3 5 Types of Elements and the Groups
















- Slides: 18

3. 5 Types of Elements and the Groups of the Periodic Table Chemistry Objectives 4: d; 5: b-d

Essential Questions l What are the types of elements and how can you tell what type an element is? l What are the different groups on the periodic table and what properties do they have?

Types of Elements l What are three types of elements? l Metals, nonmetals, and metalloids/semimetals

Metals l Metals are to the left of the stairs and make up 80% of all elements l Metals have the following properties: ¡Good conductors of heat and electric current. ¡Lustrous - shiny ¡Ductile - can be made into wires ¡Malleable – can be made into sheets ¡Many react with acids

Nonmetals l Nonmetals are found to the right of the stairs l Nonmetals have the opposite properties of metals ¡Most nonmetals are gases at room temperature but a few are solids and one is a liquid ¡Dull ¡Brittle (shatter when hit) ¡Most are poor conductors and do not react with acid

Metalloids l Metalloids are along the stairs l Metalloids have properties of both metals and nonmetals which vary depending on the metalloid.

Trends l As you move across the periodic table the elements are becoming less metallic and more nonmetallic l Moving down the periodic table elements become more metallic and less nonmetallic l Elements near the staircase have a mixture of both types of characteristics

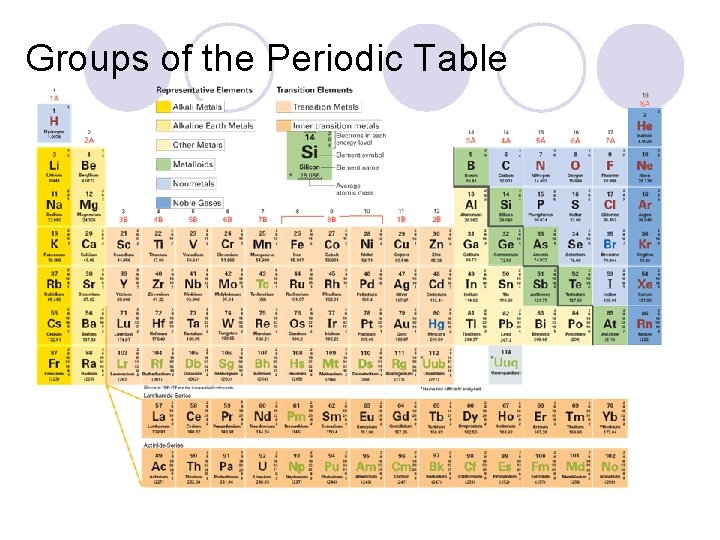
Groups of the Periodic Table

Groups of the Periodic Table l There are several main groups of the periodic table l They are the alkali metals, alkaline earth metals, transition metals, halogens, and noble gases l They have properties in common because of their same valence electrons

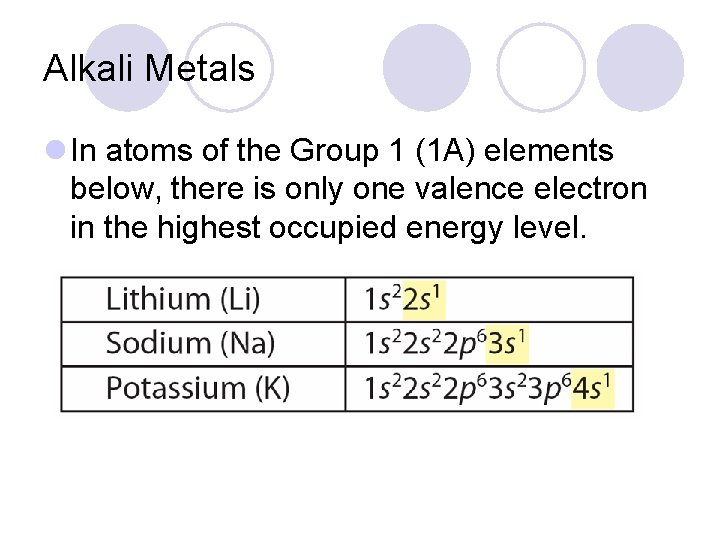
Alkali Metals l In atoms of the Group 1 (1 A) elements below, there is only one valence electron in the highest occupied energy level.

Alkali Metals l Alkali metals are soft, silver in color, shiny, and good conductors l Alkali metals are reactive and must be stored under oil l They also are highly reactive with water. l Reactivity increases as you go down the periodic table due to decreasing first ionization energy

Alkaline Metals l Alkaline metals are group 2 (2 A) l They have 2 valence electrons in their s orbital l They have the same basic properties as alkali metals, but are harder and less reactive

6. 2 Transition Elements l There are two types of transition elements —transition metals and inner transition metals. l Transition metals are in the d block l Inner transition metals are in the f block l They have the same properties as metals, but some properties can vary throughout the whole group

Halogens l Group 17 (7 A) are the halogens and have 7 valence electrons l All of the halogens are nonmetals and come in all 3 states l The halogens along with the alkali metals are the most reactive elements on the periodic table. l Reactivity decreases as you go down due to decreasing electron affinity

6. 2 Noble Gases l The noble gases are the elements in Group 18 (8 A) of the periodic table. They have full valence shells of 8 except for He which has a full shell of 2.

Noble Gases (cont) l The noble gases are called noble gases because they are inert ¡Inert means they do not react under normal conditions, but some can form compounds with F l They do not react because of the full electron shell l They are all gases and nonmetals

Essential Questions l What are the types of elements and how can you tell what type an element is? l What are the different groups on the periodic table and what properties do they have?

3. 5 Tracked Assignment l p 160 #4, 6 l p 167 #12, 14, 15 l p 181 -182 #28 -30, 32, 33, 48, 51, 54