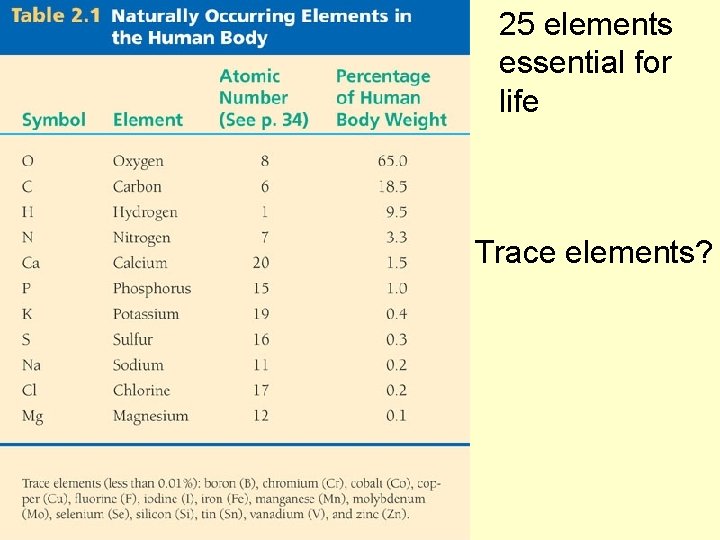
25 elements essential for life Trace elements Symptom






















- Slides: 54

25 elements essential for life Trace elements?

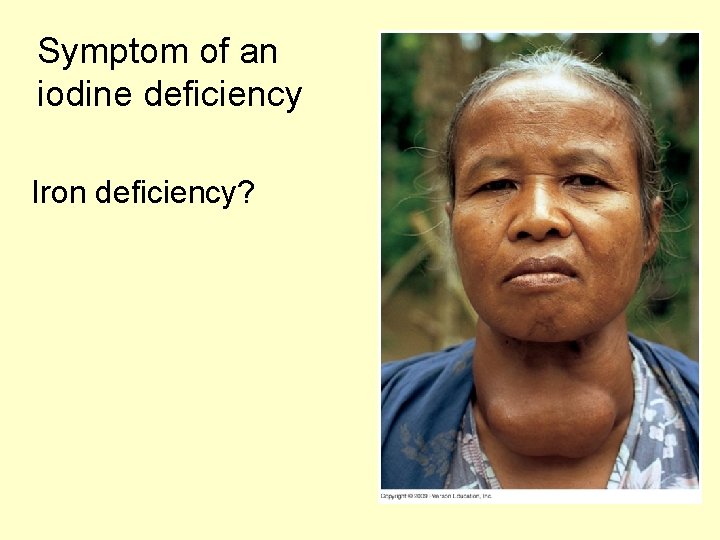
Symptom of an iodine deficiency Iron deficiency?

