3 CHAPTER engel Boles Thermodynamics The First Law

3 CHAPTER Çengel Boles Thermodynamics The First Law of Thermodynamics: Closed Systems Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
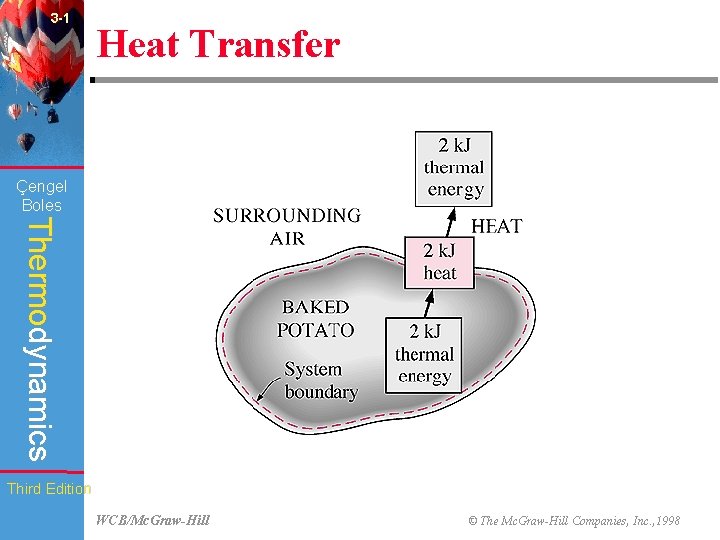
3 -1 Heat Transfer (Fig. 3 -3) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
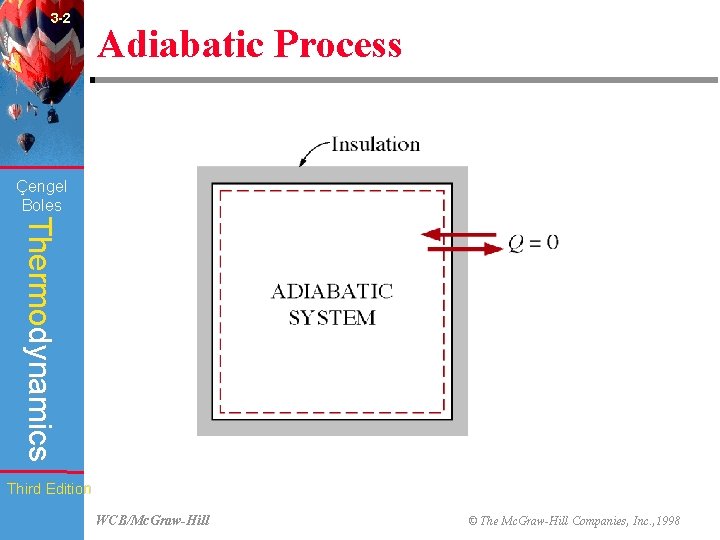
3 -2 Adiabatic Process (Fig. 3 -4) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
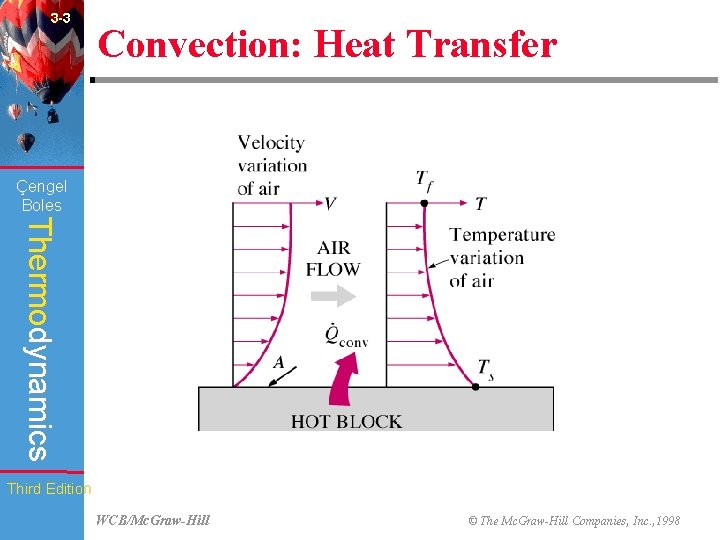
3 -3 Convection: Heat Transfer (Fig. 3 -8) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
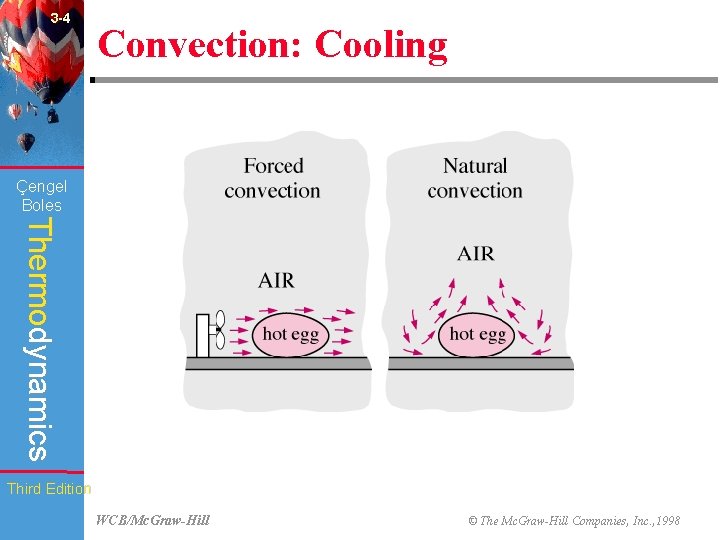
3 -4 Convection: Cooling (Fig. 3 -9) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
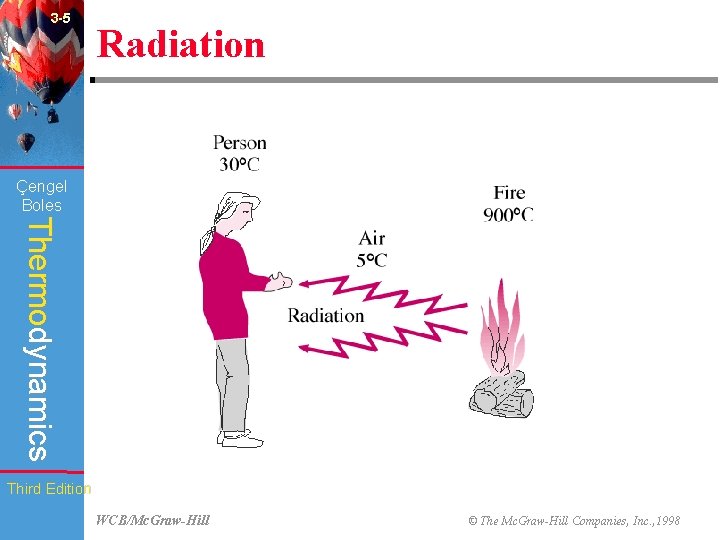
3 -5 Radiation (Fig. 3 -10) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
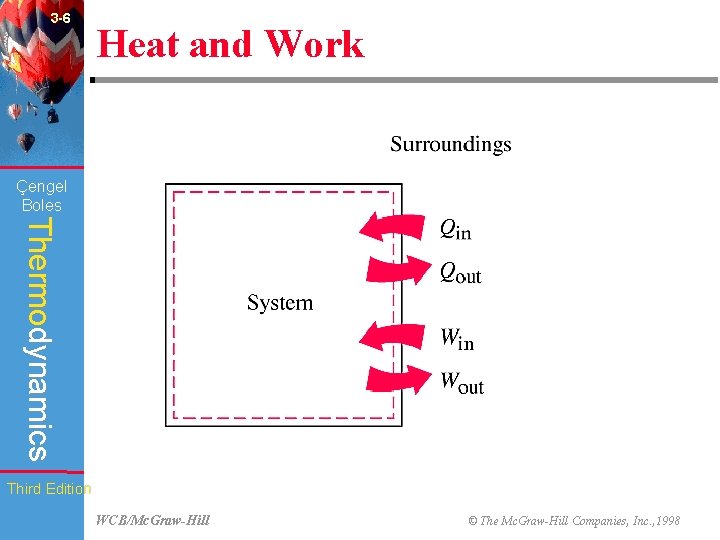
3 -6 Heat and Work (Fig. 3 -15) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
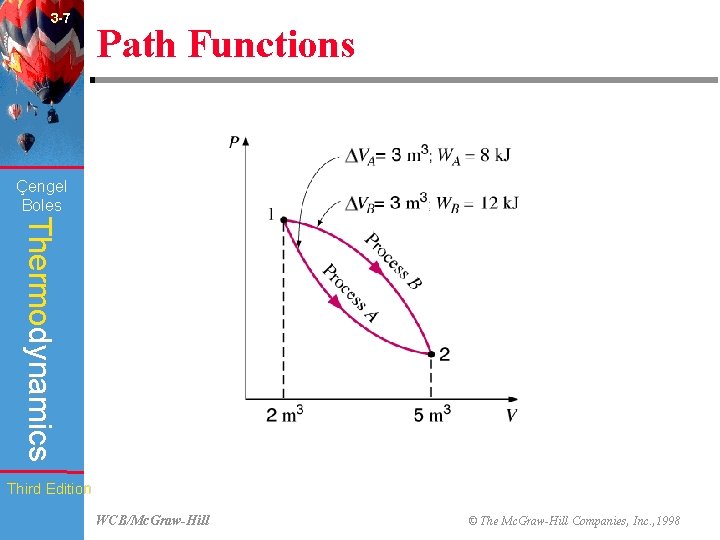
