25 Global Ecology Chapter 25 Global Ecology CONCEPT

25 Global Ecology

Chapter 25 Global Ecology CONCEPT 25. 1 Elements move among geologic, atmospheric, oceanic, and biological pools at a global scale. CONCEPT 25. 2 Earth is warming due to anthropogenic emissions of greenhouse gases.

Chapter 25 Global Ecology CONCEPT 25. 3 Anthropogenic emissions of sulfur and nitrogen cause acid deposition, alter soil chemistry, and affect the health of ecosystems. CONCEPT 25. 4 Losses of ozone in the stratosphere and increases in ozone in the troposphere each pose risks to organisms.

Introduction Movements of biologically important elements are linked at a global scale that transcends ecological boundaries. Humans are increasingly changing the physical and chemical environment on a global scale.

Introduction Atmospheric emissions of pollutants, dust, and greenhouse gases have caused widespread environmental problems. A major focus of global ecology is the study of the environmental effects of human activities.

CONCEPT 25. 1 Elements move among geologic, atmospheric, oceanic, and biological pools at a global scale.

Concept 25. 1 Global Biogeochemical Cycles Global cycling of carbon (C), nitrogen (N), phosphorus (P), and sulfur (S) are emphasized because of their biological importance and their roles in human alteration of the global environment.

Concept 25. 1 Global Biogeochemical Cycles Pool, or reservoir: Amount of an element in a component of the biosphere. Flux: Rate of movement of an element between pools. Examples: Terrestrial plants are a pool of carbon; photosynthesis represents a flux.

Concept 25. 1 Global Biogeochemical Cycles The Carbon Cycle: C is critical for energy transfer and the construction of biomass. C is dynamic, moving between different pools over time scales of weeks to decades.

Concept 25. 1 Global Biogeochemical Cycles Changes in the global C cycle are influencing Earth’s climate. C in the atmosphere occurs primarily as carbon dioxide (CO 2) and methane (CH 4). Both are greenhouse gases that affect the global climate.
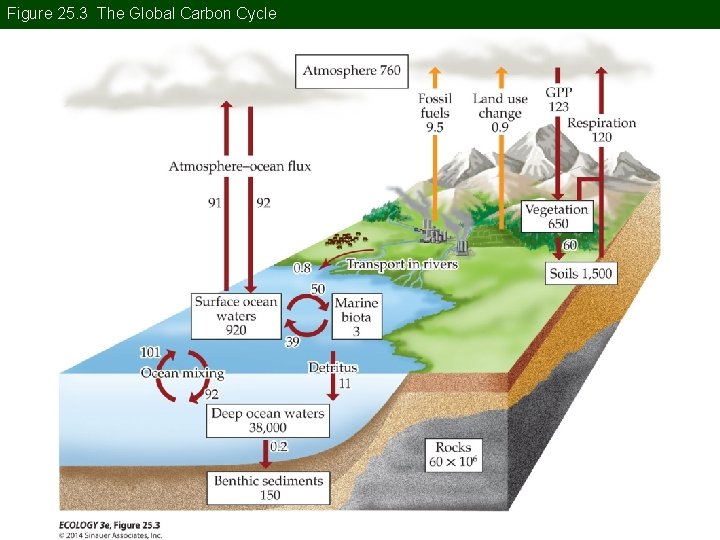
Figure 25. 3 The Global Carbon Cycle

Concept 25. 1 Global Biogeochemical Cycles Anthropogenic release of C to the atmosphere from the terrestrial pool results from land use change, mostly deforestation (20%); and from burning fossil fuels (80%). Before the mid-nineteenth century, deforestation was the main anthropogenic flux.

Concept 25. 1 Global Biogeochemical Cycles Removing the forest canopy warms the soil, increasing rates of decomposition and respiration. Burning trees releases CO 2, and small amounts of CO and CH 4. In the 20 th century, major deforestation shifted from the mid-latitudes to the tropics.

Concept 25. 1 Global Biogeochemical Cycles Anthropogenic emissions of CO 2 more than doubled from 1970 to 2011. About half is taken up by the oceans and terrestrial biota. But this proportion will decrease because terrestrial and ocean uptake will not keep pace with the rate of atmospheric increase.

Concept 25. 1 Global Biogeochemical Cycles Anthropogenic emissions of CH 4 have also increased. Atmospheric CH 4 levels are much lower than CO 2, but CH 4 is a more effective greenhouse gas.

