3 1 Bonding Ionic Bonding Form between a


3. 1 Bonding
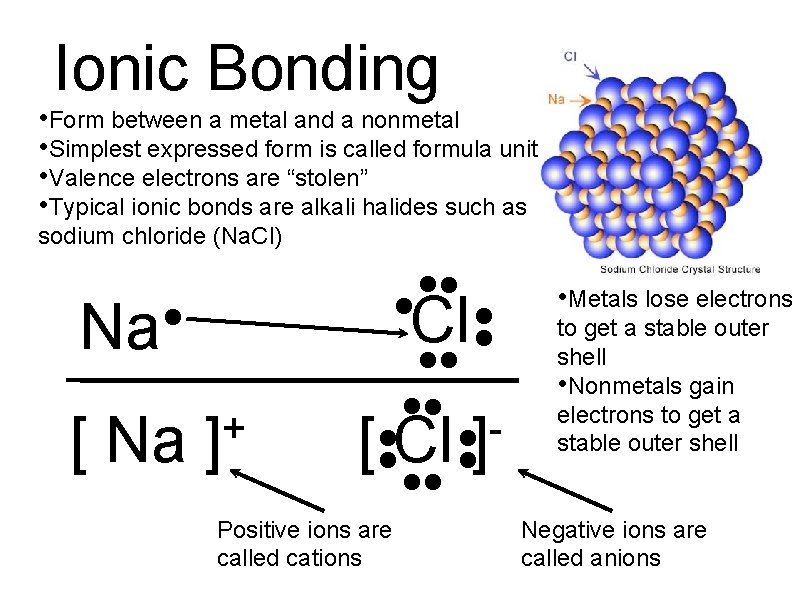
Ionic Bonding • Form between a metal and a nonmetal • Simplest expressed form is called formula unit • Valence electrons are “stolen” • Typical ionic bonds are alkali halides such as sodium chloride (Na. Cl) Na • [ Na + ] • • • Cl • • • • [ • • Cl • • ] • • Positive ions are called cations • Metals lose electrons to get a stable outer shell • Nonmetals gain electrons to get a stable outer shell Negative ions are called anions