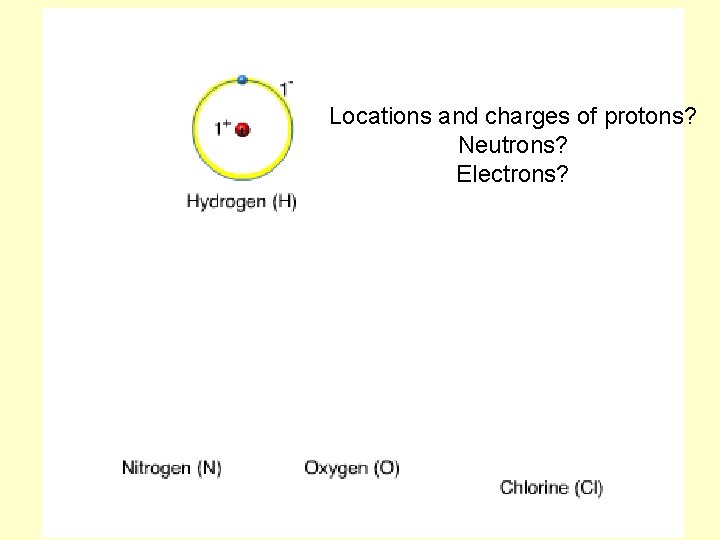
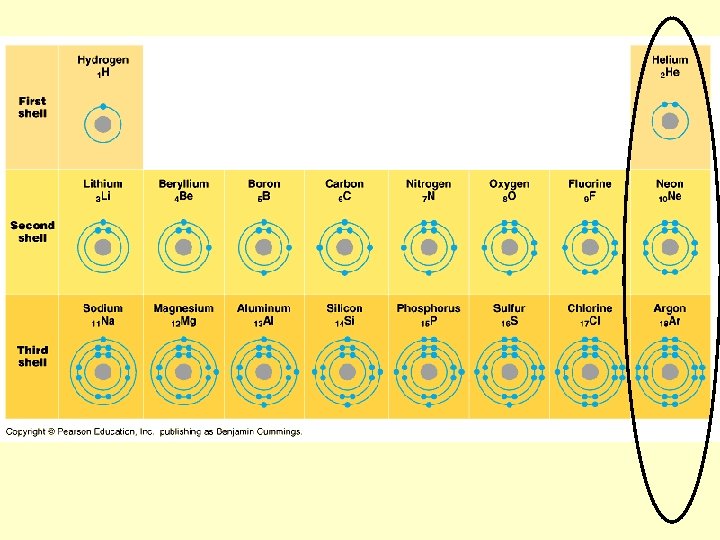
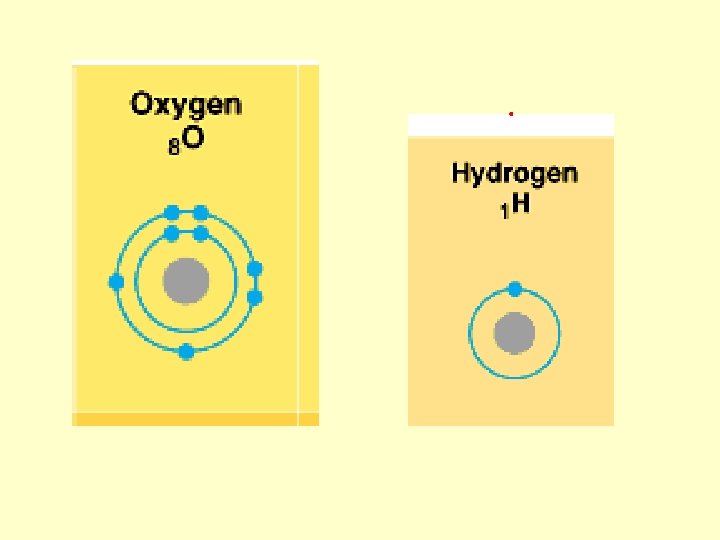
Locations and charges of protons? Neutrons? Electrons?

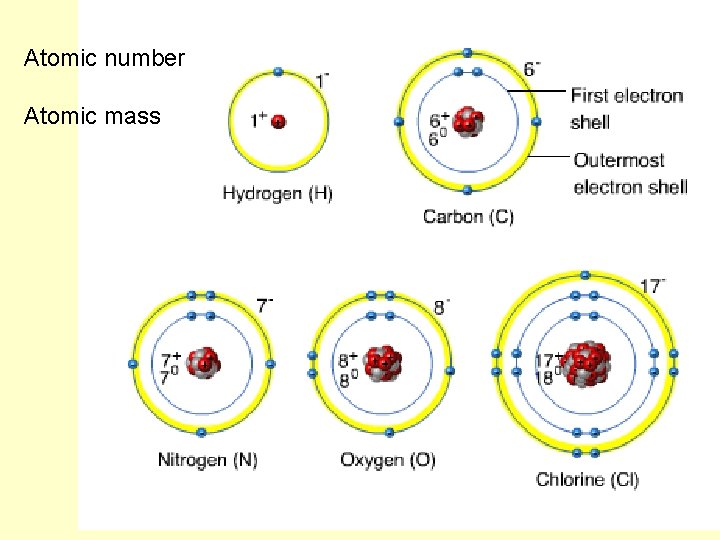
Atomic number Atomic mass

Atomic Mass vs. Atomic Number? ? ?

What is an isotope?