Concept 25. 1 Global Biogeochemical Cycles Anthropogenic sources of CH 4 include: • Burning fossil fuels • Agricultural development (primarily rice grown in flooded fields) • Burning of forests and crops • Livestock production

Concept 25. 1 Global Biogeochemical Cycles Higher concentrations of CO 2 may stimulate photosynthesis. But experiments have shown that increased photosynthetic rates may be short lived, and plants will acclimate to higher concentrations. For forest trees, increased CO 2 uptake may be sustained longer.

Concept 25. 1 Global Biogeochemical Cycles Ocean acidity has increased 30% over the last century. Further increase is predicted by models. Many marine organisms form shells of carbonate. Increasing acidity will dissolve existing shells and lower carbonate concentrations will decrease the ability to synthesize new shells.

Concept 25. 1 Global Biogeochemical Cycles On Australia’s Great Barrier Reef, calcium carbonate formation declined by 14% from 1990 to 2009. Anthropogenic CO 2 emissions therefore have potential to tremendously alter the diversity and function of marine ecosystems.
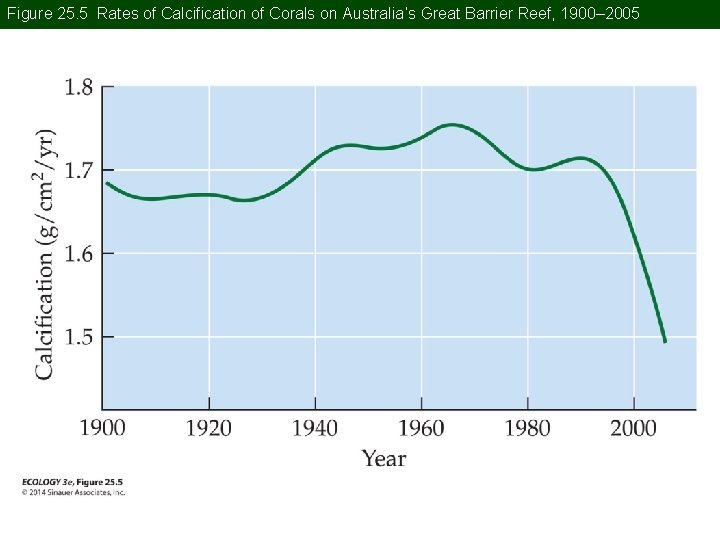
Figure 25. 5 Rates of Calcification of Corals on Australia’s Great Barrier Reef, 1900– 2005

Concept 25. 1 Global Biogeochemical Cycles Since the mid-19 th century, CO 2 concentrations have increased at a rate faster than at any other time in the past 400, 000 years. Even if CO 2 emissions are reduced dramatically, CO 2 levels will remain high due to a time lag in ocean uptake (decades to centuries).

Concept 25. 1 Global Biogeochemical Cycles The Nitrogen Cycle: N is a constituent of enzymes and proteins and often limits primary productivity. N and C cycles are tightly coupled through the processes of photosynthesis and decomposition.

Concept 25. 1 Global Biogeochemical Cycles The largest N pool is atmospheric N 2, which is not available to most organisms. N-fixing bacteria are able to convert it to a useable form. Fixed N compounds are called reactive— they can participate in chemical reactions.

Concept 25. 1 Global Biogeochemical Cycles Humans have altered the N cycle even more than the C cycle. Rate of N 2 fixation by humans now exceeds natural biological rates. Emissions of N from industrial and agricultural activities cause widespread environmental changes, including acid precipitation.
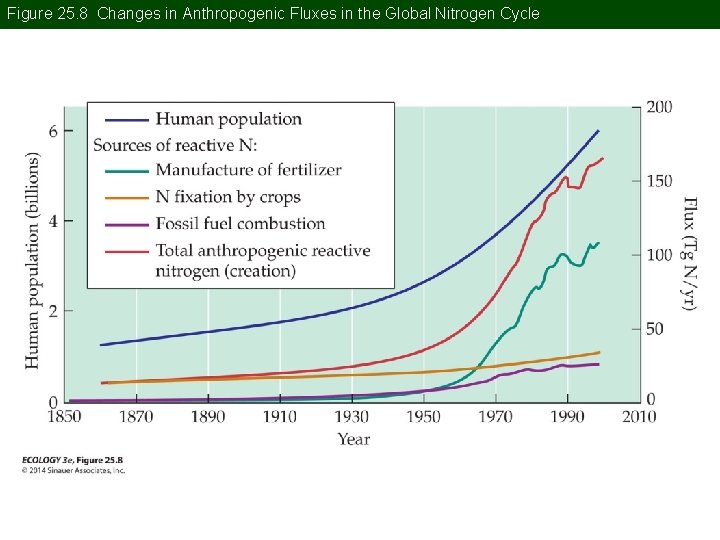
Figure 25. 8 Changes in Anthropogenic Fluxes in the Global Nitrogen Cycle

Concept 25. 1 Global Biogeochemical Cycles Fertilizers are made using the Haber– Bosch process. Growing N-fixing crops such as alfalfa, soybeans, and peas has increased biological N 2 fixation. Flooding of agricultural fields for rice has increased N 2 fixation by cyanobacteria.