Ionic Properties • Very high melting points. • Brittle crystals. • Many dissolve in water. • Good conductors of electricity if melted or dissolved. Acids • Ionic compounds that have H+ and an anion • Molecular formula always starts with H • Name is derived from its anion and always ends with the word, acid.
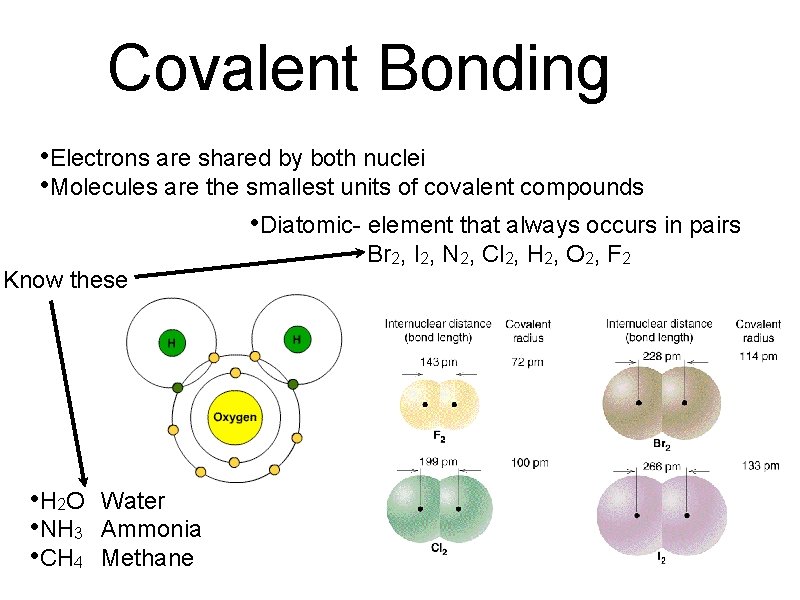
Covalent Bonding • Electrons are shared by both nuclei • Molecules are the smallest units of covalent compounds • Diatomic- element that always occurs in pairs Know these • H 2 O • NH 3 • CH 4 Water Ammonia Methane Br 2, I 2, N 2, Cl 2, H 2, O 2, F 2
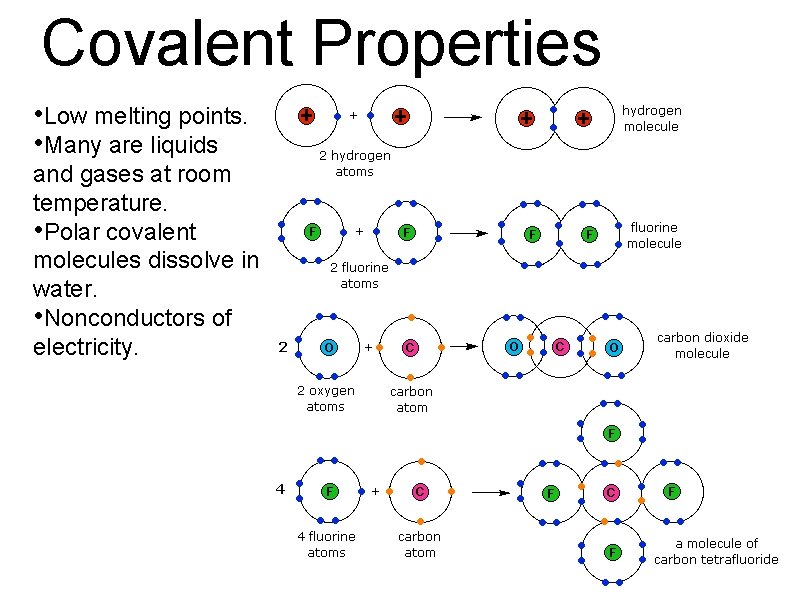
Covalent Properties • Low melting points. • Many are liquids and gases at room temperature. • Polar covalent molecules dissolve in water. • Nonconductors of electricity.
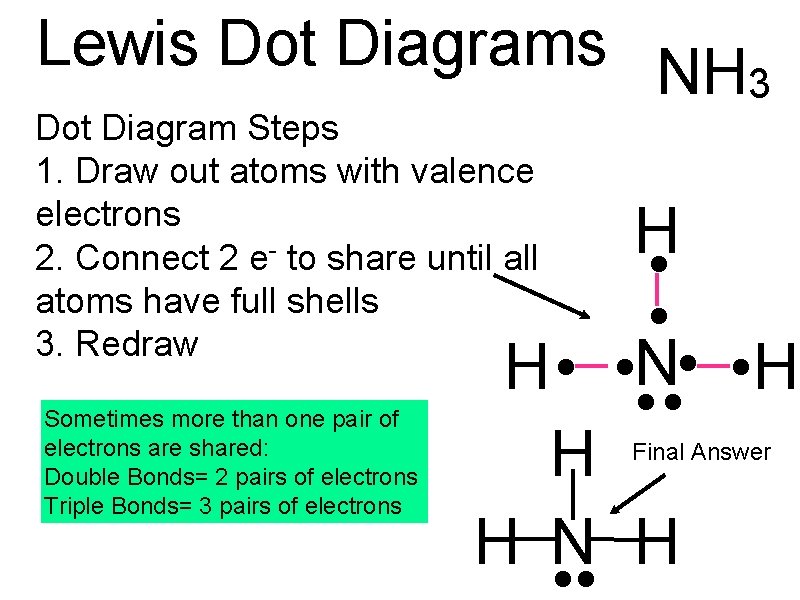
Lewis Dot Diagrams NH 3 Dot Diagram Steps 1. Draw out atoms with valence electrons 2. Connect 2 