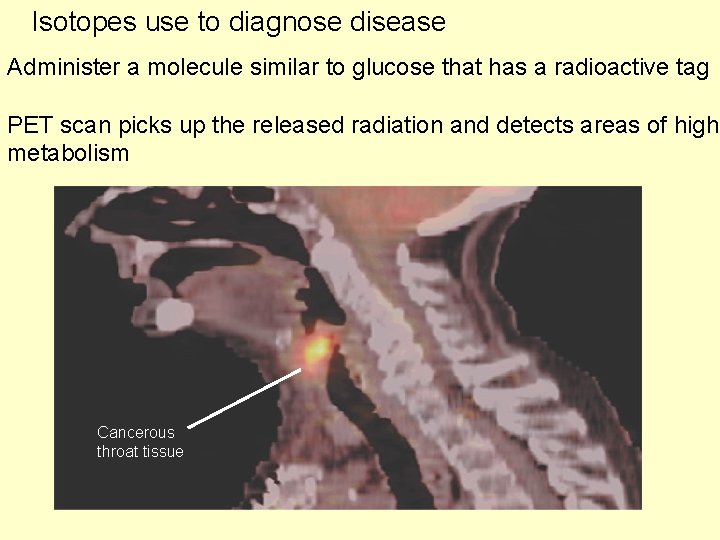
Isotopes use to diagnose disease Administer a molecule similar to glucose that has a radioactive tag PET scan picks up the released radiation and detects areas of high metabolism Cancerous throat tissue

Urea Breath Tests for stomach bacteria: Helicobacter pylori Give urea with some of the carbon being 14 C. If there is Helicobacter pylori in the stomach, then this bacteria will break down the urea and produce C 02. The CO 2 will be exhaled by the patient and collected in a balloon. If some of the carbon in the exhaled CO 2 contains is 14 C then there must be some bacteria present in the stomach. The more 14 C exhaled, the more Helicobacter pylori present. The breath test can be repeated to determine the success of the treatment.

Radioactive iodine Used treat hyperthyroidism and thyroid cancer, Small doses used to test thyroid function.

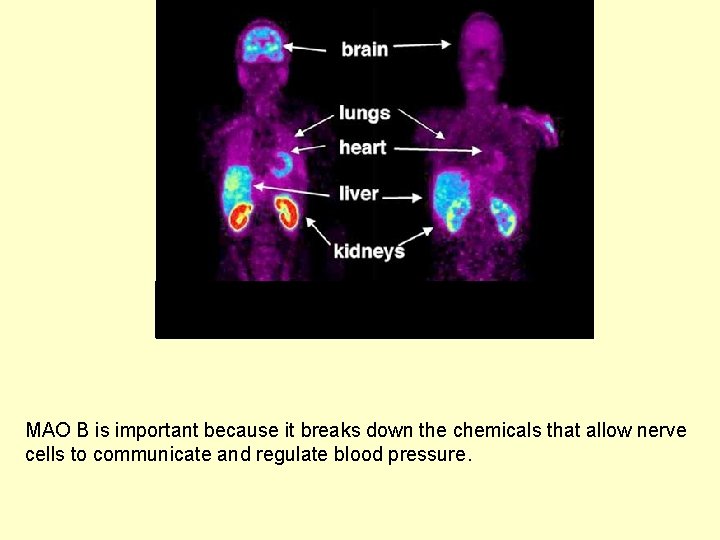
shows the concentration of radioactive tracer bound to monoamine oxidase B (MAO B). Red shows the highest concentration. MAO B is important because it breaks down the chemicals that allow nerve cells to communicate and regulate blood pressure.

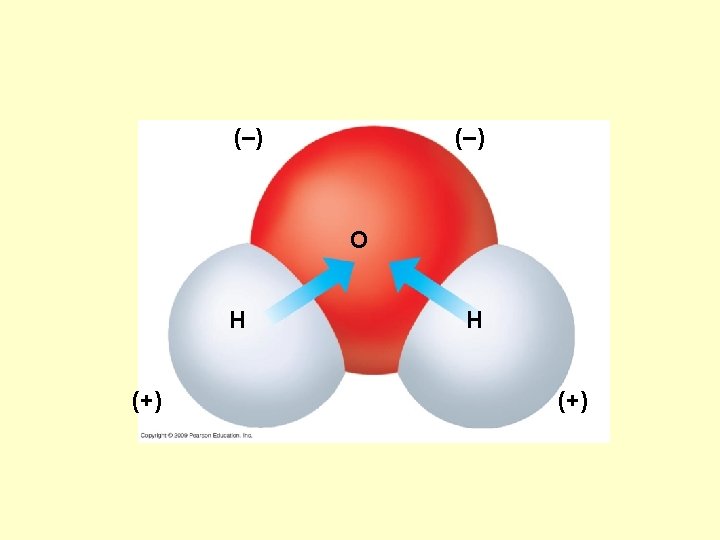
What causes an atom to react with other atom? Or… would cause it to be nonreactive (stable)?