Concept 25. 1 Global Biogeochemical Cycles Many other forms of reactive N are emitted to the atmosphere, mostly from fossil fuel combustion. These compounds can undergo chemical reactions in the atmosphere and are potentially available for biological uptake. They are returned to ecosystems by atmospheric deposition.

Concept 25. 1 Global Biogeochemical Cycles The Phosphorus Cycle: P can limit primary productivity in aquatic ecosystems and some terrestrial ecosystems. P availability can control the rate of Nfixation, which has a high metabolic demand for P.

Concept 25. 1 Global Biogeochemical Cycles The C, N, and P cycles are linked through photosynthesis and NPP, decomposition, and N 2 fixation. P has no atmospheric pool, except as dust. The largest pools are in soils and marine sediments.
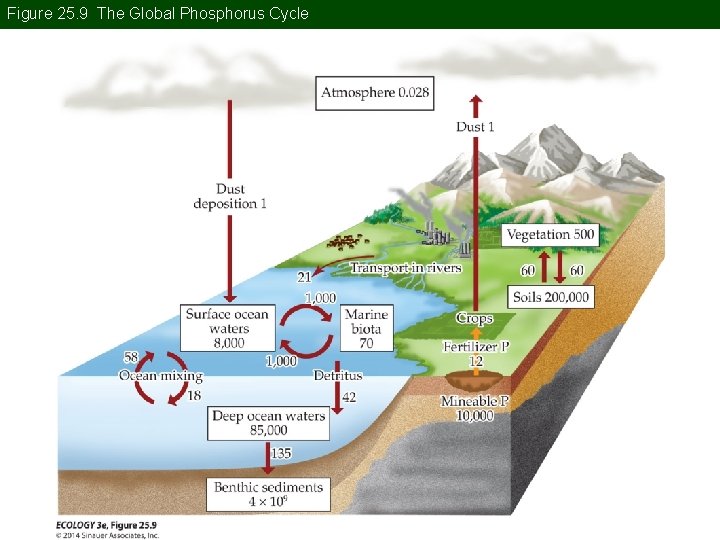
Figure 25. 9 The Global Phosphorus Cycle

Concept 25. 1 Global Biogeochemical Cycles P in aquatic systems is lost to the sediments. This is cycled again with tectonic uplift and weathering of rocks. Human influences on the P cycle include agricultural fertilizers, sewage and industrial wastes, and increased terrestrial erosion.

Concept 25. 1 Global Biogeochemical Cycles P fertilizers are made from marine sedimentary rock. Mining releases four times more P than natural rock weathering. Flux of anthropogenic P from terrestrial to aquatic ecosystems can have negative impacts such as eutrophication.

Concept 25. 1 Global Biogeochemical Cycles The Sulfur Cycle: S is a constituent of some amino acids, DNA, and RNA, but is probably never limiting to growth. Major pools of S are in rocks, sediments, and oceans as dissolved sulfate (SO 42–).
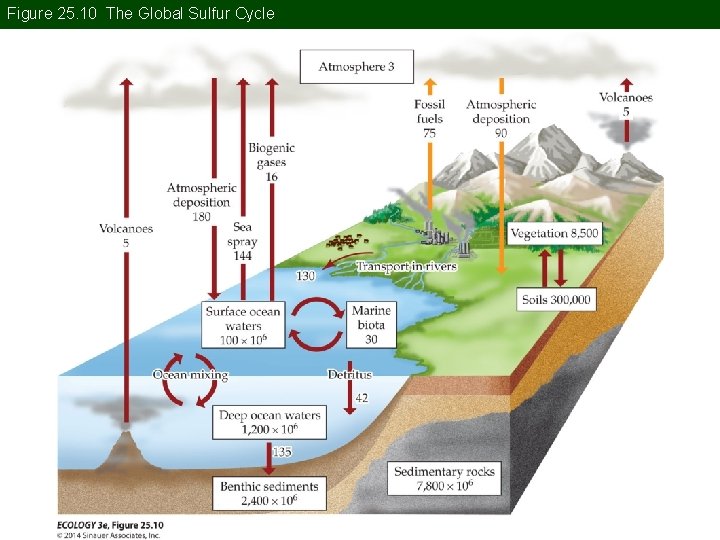
Figure 25. 10 The Global Sulfur Cycle

Concept 25. 1 Global Biogeochemical Cycles In the atmosphere, S compounds are transformed to SO 42– and H 2 SO 4 (sulfuric acid), which are removed quickly by precipitation. Anthropogenic emissions have quadrupled since the Industrial Revolution. Most comes from burning sulfur-containing coal and oil and from smelting.