e- to share until all atoms have full shells 3. Redraw Sometimes more than one pair of electrons are shared: Double Bonds= 2 pairs of electrons Triple Bonds= 3 pairs of electrons H • • N • • H • • H H N H Final Answer
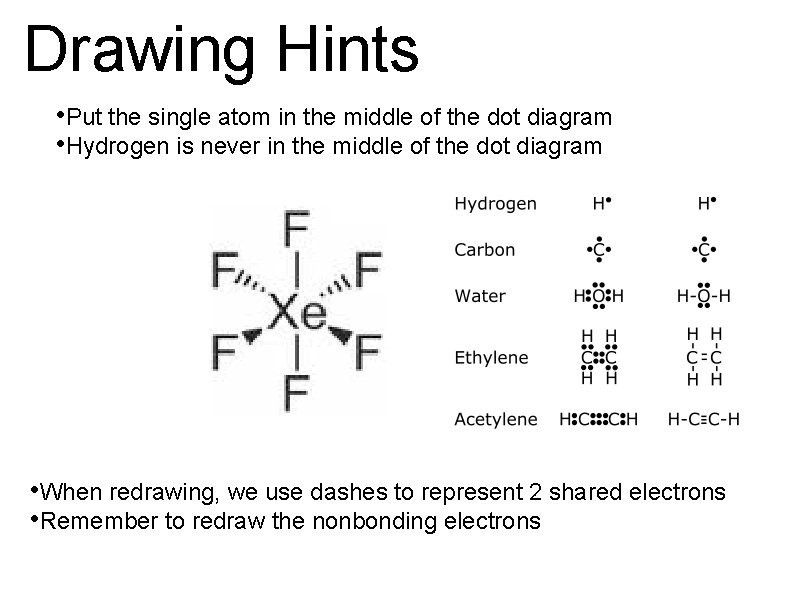
Drawing Hints • Put the single atom in the middle of the dot diagram • Hydrogen is never in the middle of the dot diagram • When redrawing, we use dashes to represent 2 shared electrons • Remember to redraw the nonbonding electrons