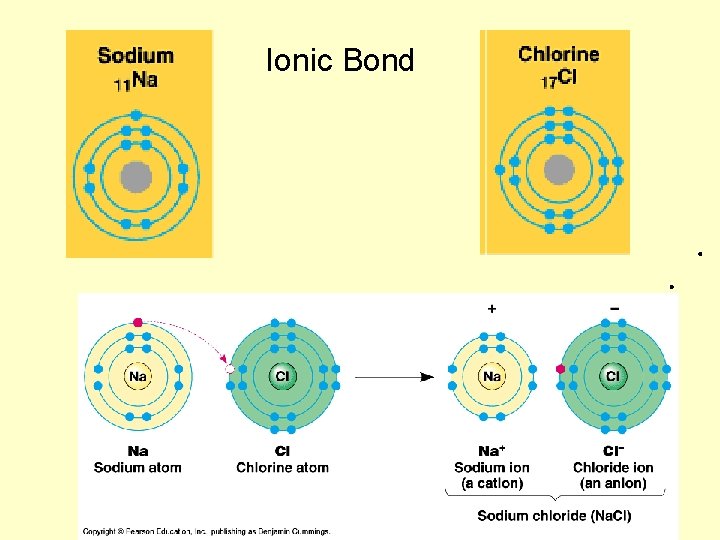
Ionic Bond










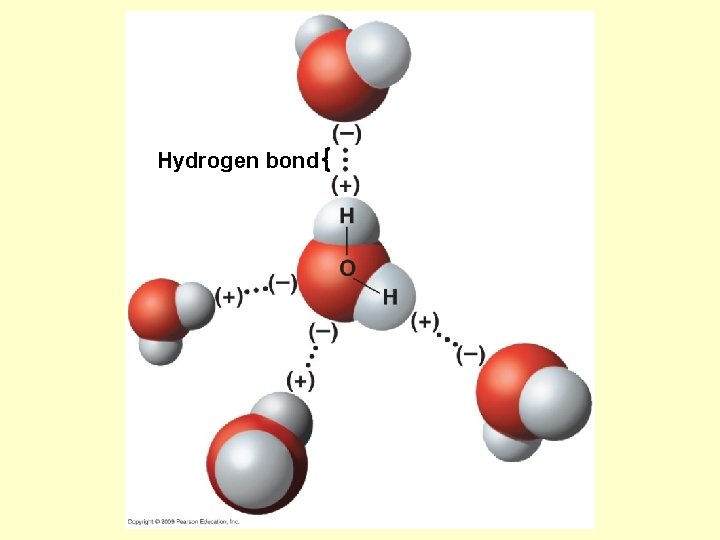
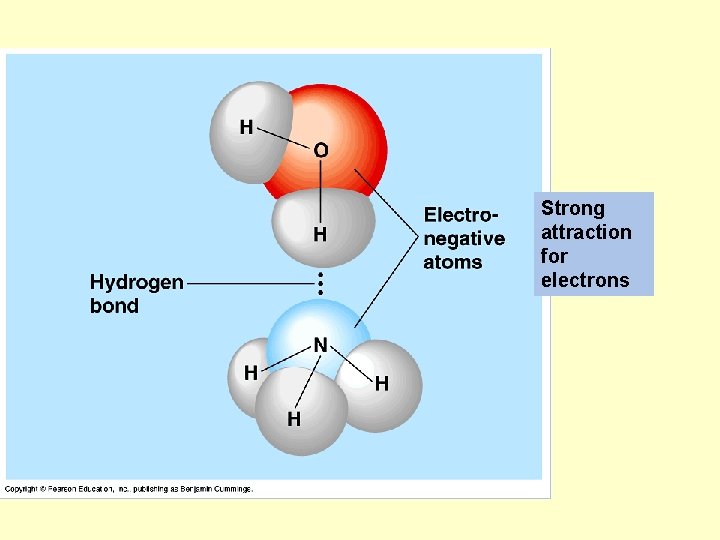
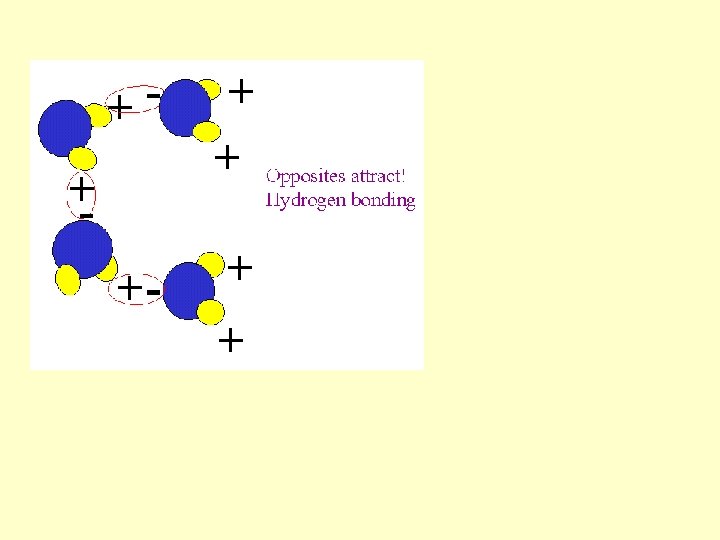
Hydrogen bond