CONCEPT 25. 2 Earth is warming due to anthropogenic emissions of greenhouse gases.

Concept 25. 2 Global Climate Change Climate change, especially change in frequency of extreme events (droughts, storms, etc. ) will have profound effects on ecosystems. Extreme events are often critical in determining species’ geographic ranges.

Concept 25. 2 Global Climate Change Weather: Current state of the atmosphere at any given time. Climate: Long term description of weather, including average conditions and the full range of variation. Climate variation occurs at multiple time scales—from daily and seasonal to decadal.

Concept 25. 2 Global Climate Change Climate change refers to directional change in climate over a period of at least three decades. Earth is currently experiencing significant climate change (IPCC 2013). Average global surface temperature increased 0. 8°C between 1880 and 2012.
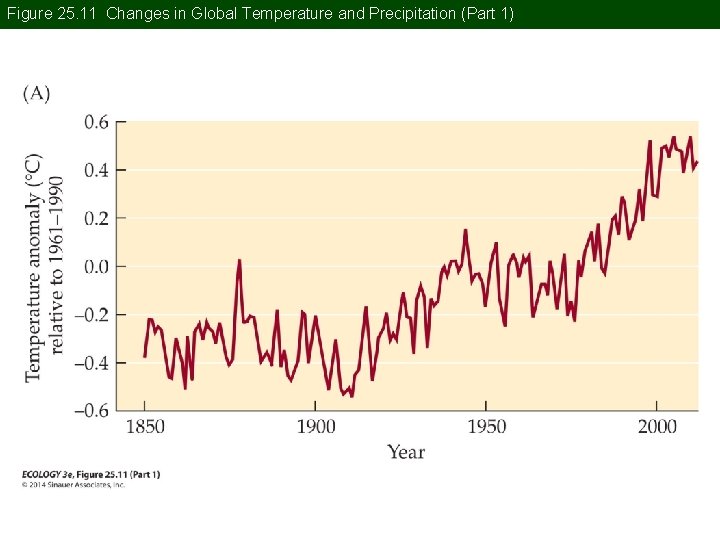
Figure 25. 11 Changes in Global Temperature and Precipitation (Part 1)

Concept 25. 2 Global Climate Change Associated with this warming, there has been: • Widespread retreat of mountain glaciers • Thinning of the polar ice caps • Melting permafrost • A 19 -cm rise in sea level since 1900

Concept 25. 2 Global Climate Change The warming trend has not been consistent around the globe. Some regions have seen greater warming, especially mid- to high latitudes in the Northern Hemisphere.
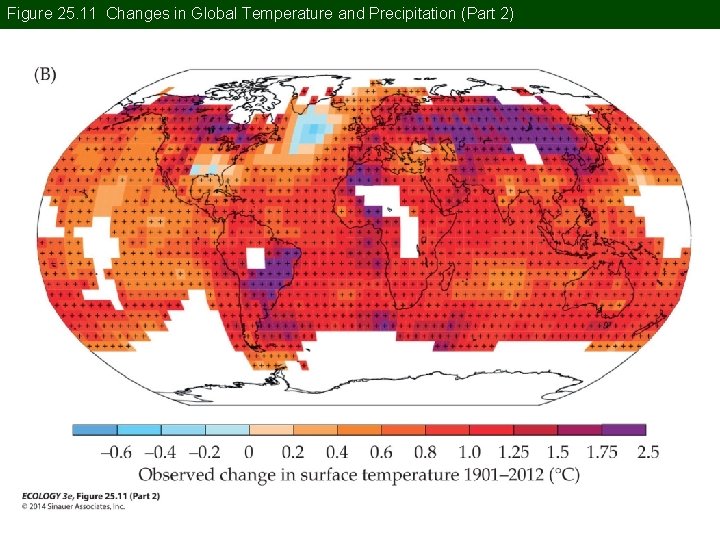
Figure 25. 11 Changes in Global Temperature and Precipitation (Part 2)

Concept 25. 2 Global Climate Change Precipitation in the high latitudes of the Northern Hemisphere has increased, but weather has been drier in the subtropics and tropics. There is also a trend of increasing frequency of extreme weather events such as hurricanes and heat waves.

