3 5 NOTES Mixtures and Pure Substances III

3. 5 NOTES Mixtures and Pure Substances

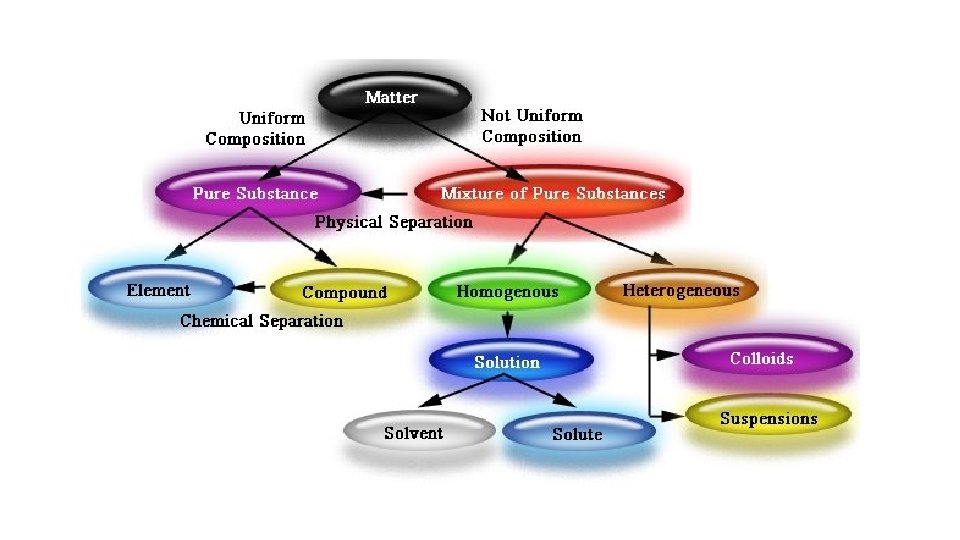
III. Mixtures of Matter • A. Mixtures • 1. Definition two or more substances that have been combined but have not been bonded (chemically) •

• 2. Types of mixtures • a. Heterogeneous non-uniform, can see the various parts, looks different; large particles • examples – mud puddle, Italian dressing; supreme pizza; salad • b. Homogeneous = solution uniform; appears the same throughout; particles are not visible • examples – Kool-Aid; salt water; formed when one substance dissolves in another; solute – what gets dissolved; solvent – does the dissolving; unique properties • c. Alloys, amalgams metal – metal solution • • brass (Cu + Zn) bronze (Cu + Sn) steel (Fe + Cr/Mn/others) amalgams – Hg based alloys; used to be found in dental work

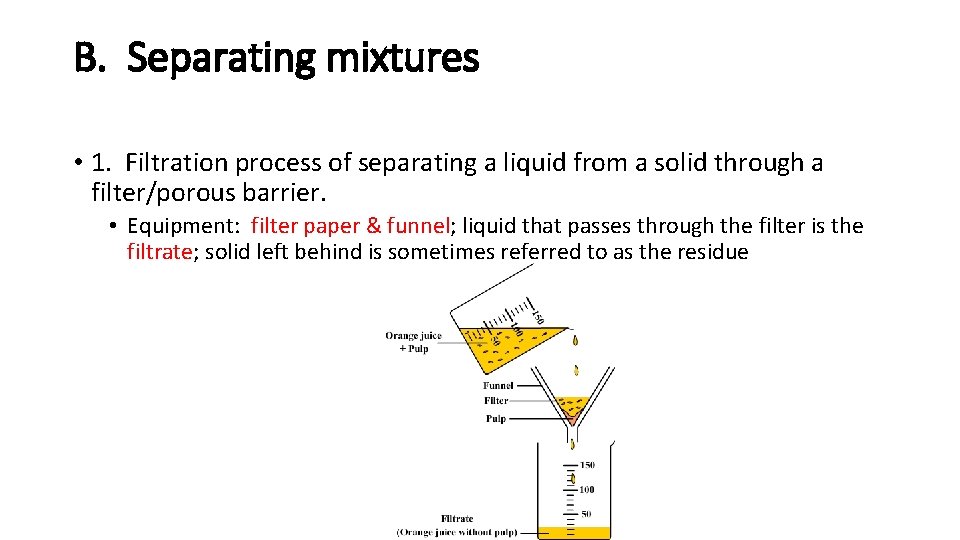
B. Separating mixtures • 1. Filtration process of separating a liquid from a solid through a filter/porous barrier. • Equipment: filter paper & funnel; liquid that passes through the filter is the filtrate; solid left behind is sometimes referred to as the residue
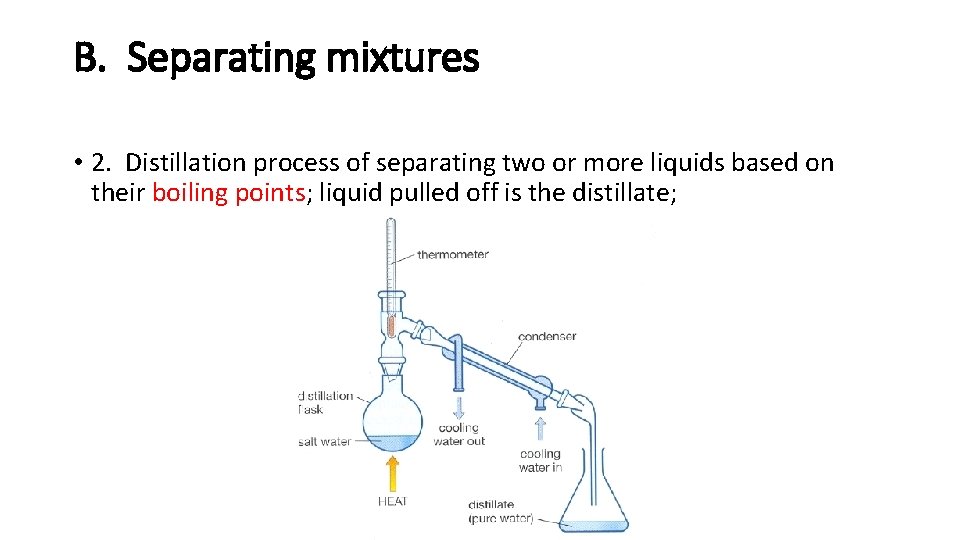
B. Separating mixtures • 2. Distillation process of separating two or more liquids based on their boiling points; liquid pulled off is the distillate;