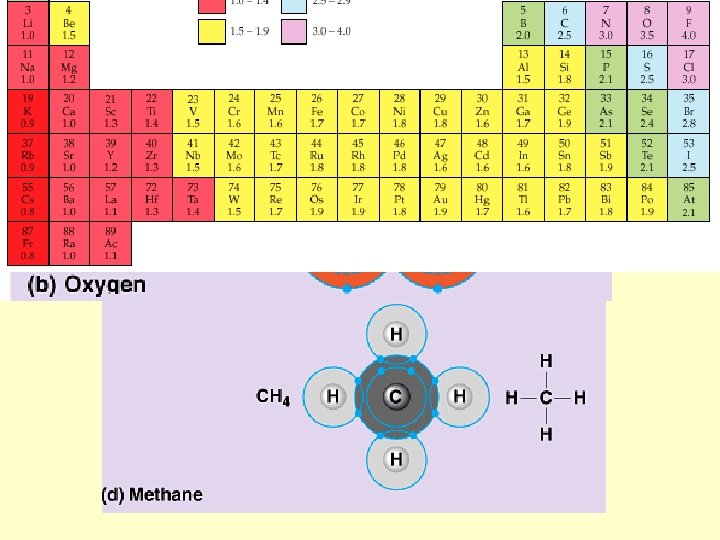
Strong attraction for electrons

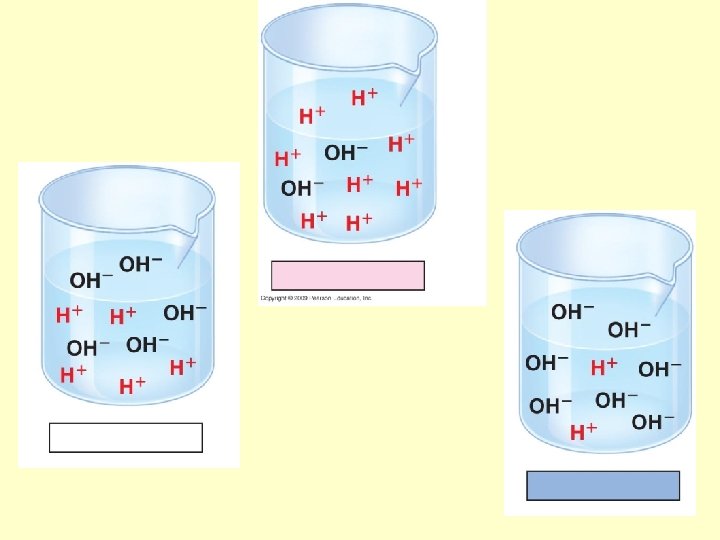
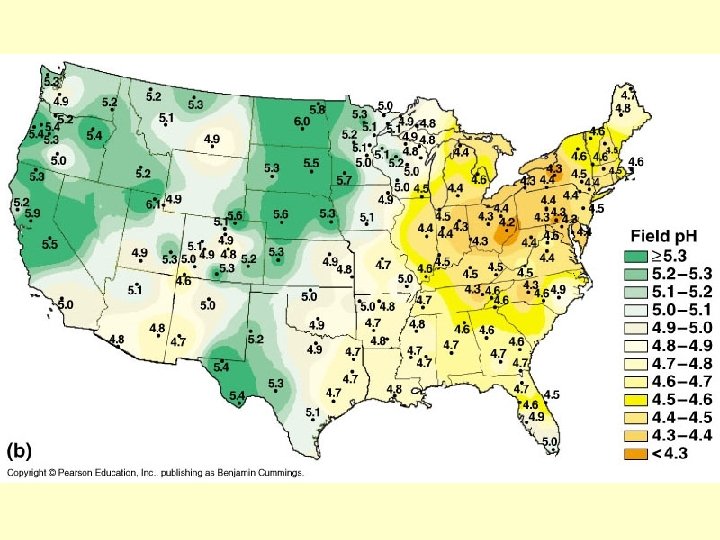
p. H • A few water molecules can break apart into ions – hydrogen ions (H+) – hydroxide ions (OH–) Copyright © 2009 Pearson Education, Inc.

p. H • Acids vs. bases Copyright © 2009 Pearson Education, Inc.

