3 5 3018 OBJ SWBAT determine the reaction

3 5 3018 OBJ: SWBAT determine the reaction mechanism of two reactions. 1. How do you determine the half life if you are provided a graph of a first order reaction?

Transition State Theory • Theory postulates that reactants form a high energy intermediate, the transition state, which then falls apart into the products. • For a reaction to occur, the reactants must acquire sufficient energy to form the transition state. – Activation energy or Ea • Mechanical analogy for activation energy 2

Transition State Theory Boulder Eactivation Epot=mgh 2 DEpot = mg. Dh h 2 Height Cross section of mountain Dh h 1 Epot=mgh 1 3
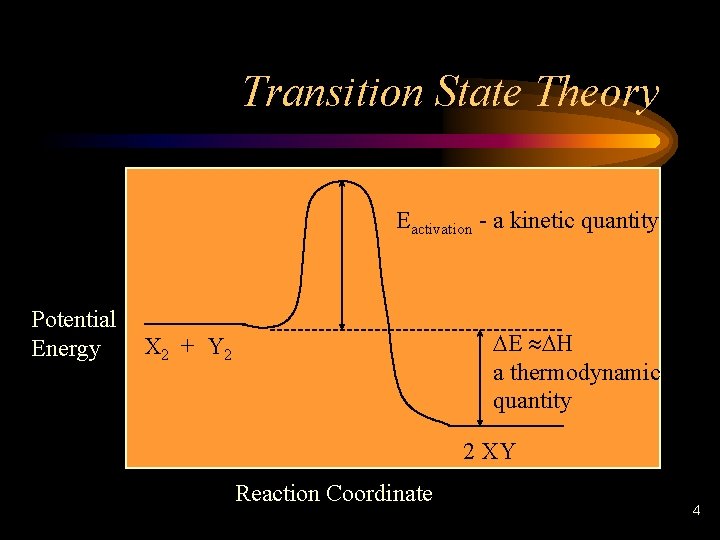
Transition State Theory Eactivation - a kinetic quantity Potential Energy DE » DH a thermodynamic quantity X 2 + Y 2 2 XY Reaction Coordinate 4

Transition State Theory • The relationship between the activation energy forward and reverse reactions is – Forward reaction = Ea – Reverse reaction = Ea + DE – difference = DE 5

Transition State Theory • The distribution of molecules possessing different energies at a given temperature may be represented as 6

Reaction Mechanisms & the Rate -Law Expression • Use experimental rate-law to postulate a mechanism. • The slowest step in a reaction mechanism is the rate determining step. • Elementary step: any process that occurs in a single step. • Elementary steps must add to give the balanced chemical equation. • Intermediate: a species which appears in an elementary step which is not a reactant or product. 7

Reaction Mechanisms & the Rate -Law Expression • Molecularity: the number of molecules present in an elementary step. – Unimolecular: one molecule in the elementary step, – Bimolecular: two molecules in the elementary step, and – Termolecular: three molecules in the elementary step. • It is not common to see termolecular processes (statistically improbable). 8
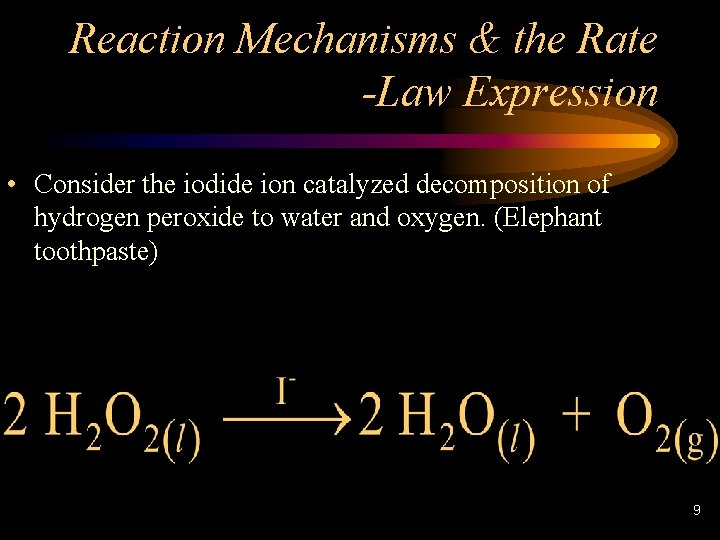
Reaction Mechanisms & the Rate -Law Expression • Consider the iodide ion catalyzed decomposition of hydrogen peroxide to water and oxygen. (Elephant toothpaste) 9

Reaction Mechanisms & the Rate -Law Expression • Reaction is known to be first order in H 2 O 2 , first order in I- , and second order overall. • Mechanism is thought to be: 10

Reaction Mechanisms & the Rate -Law Expression • Important notes: vone hydrogen peroxide molecule and one iodide ion are involved in the rate determining step vthe iodide ion catalyst is consumed in step 1 and produced in step 2 in equal amounts vhypoiodite ion has been detected in reaction mixture as a short-lived reaction intermediate 11

Reaction Mechanisms & the Rate -Law Expression • Ozone, O 3, reacts very rapidly with nitrogen oxide, NO, in a reaction that is first order in each reactant and second order overall. 12

Reaction Mechanisms & the Rate -Law Expression • A possible mechanism is: 13

Reaction Mechanisms & the Rate -Law Expression • A mechanism that is inconsistent with the rate-law expression is: 14

Reaction Mechanisms & the Rate -Law Expression • Experimentally determined reaction orders indicate the number of molecules involved in: the slow step only or the slow step and the equilibrium steps preceding the slow step. 15
- Slides: 15