency This signal, called Free Induction Delay (FID) decays with time Fourier analysis is used to transform the signal from time domain to frequency domain.

“Here’s the plan: We put the band back together, do some gigs, earn some bread, bang!” – Jake Blues

TO INFINITY AND BEYOND The Future: The Protein Data Bank continues to expand, including more proteins in more of their naturally occurring conformations NMR microscopy and clinical MRI merge to offer real time imaging of biological processes on an atomic scale
![MAGNET THERAPY WORKS, MAN REFERENCES [1] URI BME 281 BME Seminar II <www. ele. MAGNET THERAPY WORKS, MAN REFERENCES [1] URI BME 281 BME Seminar II <www. ele.](http://slidetodoc.com/presentation_image_h/a51d2c6dd130cc844d833eb45bb82547/image-10.jpg)
MAGNET THERAPY WORKS, MAN REFERENCES [1] URI BME 281 BME Seminar II <www. ele. uri. edu/courses/bme 281>. [2] J Puerta-Fonolla, T Vasquez-Osorio, J ruiz-Cabello, J Murillo-Gonzalez, A Pena-Melian. "Margentic resonance microscopy versus light microscopy in embryology teaching. " Clinical Anatomy (2004): 429 -435. [3] Oleg Yu Federoff, Miguel Salazar, Haiyong Han, Violetta Chemeris, Sean Kerwin, Laurence Hurley. "NMR-Based Model of a Telomerase-Inhibiting Compound Bound to G-quadruplex DNA. " Biochemistry (1998): 12367 -12374. [4] Seui Ogawa, david Tank, Ravi Menon, Juitta Ellerman, Seong-Gi Kim, Hellmut Merkle, Kamil Ugurbil. "Intrinsic signal changes accompanying sensory stiimulation: Functional brain mapping with magnetic resonabce imaging. " Proceedings of the National Academy of Science (1992): 5951 -5955. [5] wiki. "Nuclear magnetic Resonance. " 22 October 2011. Wikipedia. 5 November 2011 <http: //en. wikipedia. org/wiki/Nuclear_magnetic_resonance#NMR_spectroscopy>.
- Slides: 10