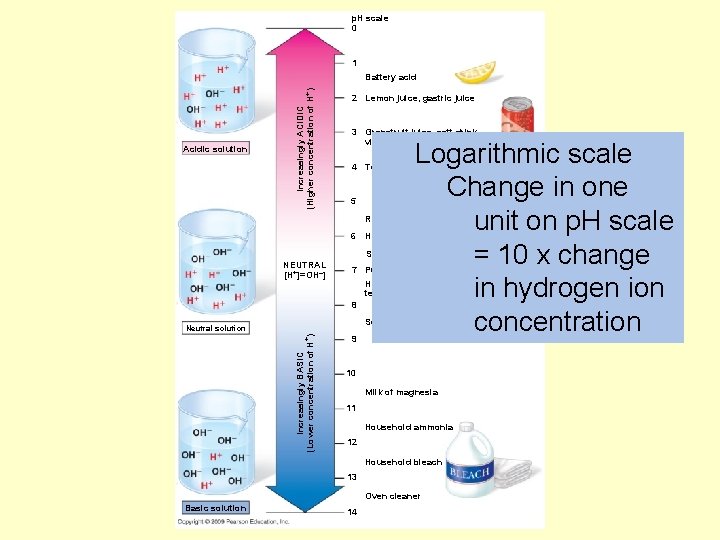
• A p. H scale (p. H = potential of hydrogen) is used to describe whether a solution is acidic or basic – p. H ranges from 0 (most acidic) to 14 (most basic) – A solution that is neither acidic or basic is neutral (p. H = 7) Copyright © 2009 Pearson Education, Inc.

p. H scale 0 1 Acidic solution Increasingly ACIDIC (Higher concentration of H+) Battery acid 2 Lemon juice, gastric juice 3 Grapefruit juice, soft drink, vinegar, beer Logarithmic scale Change in one unit on p. H scale = 10 x change in hydrogen ion concentration 4 Tomato juice 5 Rain water 6 Human urine Saliva NEUTRAL [H+]=OH–] 7 Pure water Human blood, tears 8 Seawater Increasingly BASIC (Lower concentration of H+) Neutral solution 9 10 Milk of magnesia 11 Household ammonia 12 Household bleach 13 Oven cleaner Basic solution 14

Solution A has a p. H = 4 Solution B has a p. H = 2 How much more acidic is Solution B? Solution C has a p. H = 9 Solution D has a p. H = 12 How much more H+ ions does solution C have compared to solution D?

Why is p. H important? • Different areas of the body have different p. H values – Ex: stomach blood • If the p. H varies from this value, proteins lose their shape and don’t work

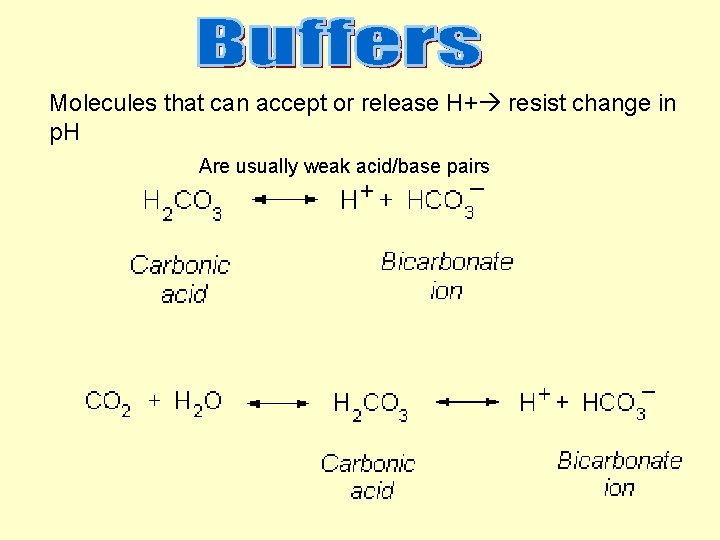
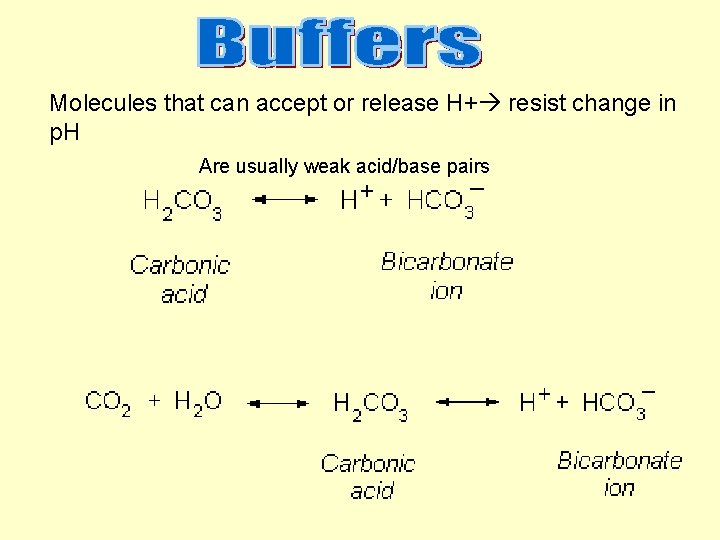
Molecules that can accept or release H+ resist change in p. H Are usually weak acid/base pairs



