3 2 The Nature of Light Visible light
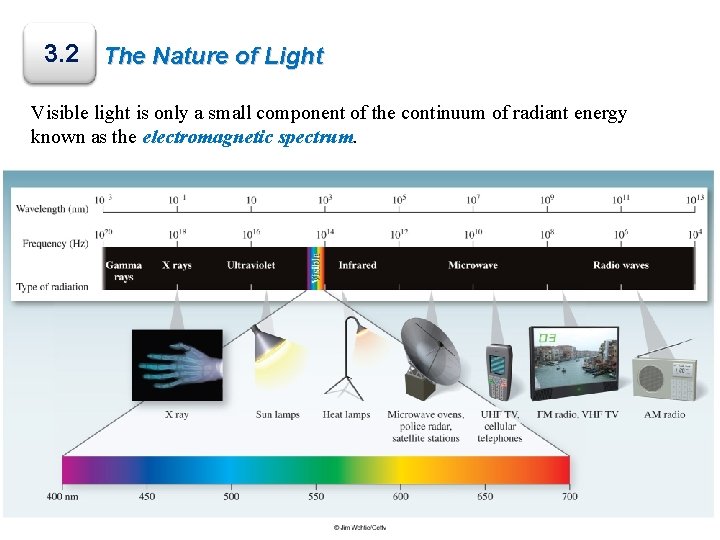
3. 2 The Nature of Light Visible light is only a small component of the continuum of radiant energy known as the electromagnetic spectrum.
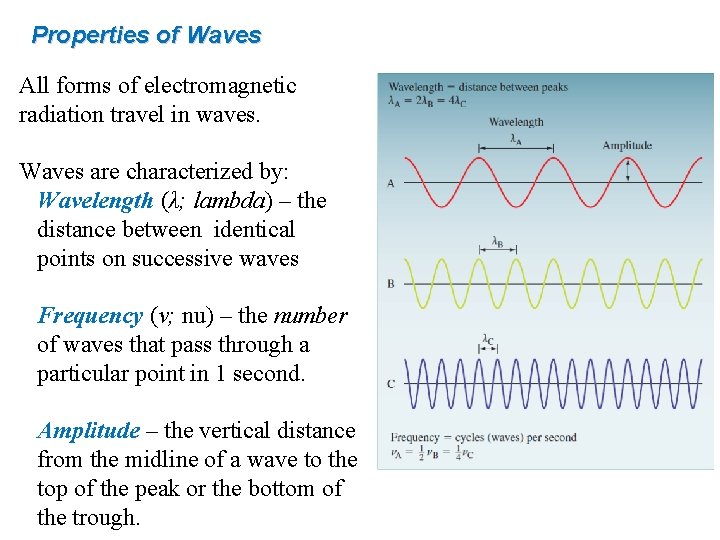
Properties of Waves All forms of electromagnetic radiation travel in waves. Waves are characterized by: Wavelength (λ; lambda) – the distance between identical points on successive waves Frequency (ν; nu) – the number of waves that pass through a particular point in 1 second. Amplitude – the vertical distance from the midline of a wave to the top of the peak or the bottom of the trough.

The Nature of Light The speed of light (c) through a vacuum is a constant: c = 2. 99792458× 108 m/s Normally rounded to, c = 3. 00× 108 m/s. Speed of light, frequency and wavelength are related: Ø λ is expressed in meters Ø v is expressed in reciprocal seconds (s− 1) Ø s-1 is also known as hertz (Hz)