Concept 25. 2 Global Climate Change Greenhouse effect: Warming of Earth by atmospheric absorption and reradiation of infrared radiation emitted by Earth’s surface. It is due to greenhouse gases in the atmosphere, primarily water vapor, CO 2, CH 4, and N 2 O.
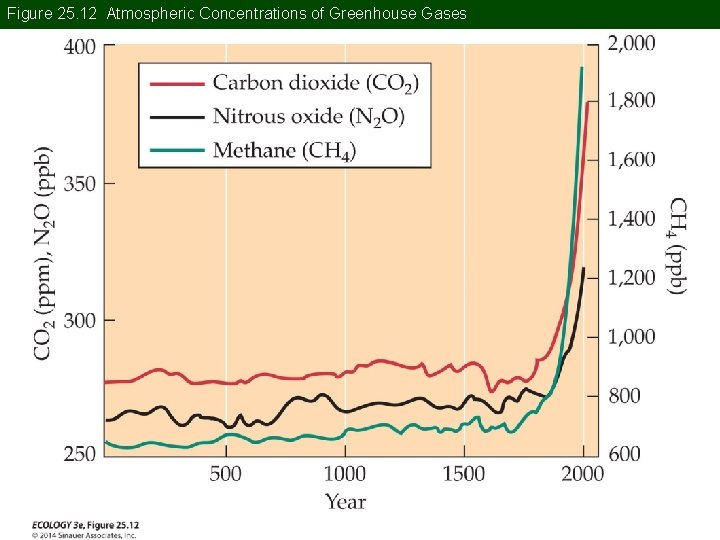
Figure 25. 12 Atmospheric Concentrations of Greenhouse Gases

Concept 25. 2 Global Climate Change The Intergovernmental Panel on Climate Change (IPCC) was established in 1988. It includes experts in atmospheric and climate science from around the world. They use modeling and analysis of data from the scientific literature to evaluate underlying causes of observed climate change and scenarios for the future.

Concept 25. 2 Global Climate Change The IPCC releases assessment reports to promote understanding of climate change among scientists, policymakers, and the general public. In recognition of their efforts to spread “knowledge about man-made climate change, ” the IPCC was awarded the Nobel Peace Prize in 2007.

Concept 25. 2 Global Climate Change In the third report (2001), the IPCC concluded that the majority of the observed global warming is attributable to human activities. While this conclusion is debated in the political arena, it is backed by the majority of the world’s leading atmospheric scientists.
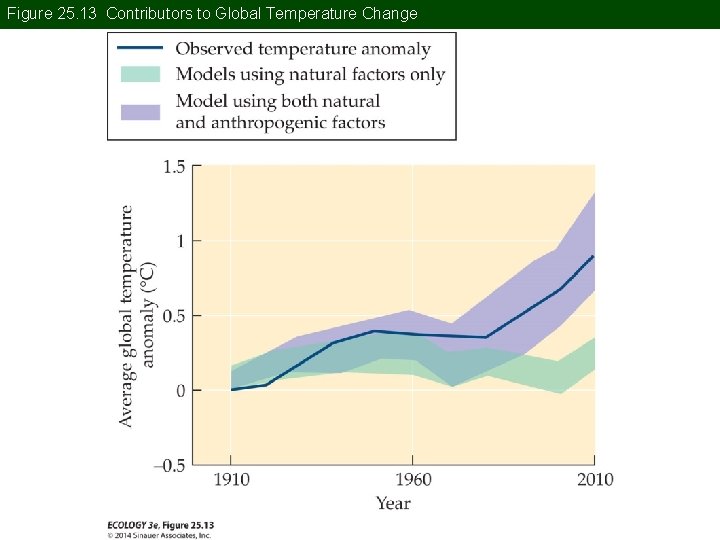
Figure 25. 13 Contributors to Global Temperature Change

Concept 25. 2 Global Climate Change The certainty of anthropogenic cause of climate change has increased with each new IPCC report. The 2013 report states “It is extremely likely (95%– 100% probability) that human influence has been the dominant cause of the observed warming since the mid-20 th century. ”

Concept 25. 2 Global Climate Change Paul Crutzen and Eugene Stoermer have suggested that we have entered a new geological period, called the Anthropocene epoch, to indicate the extensive impact of humans on our environment.

Concept 25. 2 Global Climate Change IPCC models predict an additional temperature increase of 1. 1 to 4. 8°C in the 21 st century. The range is associated with uncertainty of future greenhouse gas emissions and the behavior of Earth’s climate system.

