3 3 NOTES Nuclear Radiation III Nuclear Reactions


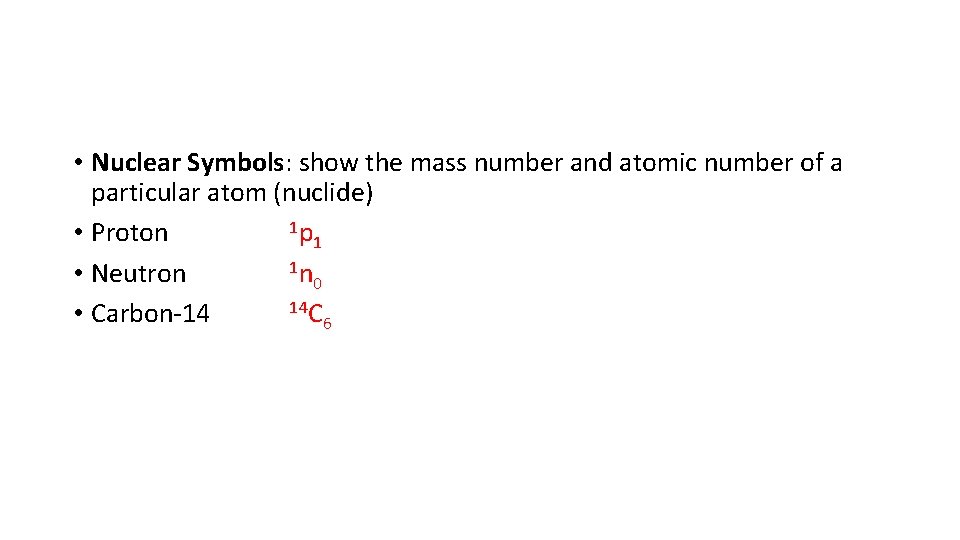





- Slides: 13

3. 3 NOTES Nuclear Radiation

III. Nuclear Reactions • Radioactivity: any process that involves particles or energy as a result of nuclear instability; often involves transmutation (the change of elements) • Fission: splitting of atoms • Fusion: joining of atoms •
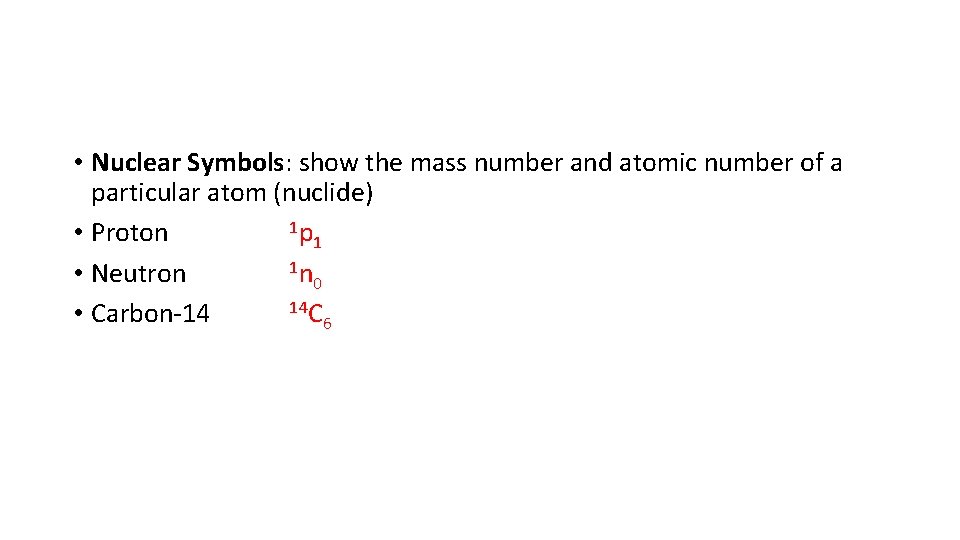
• Nuclear Symbols: show the mass number and atomic number of a particular atom (nuclide) 1 p • Proton 1 1 n • Neutron 0 14 C • Carbon-14 6