B. Separating mixtures • 3. Crystallization process of separating a solute from the solvent; solvent is evaporated; solute will crystallize as the solvent is removed; example - rock candy.

B. Separating mixtures • 4. Chromatography separates the components of a mixture (mobile phase) based on the likelihood the components will travel through a medium (stationary phase)

• IV. Elements and Compounds • A. Elements substance composed of one type of atom; cannot be separated into smaller particles without losing its properties; 91 naturally occurring elements, remaining elements are synthetic; pure homogeneous substance;

• 1. Symbols two letters (1 st – capital, 2 nd – lower case); most are based on English name; • 10 are based on the Latin name • Lead – plumbum (Pb) • Mercury – hydrargyrum (Hg) • Sodium – natrium (Na) • Potassium – kalium (K) • Antimony – stibnium (Sb), • Copper – cuprum (Cu) • Iron – ferrum (Fe) • Silver – argentum (Ag) • Gold – aurum (Au) • Tin – Stannium (Sn)); • 1 is based on German name (Tungsten – wolfrum (W)); symbols/names are determined by IUPAC

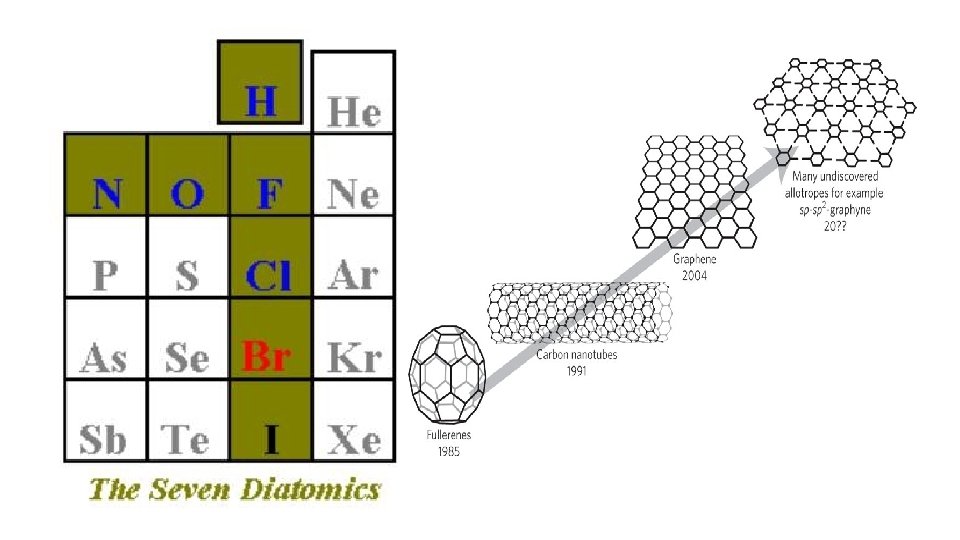
• 2. Atomic mass: average mass of compound (determined by numbers of protons and neutrons) • 3. Diatomic elements seven elements that bond to each for stability; will always be found together when they are in the elemental form; symbol written with a subscript 2; • H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2; Professor Br. INCl. HOF • • 4. Allotropes atoms of the same element found in the same state but with different structure; • carbon - graphite, diamond, buckminster fullerenes; • oxygen –molecular oxygen, ozone

• B. Compounds • 1. Definition substance composed of more than one type of atom; • homogeneous substances with a definite composition by mass; • when elements combine/bond together, new properties are seen; cannot be separated into smaller particles without losing its new properties, but can be separated back into atoms; written with formulas where the subscript describes how many of each type of atom is present.
- Slides: 14