Concept 25. 2 Global Climate Change What does a 1. 1° to 4. 8°C change in temperature mean for biological communities? It can be compared with elevational climate change on a mountain. The median value (2. 9°C) would correspond to a 500 m shift in elevation.

Concept 25. 2 Global Climate Change Because climate change will be rapid, most plants and animals will not be able to respond with evolutionary change. Dispersal may be the only way to avoid extinction. Dispersal barriers and habitat fragmentation will be important constraints.

Concept 25. 2 Global Climate Change Plant dispersal rates are generally much slower than the predicted rate of climate change. Ruderal (weedy) herbaceous plants and plants with animal-dispersed seeds can disperse and establish quickly. Shrubs and trees have much slower dispersal rates; there may be time lags in their response.

Concept 25. 2 Global Climate Change For animals, their habitat and food requirements are associated with specific vegetation types. Barriers to dispersal can prevent migration of many kinds of organisms— dams, habitat fragmentation, etc.

Concept 25. 2 Global Climate Change Organisms have already begun to respond to climate change (e. g. , earlier migration of birds, local extinction of amphibian and reptile populations, earlier leaf-out of vegetation). Geographic ranges of many species have shifted.

Concept 25. 2 Global Climate Change Ranges of plant species in the European Alps were compared with historical records (Grabherr et al. 1994). A consistent trend of upward movement of species from lower elevations onto the summits was reported.
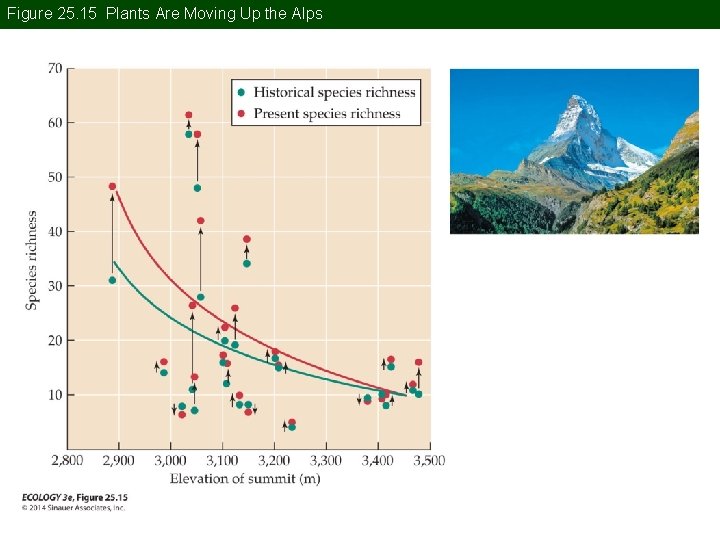
Figure 25. 15 Plants Are Moving Up the Alps

Concept 25. 2 Global Climate Change Extinction of lizard populations in Mexico has been linked to warmer spring temperatures, which limits foraging time during the breeding season (Sinervo et al. 2010). Using models of lizard physiology and projections for climate warming, they predict 39% of lizard populations will go extinct by 2050.

Concept 25. 2 Global Climate Change Migratory animals may be affected: • Fish and whales may have to make longer journeys to find prey. • Birds arrive earlier in spring, but plants and invertebrates they depend on for food may not be available at the same time.

Concept 25. 2 Global Climate Changes in community composition and local extinctions may be indicators of climate change. Example: Warmer water has affected coral reef community structure.

CONCEPT 25. 3 Anthropogenic emissions of sulfur and nitrogen cause acid deposition, alter soil chemistry, and affect the health of ecosystems.

Concept 25. 3 Acid and Nitrogen Deposition Since the Industrial Revolution, air pollution has been associated with urban industrial centers, power plants, and oil and gas refineries. Increasing emissions from cars, taller smokestacks, and widespread industrial development have increased the spatial extent of air pollution.

Concept 25. 3 Acid and Nitrogen Deposition Emissions of N and S have resulted in two related issues: • Acid precipitation • N deposition Sites affected by these problems now include national parks and wilderness areas.

Figure 25. 17 Air Quality Monitoring in Grand Canyon National Park

Concept 25. 3 Acid and Nitrogen Deposition Awareness of the widespread effects of acid precipitation, even in pristine areas, increased during the 1960 s. Damage to forests and aquatic ecosystems became well-known.

Concept 25. 3 Acid and Nitrogen Deposition Large-scale mortality of trees in European forests during the 1970 s and 1980 s was associated with acid precipitation, Ca and Mg deficiencies, and other stresses.