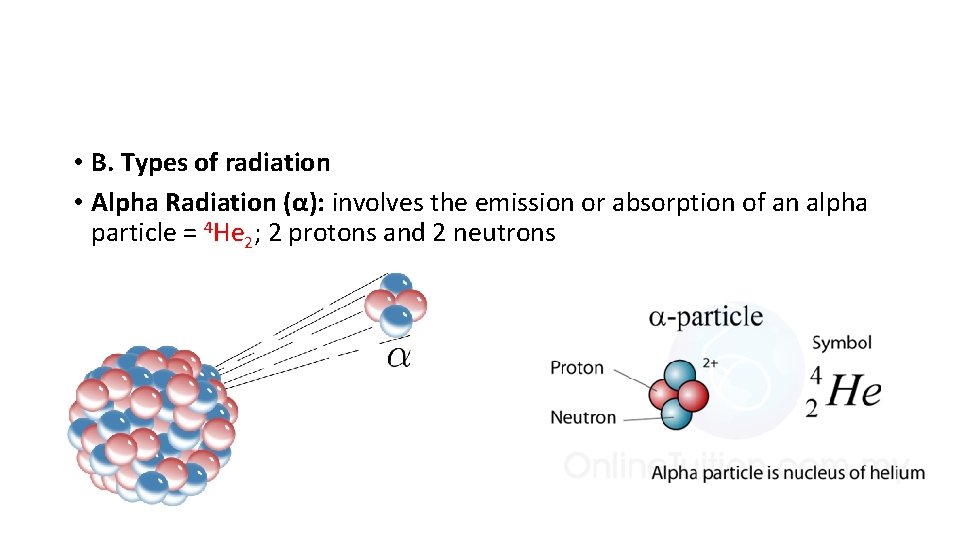
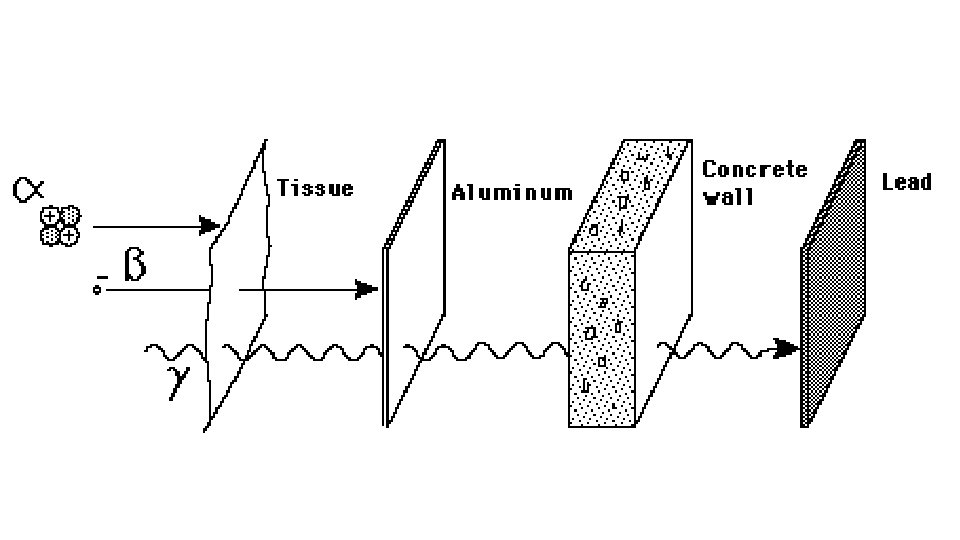
• B. Types of radiation • Alpha Radiation (α): involves the emission or absorption of an alpha particle = 4 He 2; 2 protons and 2 neutrons

• Sample Problems: Ra-226 decays by alpha emission • • 226 Ra 4 ? ? Xx ans: 222 Rn 86 88 He 2 + ? ? • top: 226 = 4 + x • bottom: 88 = 2 + x

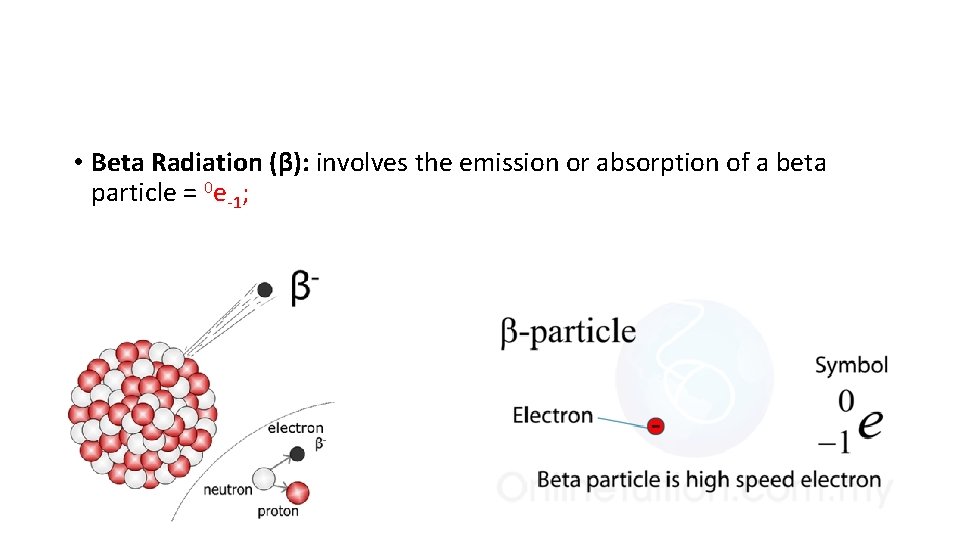
• Beta Radiation (β): involves the emission or absorption of a beta particle = 0 e-1;