3 -7 Path Functions (Fig. 3 -16) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
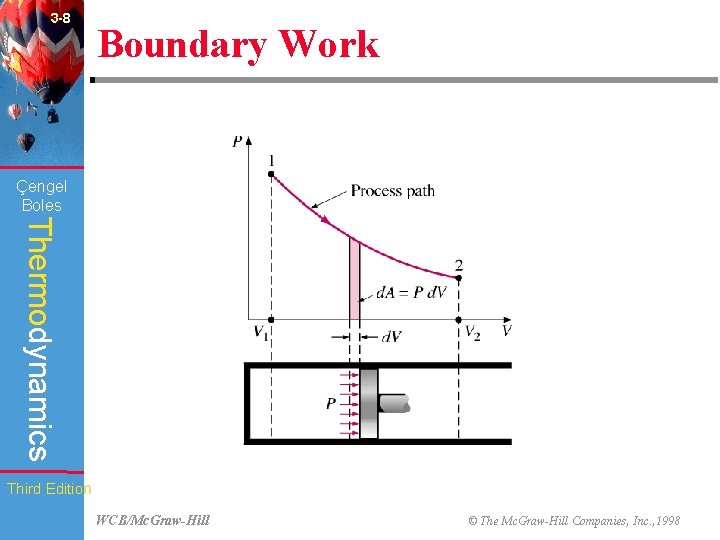
3 -8 Boundary Work (Fig. 3 -27) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
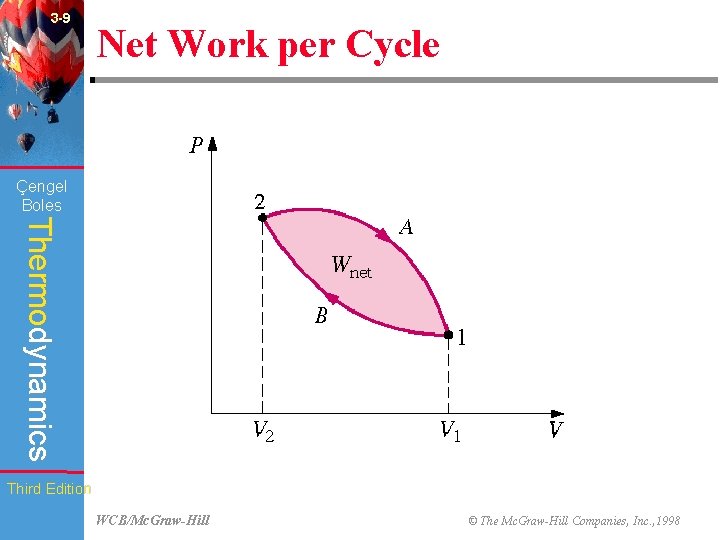
3 -9 Net Work per Cycle (Fig. 3 -29) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
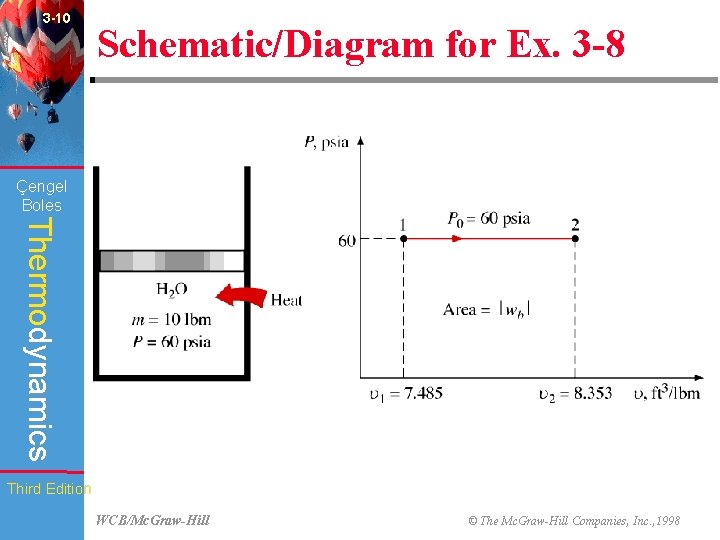
3 -10 Schematic/Diagram for Ex. 3 -8 (Fig. 3 -31) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
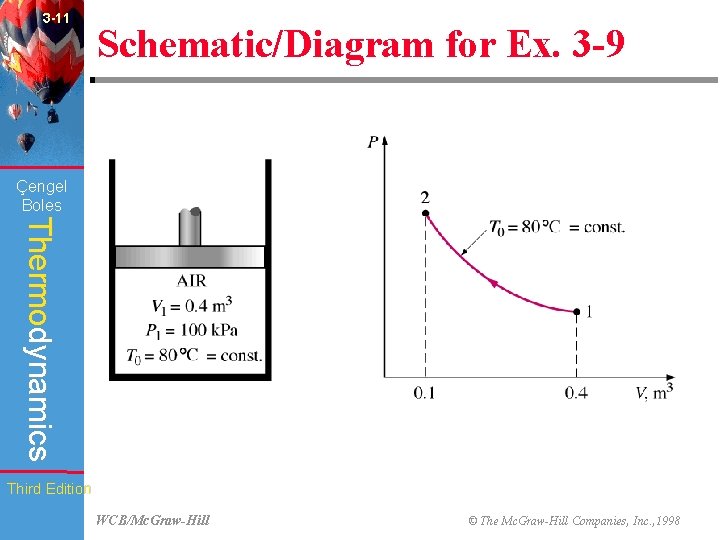
3 -11 Schematic/Diagram for Ex. 3 -9 (Fig. 3 -32) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
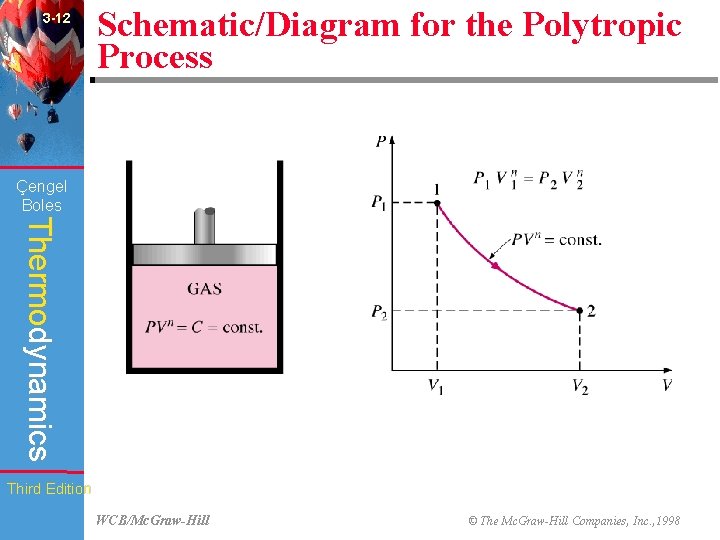
3 -12 Schematic/Diagram for the Polytropic Process (Fig. 3 -33) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
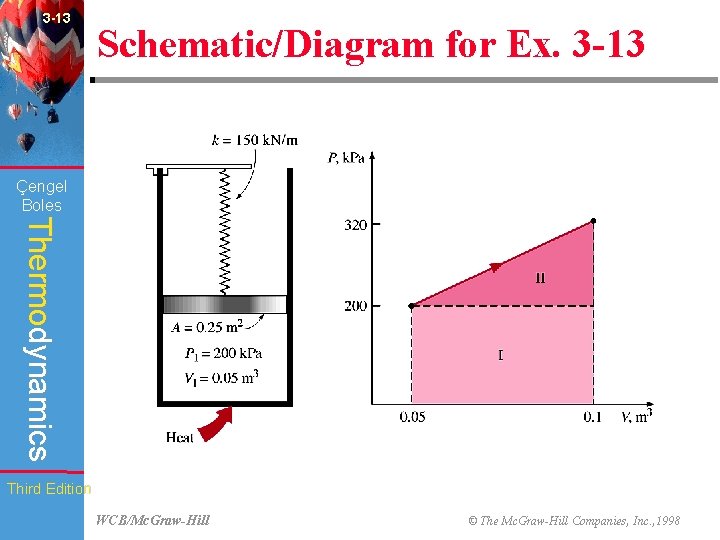
3 -13 Schematic/Diagram for Ex. 3 -13 (Fig. 3 -43) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
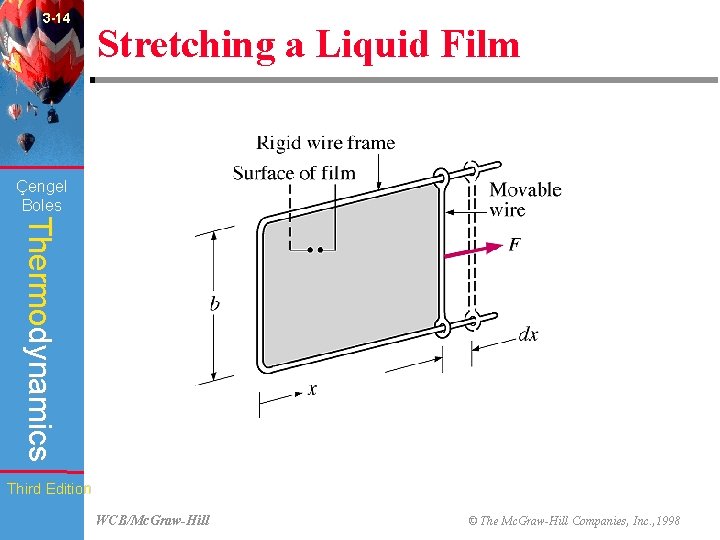