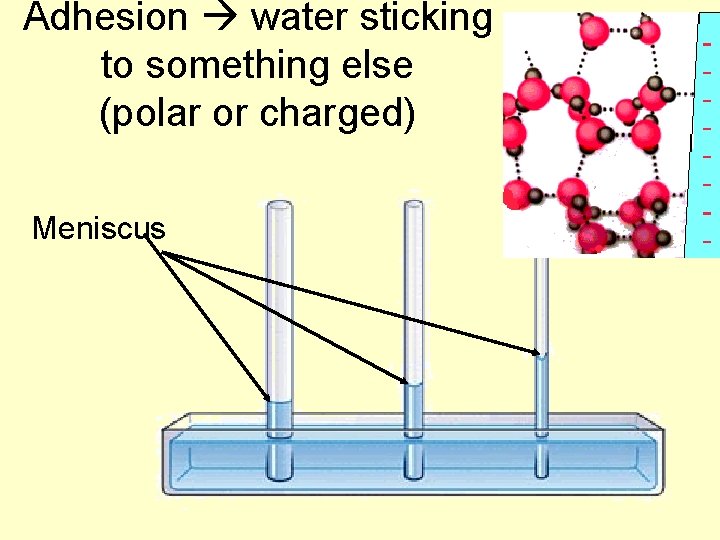
Cohesion of water sticking to itself • Does this because of hydrogen bonding • Results in water having surface tension – “film” on top of water

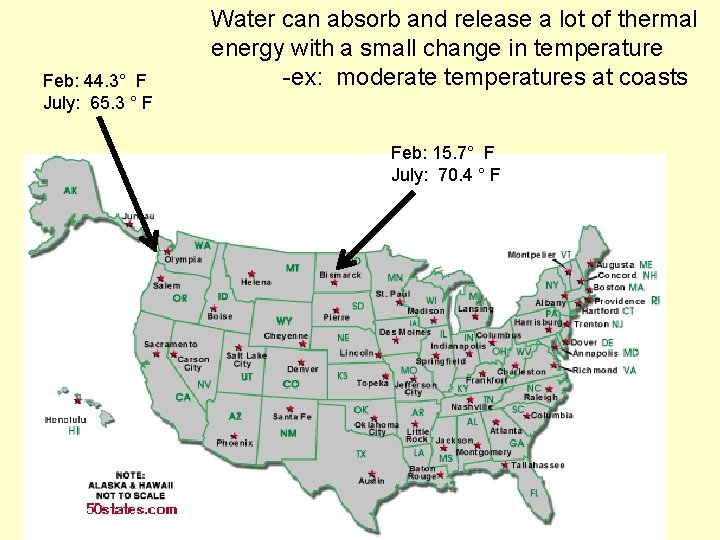
Temperature moderation Water resists changes in temperature

Feb: 44. 3° F July: 65. 3 ° F Water can absorb and release a lot of thermal energy with a small change in temperature -ex: moderate temperatures at coasts Feb: 15. 7° F July: 70. 4 ° F

Absorbs a lot of energy when heating up Releases energy when cooling down ex: evaporative cooling