• Example: C-14 decays be beta emission • 14 C 0 e + 14 N 6 -1 7

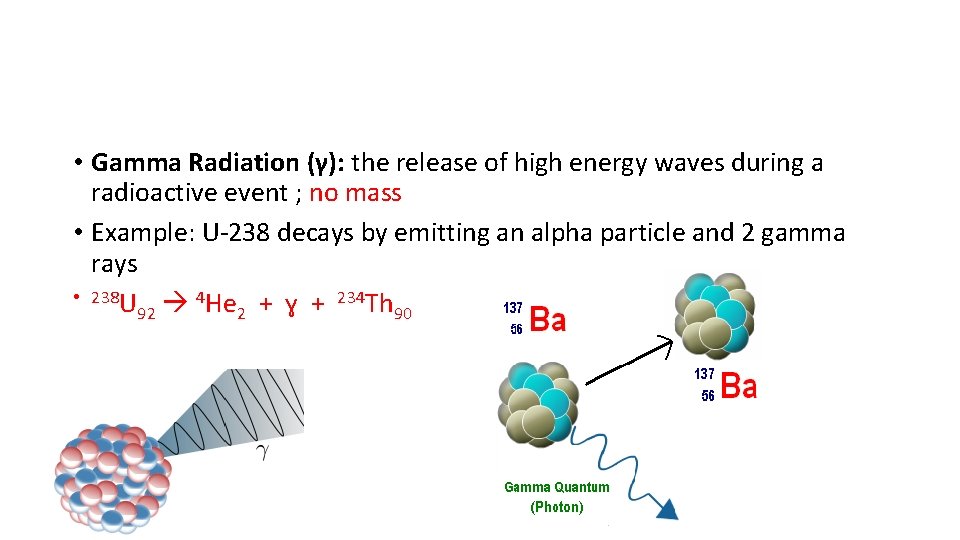
• Gamma Radiation (γ): the release of high energy waves during a radioactive event ; no mass • Example: U-238 decays by emitting an alpha particle and 2 gamma rays • 238 U 4 He + ɣ + 234 Th 92 2 90


• Induced Fission: Initiation of a fission chain reaction by a neutron striking a nucleus • Example: Plutonium-237 is struck by a neutron and induces fission into Palladium-130 and one other products. • 237 Pu 1 n 130 Pd 108 Cd + + 94 0 46 48

• Sample Problem: U-235 is bombarded with a neutron. A proton is produced as well as another element. What is the other element? Bombarded, collided, struck – all imply particles are on the left side of the arrow. • 235 U + 1 n 1 p + 235 Pa • ans: 92 0 1 91

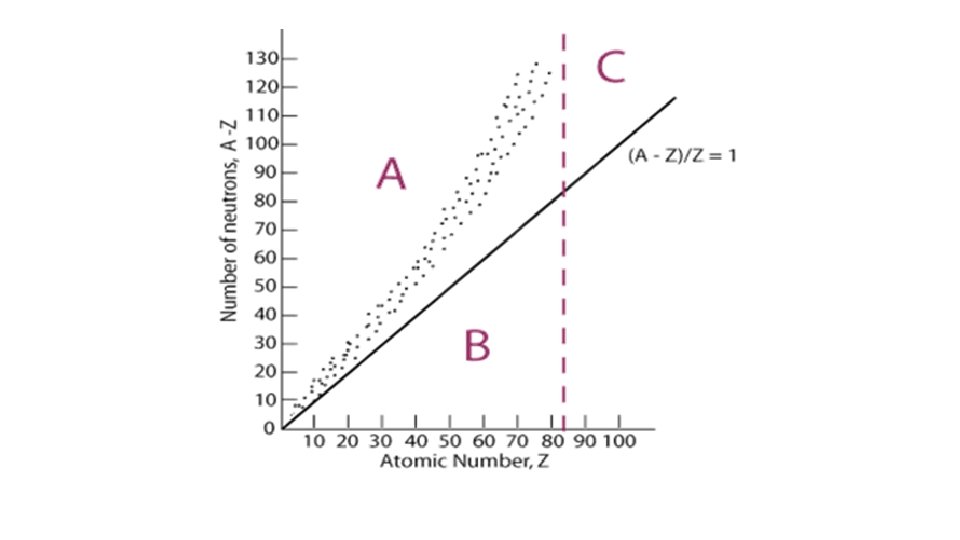
• What Causes Radioactivity? • Size of Nucleus: if a nucleus is too large, the element will always be radioactive, regardless of its isotope. • • • Proton/Neutron Ratio: Atoms need close to a correct proton: neutron ratio • Band of Stability: In general, atoms need a 1: 1 ratio, but as atomic number increases, more neutrons are needed to stabilize the atom. • If an atom strays too far from the desired ratio, that isotope may be radioactive.