3 -14 Stretching a Liquid Film (Fig. 3 -45) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
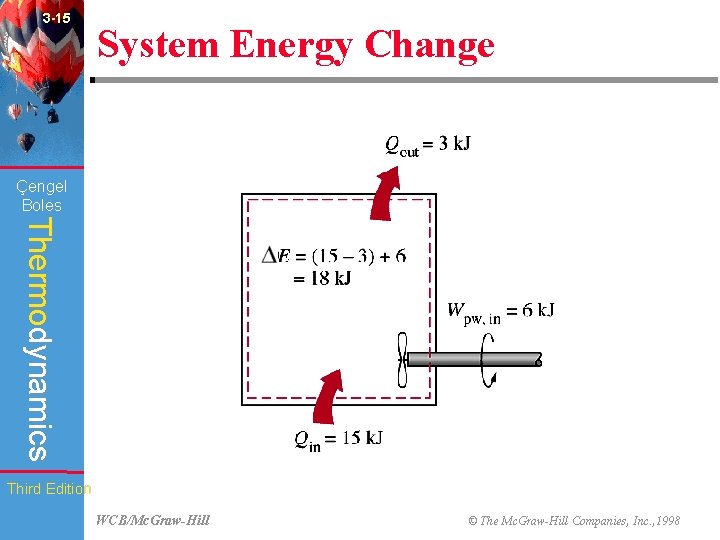
3 -15 System Energy Change (Fig. 3 -52) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
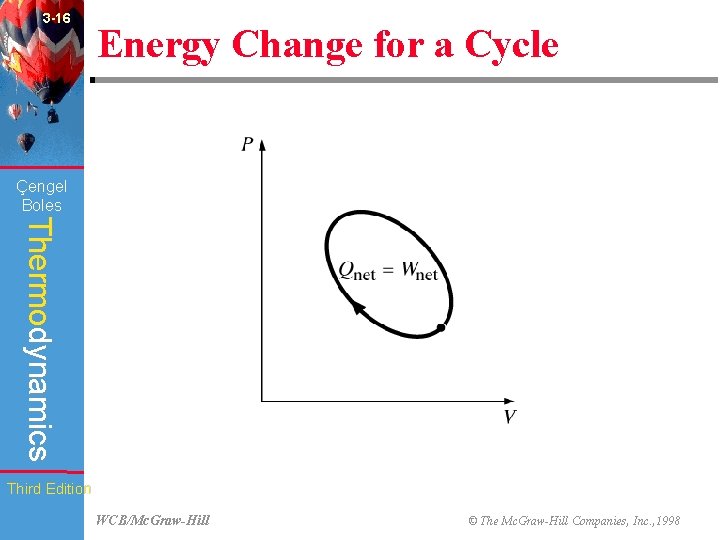
3 -16 Energy Change for a Cycle (Fig. 3 -54) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -17 Closed-Systems, First-Law (Fig. 3 -55) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -18 Formal Definitions of Cv and Cp (Fig. 3 -72) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
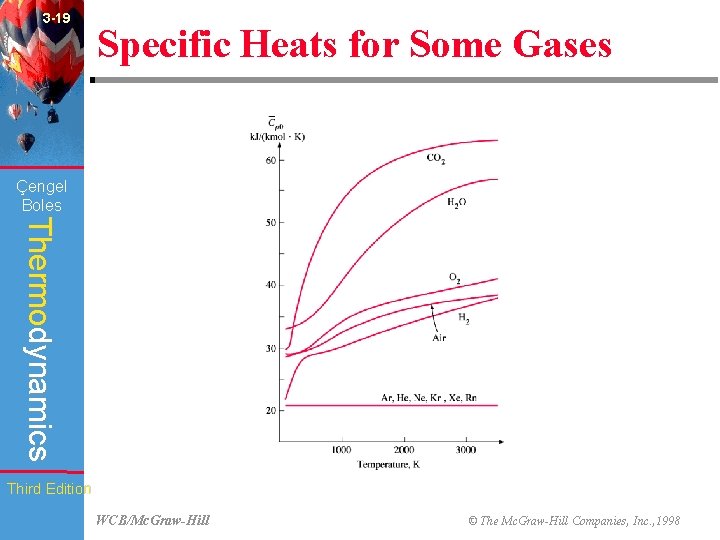
3 -19 Specific Heats for Some Gases (Fig. 3 -76) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
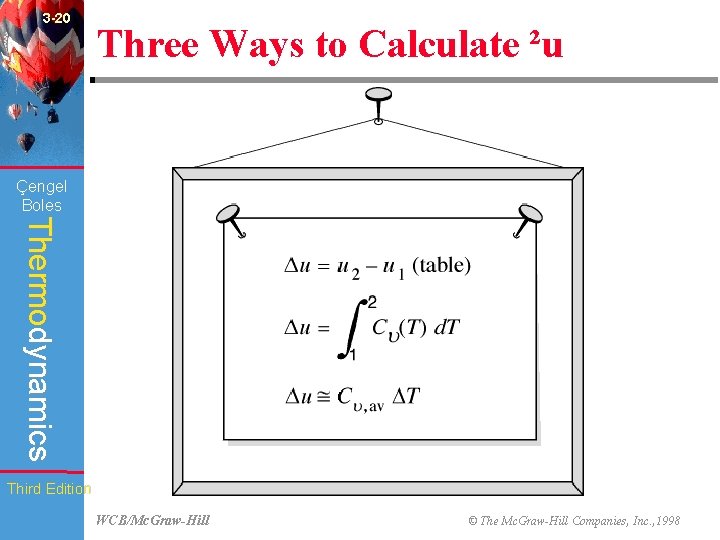
3 -20 Three Ways to Calculate ²u (Fig. 3 -80) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
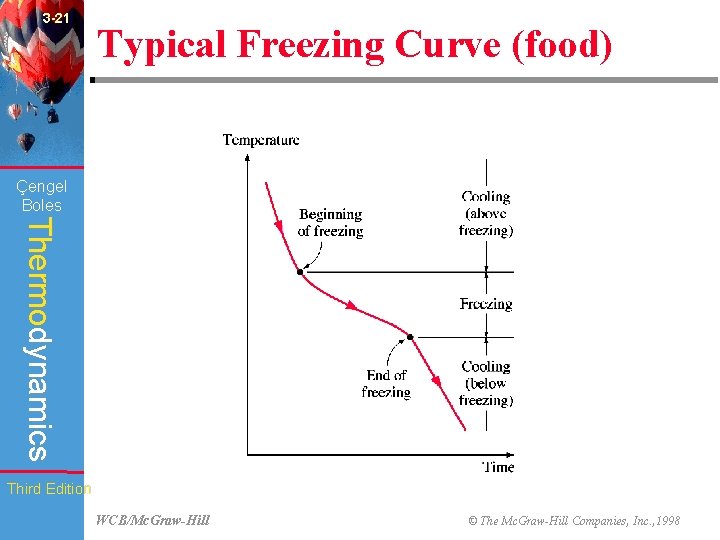