The Electromagnetic Spectrum An electromagnetic wave has both an electric field component and a magnetic component. The electric and magnetic components have the same frequency and wavelength.
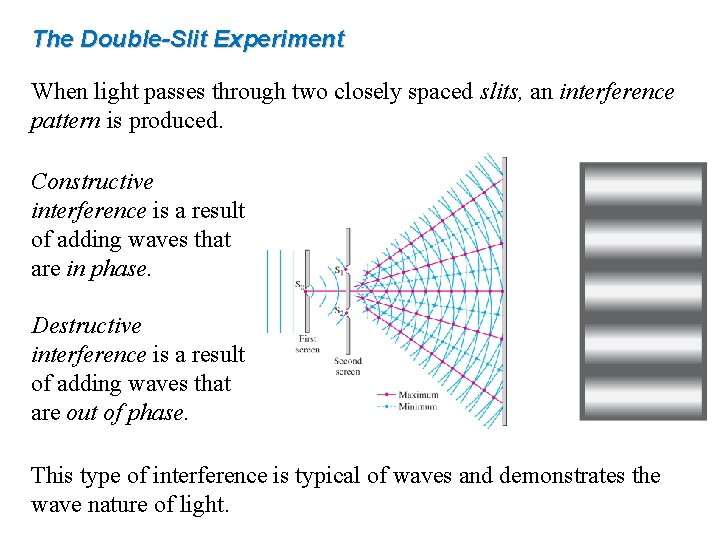
The Double-Slit Experiment When light passes through two closely spaced slits, an interference pattern is produced. Constructive interference is a result of adding waves that are in phase. Destructive interference is a result of adding waves that are out of phase. This type of interference is typical of waves and demonstrates the wave nature of light.

3. 3 Quantum Theory When a solid is heated, it emits electromagnetic radiation, known as blackbody radiation, over a wide range of wavelengths. The amount of energy given off at a certain temperature depends on the wavelength. Due to its wave properties, Max Planck suggested that radiant energy is only emitted or absorbed in discrete quantities, like small packages or bundles. A quantum of energy is the smallest quantity of energy that can be emitted (or absorbed).

Quantum Theory The energy E of a single quantum of energy is Ø h is called Planck’s constant: 6. 63× 10− 34 J∙s The idea that energy is quantized rather than continuous is like walking up a staircase or playing the piano Ø You cannot step or play anywhere (continuous), you can only step on a stair or play on a key (quantized). Activity: Should a wave with a short wavelength have a high or low energy? What if the speed of light were not constant, would the relationship between wavelength and energy still be the same?

Photons and the Photoelectric Effect Albert Einstein used Planck’s theory to explain the photoelectric effect. Electrons are ejected from the surface of a metal exposed to light of a certain minimum frequency, called the threshold frequency. The number of electrons ejected is proportional to the intensity. Below the threshold frequency no electrons were ejected, no matter how bright (or intense) the light.

Photons and the Photoelectric Effect Einstein proposed that the beam of light is really a stream of particles. These particles of light are now called photons. Each photon (of the incident light) must posses the energy given by the equation: • https: //phet. colorado. edu/en/simulation/phot oelectric
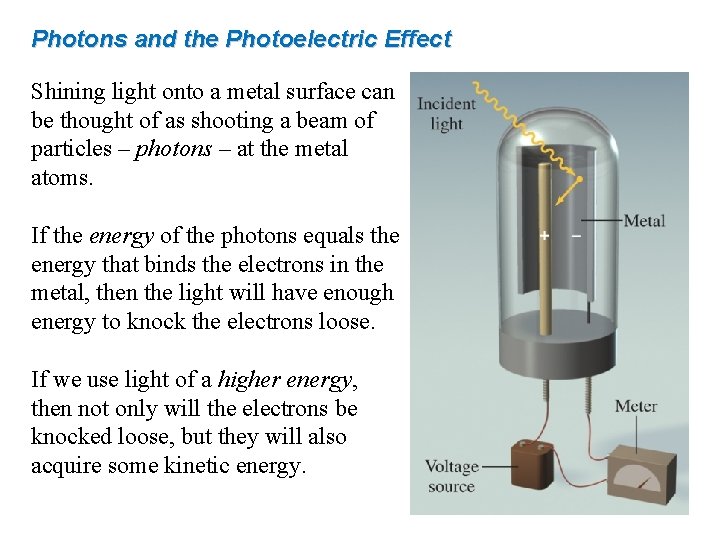
Photons and the Photoelectric Effect Shining light onto a metal surface can be thought of as shooting a beam of particles – photons – at the metal atoms. If the energy of the photons equals the energy that binds the electrons in the metal, then the light will have enough energy to knock the electrons loose. If we use light of a higher energy, then not only will the electrons be knocked loose, but they will also acquire some kinetic energy.

