2617 Get ready for Bell Work Bell Work

2/6/17 Get ready for Bell Work.

Bell Work 2/6/17 1. 2.

2. When balancing equations, you can NEVER change the. . . a. Coefficient b. Subscript c. Whole number d. Atom

3.

4.

5.

6.

7.

Get ready to check Bell Work.


2. When balancing equations, you can NEVER change the. . . a. Coefficient b. Subscript c. Whole number d. Atom

3.

4.

5.

6.

7.

Success Criteria I can name the 4 types of reactions. TOC: #19. TYPES OF CHEMICAL REACTIONS NOTES

Objectives • Describe four types of chemical reactions. • Classify a chemical equation as one of four types of chemical reactions. 4 Types of Reactions: • Synthesis Reaction • Decomposition Reaction • Single-Displacement Reaction • Double-Displacement Reaction Video

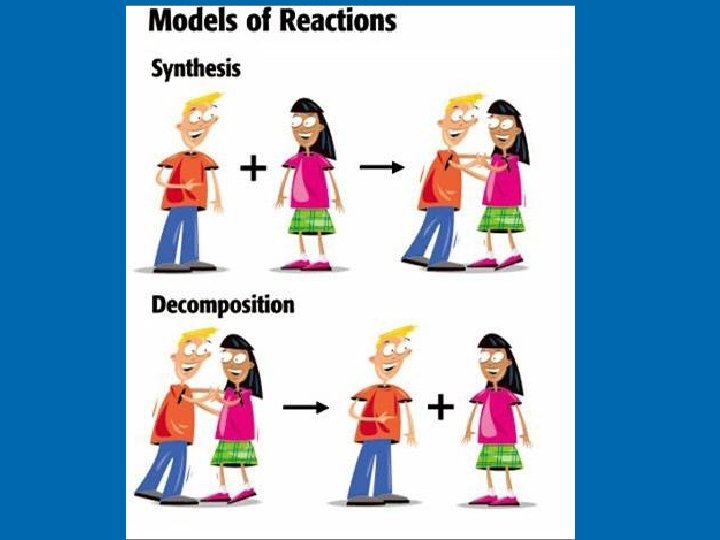
Synthesis Reactions: Synthesis reaction is a reaction in which two or more substances combine to form one new compound. Pattern: A + B AB Ex. N 2 + H 2 NH 3

Decomposition Reactions: A decomposition reaction is a reaction in which a single compound breaks down to form two or more simpler substances. Decomposition is the reverse of synthesis. Pattern: AB A + B Ex. Ag 2 O Ag + O 2

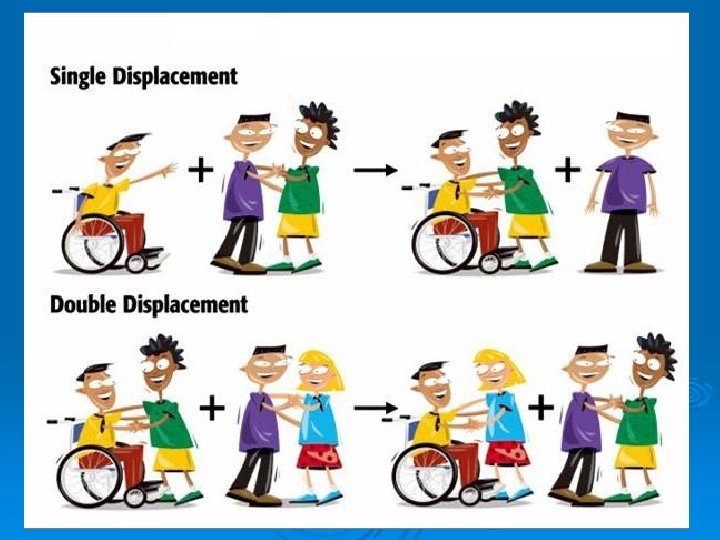
Single-Displacment Reactions: A single-displacement reaction is a reaction in which one element replaces another element that is part of a compound. The products of singledisplacement reactions are a new compound a different element. In a single-displacement reaction, a more reactive element can displace a less reactive element in a compound. Pattern: A + BX AX + B Ex. K + Mg. Br KBr + Mg

Double-Displacment Reactions: A double-displacement reaction is a reaction in which ions from two compounds exchange places. One of the products of this type of reaction is often a gas or a precipitate. Pattern: AX + BY AY + BX Ex. KNO 3 + H 2 CO 3 K 2 CO 3 + HNO 3

Brain Break! Ø Pair up with your same birthday month partner. Mirror your same month partner. Best idea gets candy!

Exit Ticket Ø Carbonic acid is an unstable compound. At room temperature, carbonic acid produces carbons dioxide and water according to the following equation: H 2 CO 3 CO 2 + H 2 O WHAT TYPE OF CHEMICAL REACTION IS THIS?
- Slides: 26