Sample Problems Draw • CH 4 • PCl 3 • SH 2 • HCl • CO 2 • BH 3
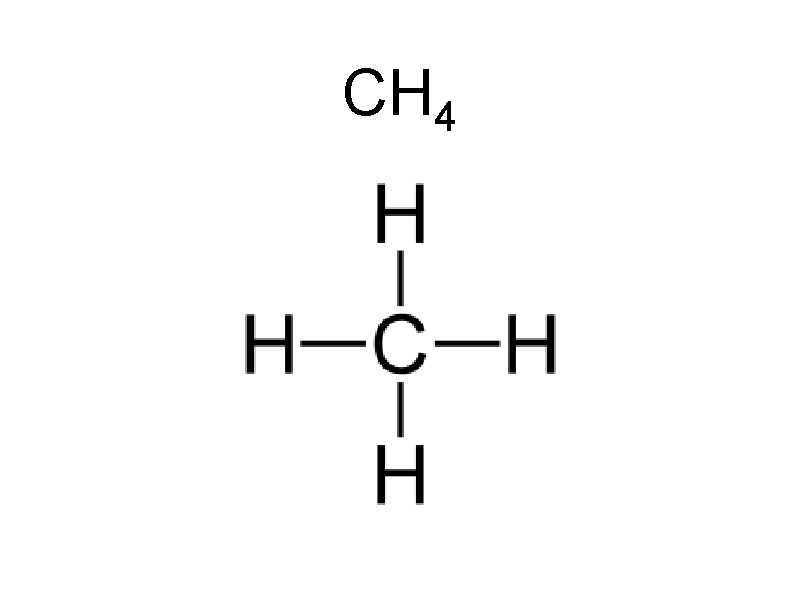
CH 4
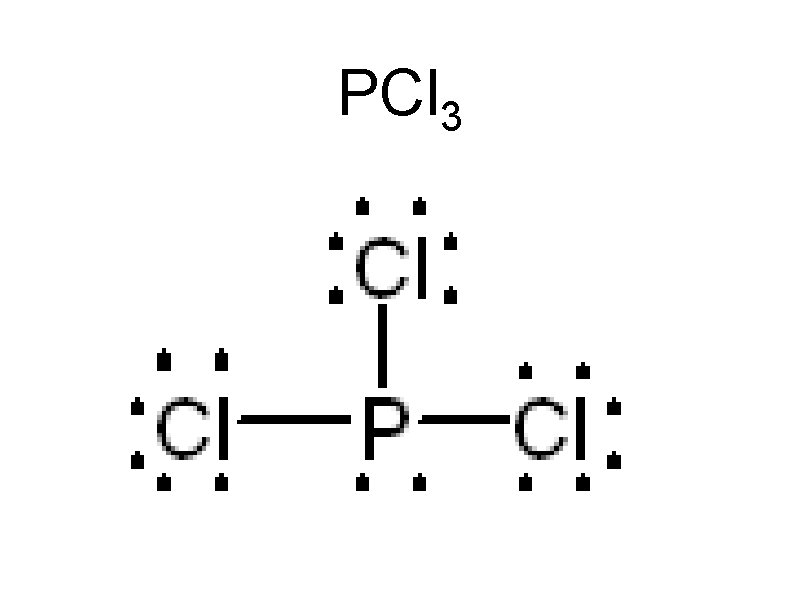
PCl 3
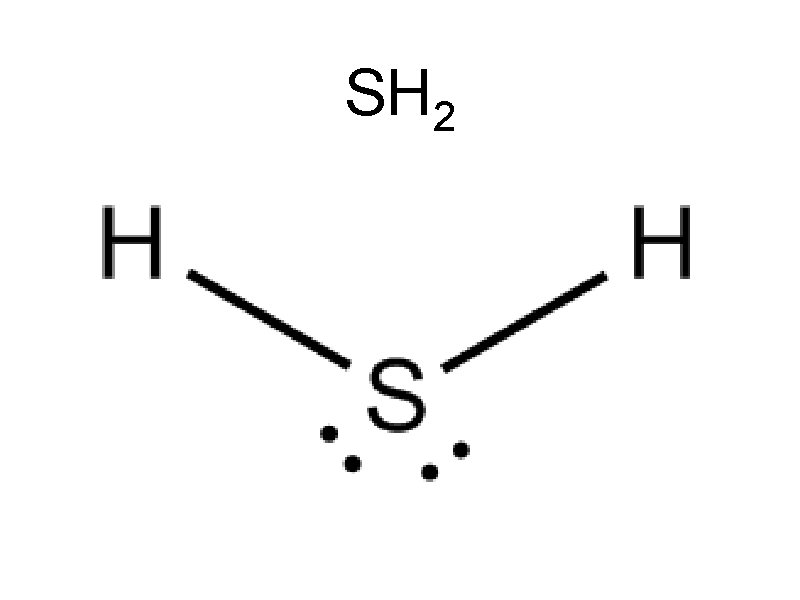
SH 2
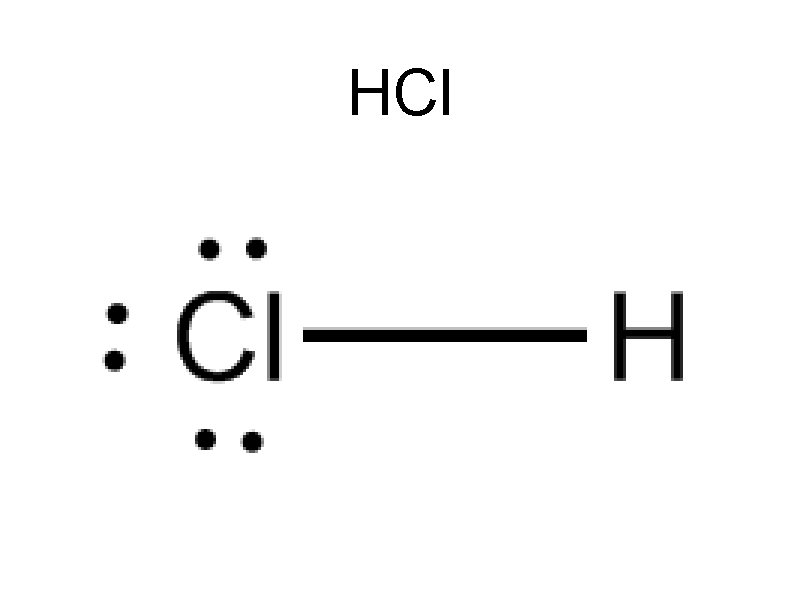
HCl
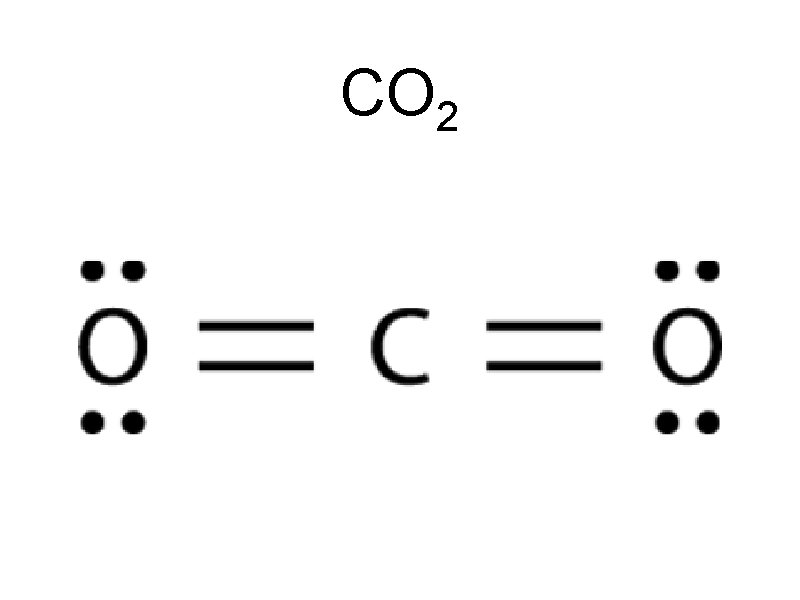
CO 2
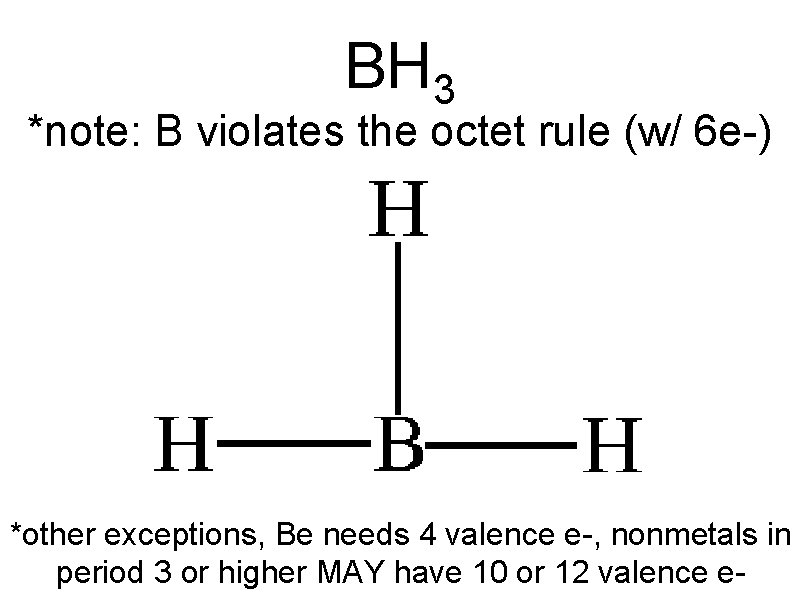
BH 3 *note: B violates the octet rule (w/ 6 e-) *other exceptions, Be needs 4 valence e-, nonmetals in period 3 or higher MAY have 10 or 12 valence e-
- Slides: 15