3. 4 Bohr’s Theory of the Hydrogen Atom Sunlight is composed of various color components that can be recombined to produce white light. The emission spectrum of a substance can be seen by energizing a sample of material with some form of energy. The “red hot” or “white hot” glow of an iron bar removed from a fire is the visible portion of its emission spectrum. The emission spectrum of both sunlight and a heated solid are approximately continuous (all wavelengths present).
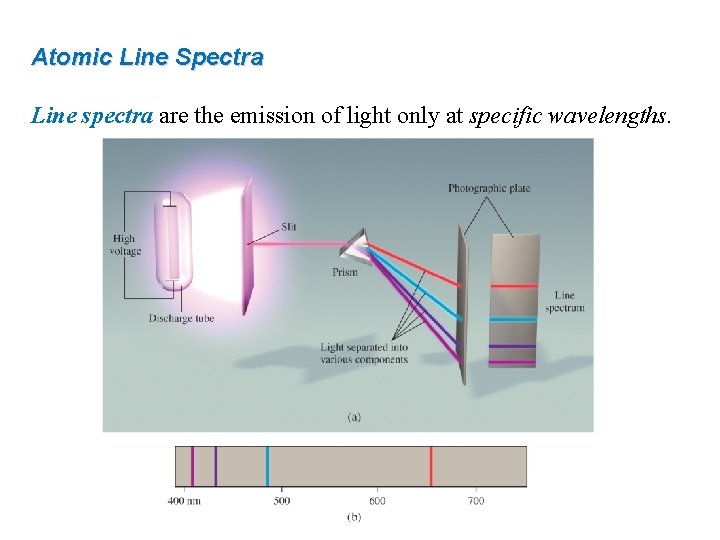
Atomic Line Spectra Line spectra are the emission of light only at specific wavelengths.
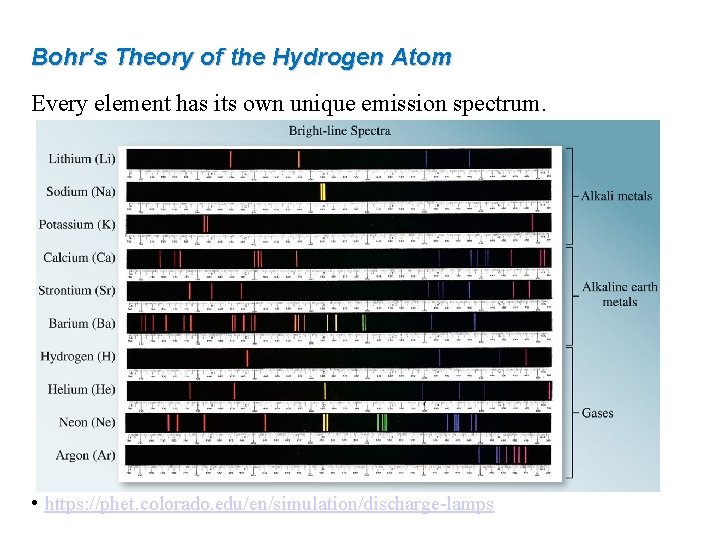
Bohr’s Theory of the Hydrogen Atom Every element has its own unique emission spectrum. • https: //phet. colorado. edu/en/simulation/discharge-lamps

The Line Spectrum of Hydrogen Neils Bohr attributed the emission of radiation by an energized hydrogen atom to the electron dropping from a higher-energy orbit to a lower one. As the electron dropped, it gave up a quantum of energy in the form of light. En is most negative when n = 1. ØCalled the ground state, the lowest energy state of the atom ØFor hydrogen, this is the most stable state The stability of the electron decreases as n increases. Each energy state in which n > 1 is called an excited state.

