3 2 Introduction to the Atom The Nucleus













- Slides: 15

3. 2 Introduction to the Atom The Nucleus

Atomic History • The term atom coined by the Ancient Greeks. – Greeks had no evidence for the atom. It was more of a philosophical idea. • John Dalton – early 1800 s – witnessed elements reacting in “set ratios. ” Assumed there must be smaller parts combining in specific ways. – “Atoms are indivisible!”

Atomic History • JJ Thompson – 1897 – showed cathode rays had mass but very low amounts. Showed these particles were negatively charged and he believed them to be evenly spaced throughout the atom. Nobel Prize – 1906 • Ernest Rutherford – 1909 – bombarded gold foil with alpha (positive) particles to see if they would pass straight through. Some particles were deflected leading to the idea that their was a tiny positive nucleus in the atom.

History of the Atom • Niels Bohr – 1913 – proposed that electrons orbited the atom in fixed positions • Many others were involved in the development of atomic theory: Shrodinger, de Broglie, Heisenberg, Chadwick, Einstein, Max Planck


Earth’s Matter Everything in the universe is composed of matter. Matter: anything that has volume and mass. Mass: the quantity of matter an object has.

Elements and Atoms § Elements are pure substances that cannot be broken down into simpler forms of matter. § All matter is composed of elements. § Each element is made up of tiny particles called atoms. § Atom: the smallest part of an element that has all the properties of that element.

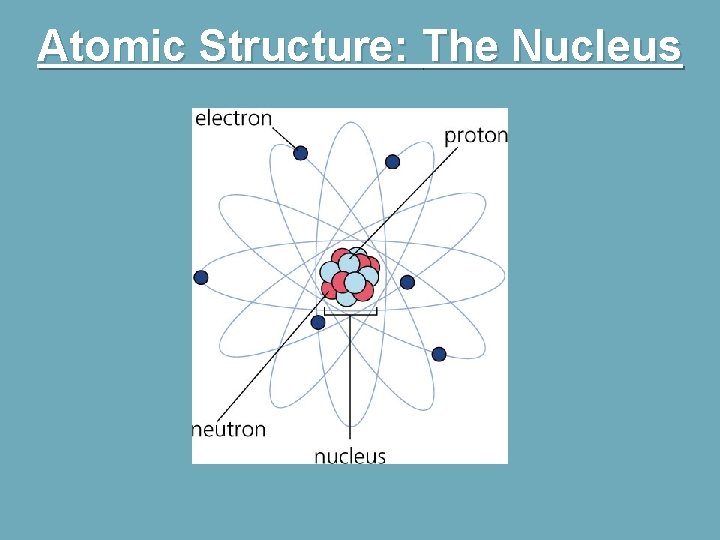
Atomic Structure: The Nucleus § The central core of the atom. § Consists of two kinds of particles: § Protons: have a positive electrical charge. § Neutrons: have a neutral charge. § Both have almost exactly equal masses of 1 amu (atomic mass unit). § Most of the mass in an atom is held in the nucleus.

Atomic Structure: The Nucleus

How do we know how many protons? BY USING THE PERIODIC TABLE!!!!! Atomic Number: Number of protons in an atom; usually appears directly above the chemical symbol.

How do we know how many neutrons? BY USING THE PERIODIC TABLE! Atomic Mass (Weight): Average masses of all the isotopes Mass (Weight) Number: Number of protons plus neutrons in an atom; usually appears directly below the chemical symbol.

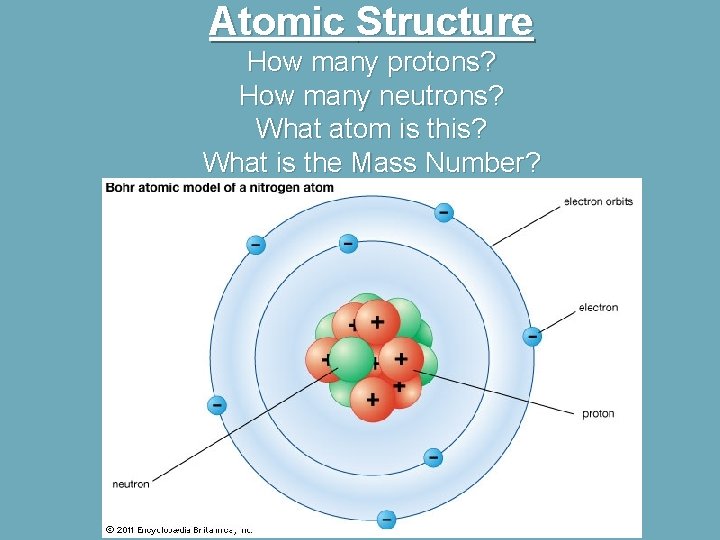
Atomic Structure How many protons? How many neutrons? What atom is this? What is the Mass Number?

Atom Equations Atomic Number = # of Protons Mass Number = Neutrons # of Neutrons = # of Protons = Neutrons # of Protons + # of Mass Number - # of Protons Mass Number - # of

Example • Copper – 65 – How many protons? – How many neutrons?

Isotopes: Have the same atomic # but different mass numbers because of a different number of neutrons.