Figure 25. 18 Air Pollution Has Damaged European Forests
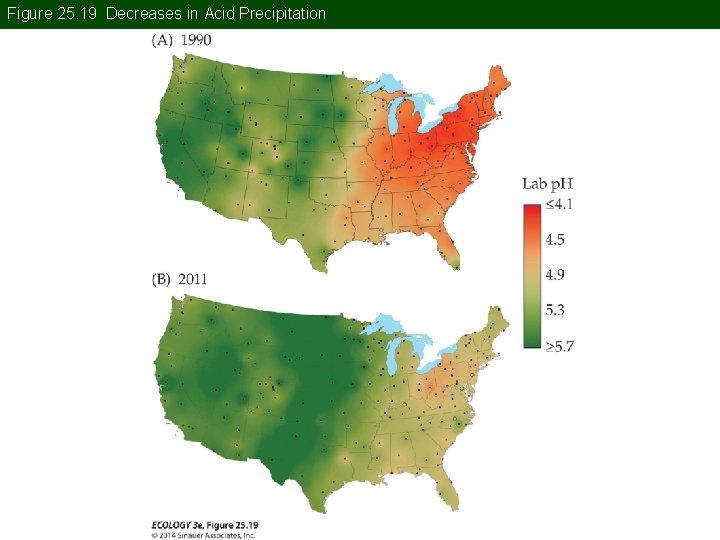
Figure 25. 19 Decreases in Acid Precipitation

Concept 25. 3 Acid and Nitrogen Deposition Increased N supplies might be expected to increase plant growth and production. Primary production has increased in some ecosystems. It may be partly responsible for a greater uptake of atmospheric CO 2 by terrestrial ecosystems.

Concept 25. 3 Acid and Nitrogen Deposition But N deposition is also associated with environmental degradation, loss of diversity, and acidification. Nitrogen saturation—N deposition may exceed the capacity of plants and microbes to take it up.
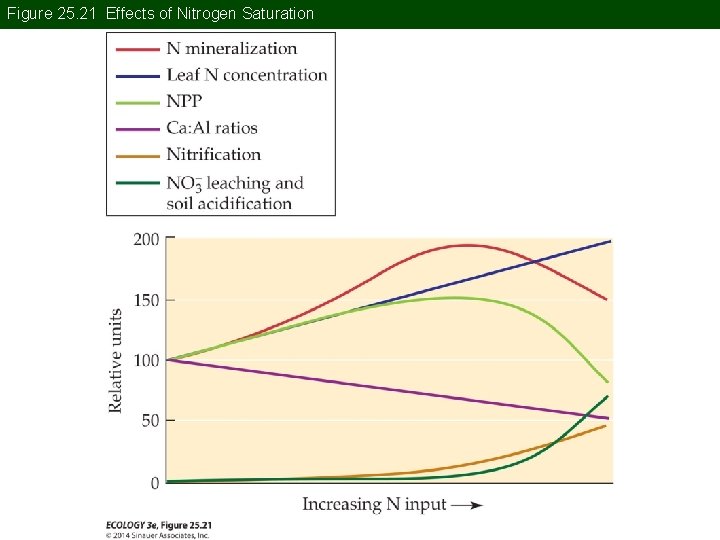
Figure 25. 21 Effects of Nitrogen Saturation

Concept 25. 3 Acid and Nitrogen Deposition • N export to nearshore marine ecosystems contributes to eutrophication and oxygen depletion. § Anoxic conditions over large areas are called “dead zones. ”

Concept 25. 3 Acid and Nitrogen Deposition • In nutrient-poor environments, many plants have adaptations that lower their nutrient requirements, which lowers their capacity to take up excess N. § Faster-growing species may then outcompete them, resulting in loss of biodiversity and change in community composition.

Concept 25. 3 Acid and Nitrogen Deposition In Holland, species-rich heath communities, adapted to low-nutrient conditions, have been replaced by species-poor grassland communities as a result of very high rates of N deposition.