3 -21 Typical Freezing Curve (food) (Fig. 3 -91) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -22 Çengel Boles Chapter Summary Thermodynamics • The first law of thermodynamics is essentially an expression of the conservation of energy principle. Energy can cross the boundaries of a closed system in the form of heat or work. • If the energy transfer across the boundaries of a closed system is due to a temperature difference, it is heat; otherwise, it is work. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -23 Chapter Summary • Heat is transferred in three ways: conduction, convection, and radiation. Çengel Boles Thermodynamics Third Edition › Conduction is the transfer of energy from the more energetic particles of a substance to the adjacent less energetic ones as a result of interactions between the particles. › Convection is the mode of energy transfer between a solid surface and the adjacent liquid or gas that is in motion, and it involves the combined effects of conduction and fluid motion. › Radiation is the energy emitted by matter in the form of electromagnetic waves (or photons) as a result of the changes in the electronic configurations of the atoms or molecules. WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -24 Chapter Summary • The three modes of heat transfer are expressed as: Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -25 Chapter Summary • Various forms of work are expressed as follows: Çengel Boles Thermodynamics › Electrical work: (k. J) › Boundary work: (k. J) › Gravitational work (=DPE): (k. J) › Acceleration work (=DKE): (k. J) › Shaft work: (k. J) › Spring work: (k. J) Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -26 Çengel Boles Chapter Summary Thermodynamics • For the ploytropic process (Pvn = constant) of real gases, the boundary work can be expressed as: Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -27 Çengel Boles Chapter Summary • The energy balance for any system undergoing any process can be expressed as: Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -28 Çengel Boles Chapter