The Line Spectrum of Hydrogen Bohr’s theory explains the line spectrum of the hydrogen atom. Radiant energy absorbed by the atom causes the electron to move from the ground state (n = 1) to an excited state (n > 1). Conversely, radiant energy is emitted when the electron moves from a higher-energy state to a lower-energy excited state or the ground state. The quantized movement of the electron from one energy state to another is analogous to a ball moving and down steps.

Bohr’s Theory of the Hydrogen Atom nf is the final state ni is the initial state

3. 5 Wave Properties of Matter Louis de Broglie reasoned that if light can behave like a stream of particles (photons), then electrons could exhibit wavelike properties. • According to de Broglie, electrons behave like standing waves. • Only certain wavelengths are allowed. • At a node, the amplitude of the wave is zero. • https: //phet. colorado. edu/e n/simulation/wave-on-astring

3. 6 Quantum Mechanics Erwin Schrödinger derived a complex mathematical formula to incorporate the wave and particle characteristics of electrons. Wave behavior is described with the wave function ψ. The probability of finding an electron in a certain area of space is proportional to ψ2 and is called electron density.

Quantum Mechanics The Schrödinger equation specifies possible energy states an electron can occupy in a hydrogen atom. The energy states and wave functions are characterized by a set of quantum numbers. Instead of referring to orbits as in the Bohr model, quantum numbers and wave functions describe atomic orbitals. • https: //phet. colorado. edu/en/simulation/hydrog en-atom

Study Guide for sections 3. 2 -3. 6 DAY 5, Terms to know: Sections 3. 2 -3. 6 light, radiant energy, electromagnetic spectrum, wavelength, frequency, amplitude, hertz, constructive and destructive interference, quantum, photoelectric effect, photon, line spectra or emission spectra, ground state, excited state, node, wave function, electron density DAY 5, Specific outcomes and skills that may be tested on exam 1: Sections 3. 2 -3. 6 • Be able to describe the properties of light existing as both a wave and a particle • Given that the speed of light is constant, be able to describe and fully explain the either directly or inversely proportional relationships between energy, frequency, and wavelength • Be able to describe the photoelectric effect in detail including how the photoelectric effect resulted in information about the nature of electrons and atoms • Be able to explain how the existence of line spectra suggest that electron as in quantized energy levels • Be able to explain two reasons why different elements give different line spectra • Be able to describe the potential energy difference between various excited states and between excited states and the ground state for an atom and explain why • Be able to explain what a node is and how the existence of nodes suggests that electrons behave as waves in addition to behaving as particles in other experiments • Be able to describe what electron density and orbitals are and how they relate to electron probability

Extra Practice Problems for sections 3. 2 -3. 6 Complete these problems outside of class until you are confident you have learned the SKILLS in this section outlined on the study guide and we will review some of them next class period. 3. 19 3. 33 3. 39 VC 3. 1 VC 3. 4 3. 51 3. 121 3. 143

Prep for day 6 Must watch videos: https: //www. youtube. com/watch? v=accy. CUzasa 0 (Bozeman: orbitals) http: //echem 1 a. cchem. berkeley. edu/modules/module-3/ (UC-Berkeley watch lesson 7) Other helpful videos: http: //ocw. mit. edu/courses/chemistry/5 -111 -principles-of-chemical-science-fall-2008/video-lectures/ (MIT lectures 6 and 7) http: //ps. uci. edu/content/chem-1 a-general-chemistry (UC-Irvine lectures 6) Read sections 3. 7 -3. 8
- Slides: 22