Concept 25. 3 Acid and Nitrogen Deposition A survey of grasslands in Great Britain looked at a range of N deposition rates (Stevens et al. 2004). Of 20 factors that may have influenced species richness, N deposition rate explained the most variation. Higher N inputs were associated with lower species richness.
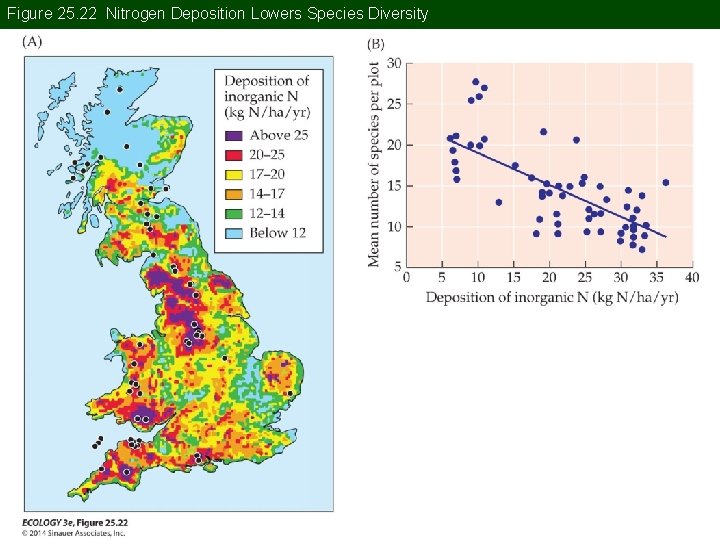
Figure 25. 22 Nitrogen Deposition Lowers Species Diversity

Concept 25. 3 Acid and Nitrogen Deposition Many experimental studies have also shown that adding N to experimental plots decreases species richness, often resulting in loss of rare species. High N deposition rates also facilitate the spread of some invasive plant species.

CONCEPT 25. 4 Losses of ozone in the stratosphere and increases in ozone in the troposphere each pose risks to organisms.

Concept 25. 4 Atmospheric Ozone Statospheric ozone (O 3) protects Earth’s surface from high-energy ultraviolet-B (UVB) radiation. UVB radiation causes damage to DNA and photosynthetic pigments, impairment of immune responses, and cancerous skin tumors in animals.

Concept 25. 4 Atmospheric Ozone Stratospheric ozone concentrations decrease in spring in polar regions. In 1980, British scientists measured an unusually large decrease in springtime ozone over Antarctica. This phenomenon is known as the ozone hole, and it has increased in intensity and spatial extent.
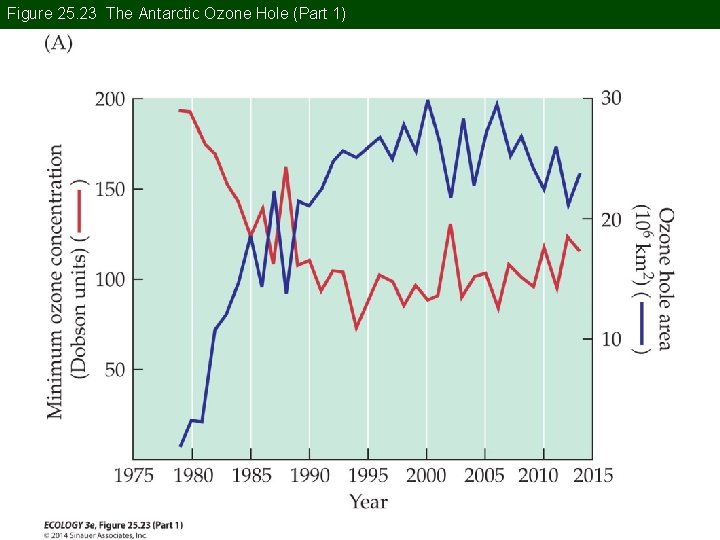
Figure 25. 23 The Antarctic Ozone Hole (Part 1)

Figure 25. 23 The Antarctic Ozone Hole (Part 2)

Concept 25. 4 Atmospheric Ozone An ozone hole is not really a hole, but an area with low ozone concentrations. In the Arctic, the decreases have not been as great (the Arctic ozone dent).

Concept 25. 4 Atmospheric Ozone Molina and Rowland (1974) predicted the decrease in stratospheric ozone due to manmade compounds called chlorofluorocarbons (CFCs). CFCs were developed in the 1930 s as refrigerants and propellants in spray cans of paint, deodorants, hair spray, etc.

Concept 25. 4 Atmospheric Ozone In the stratosphere, CFCs react with other compounds to produce reactive chlorine atoms that destroy ozone. A single free chlorine atom can destroy 105 ozone molecules. Amount of UVB radiation at Earth’s surface increased as stratospheric ozone concentration decreased.

Concept 25. 4 Atmospheric Ozone Increased UVB radiation is correlated with higher incidence of skin cancer in humans. UVB radiation influenced evolution of skin pigmentation in humans. The pigment melanin was selected for in populations at low latitudes where ozone levels are naturally lowest.

Concept 25. 4 Atmospheric Ozone Several international conferences on ozone destruction took place in the 1980 s. The Montreal Protocol is an international agreement calling for reduction and eventual ban on CFCs and other ozonedegrading chemicals.
- Slides: 90