Summary • The energy balances for any system undergoing any process can be expressed in the rate form as: Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -29 Çengel Boles Chapter Summary Thermodynamics • Taking heat transfer to the system and work done by the system to be positive quantities, the energy balance for a closed system can also be expressed as: where: Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -30 Çengel Boles Chapter Summary • For a constant-pressure process, Thus Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998 .

3 -31 Çengel Boles Chapter Summary Thermodynamics • The amount of energy needed to raise the temperature of a unit of mass of a substance by one degree is called the specific heat at constant volume Cv for a constant-volume process and the specific heat at constant pressure Cp for a constant pressure process. They are defined as: Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -32 Çengel Boles Chapter Summary Thermodynamics • For ideal gases u, h, Cv, and Cp are functions of temperature alone. The u and h of ideal gases can be expressed as: Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -33 Chapter Summary • For ideal gases Cv, and Cp are related by: Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -34 Chapter Summary • The specific heat ratio k is defined as: Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -35 Çengel Boles Chapter Summary Thermodynamics • For incompressible substances (liquids and solids), both the constant-pressure and constantvolume specific heats are identical and denoted by C: Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -36 Çengel Boles Chapter Summary • The u and given by h of incompressible substances are Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

3 -37 Chapter Summary Çengel Boles Thermodynamics • The refrigeration and freezing of foods is a major application area of thermodynamics. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
- Slides: 38