Adhesion water sticking to something else (polar or charged) Meniscus


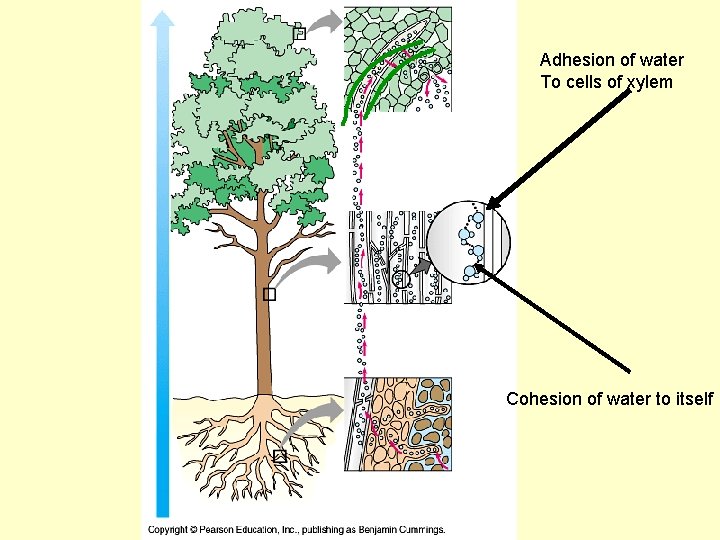
Transpiration • Uses cohesion and adhesion to move water from the roots to top of a tree

Adhesion of water To cells of xylem Cohesion of water to itself

Density Water is densest at 4° C Provides insulation and allows wildlife to survive under a layer of ice Ice is less dense than liquid water Ice floats

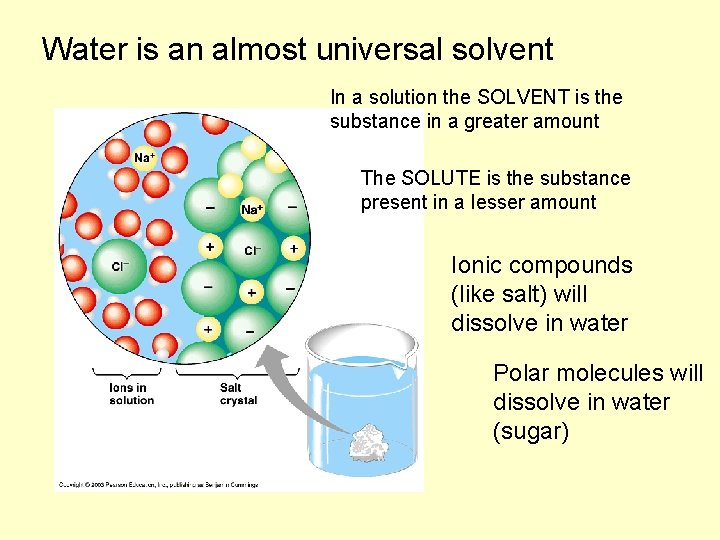
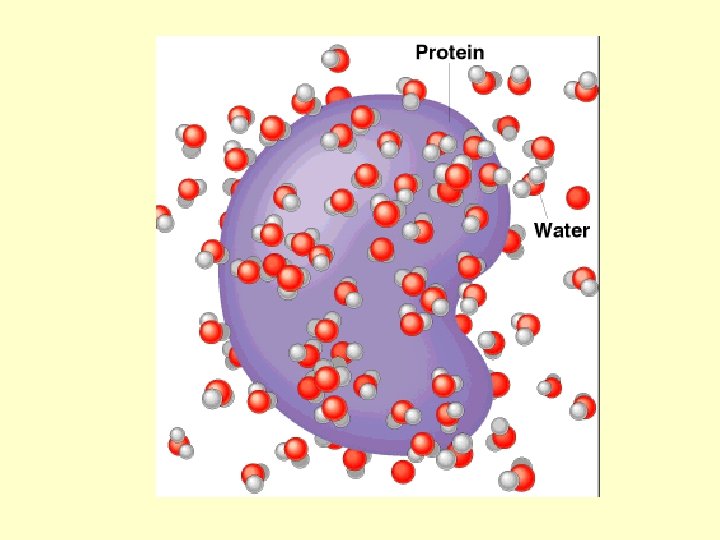
Water is an almost universal solvent In a solution the SOLVENT is the substance in a greater amount The SOLUTE is the substance present in a lesser amount Ionic compounds (like salt) will dissolve in water Polar molecules will dissolve in water (sugar)


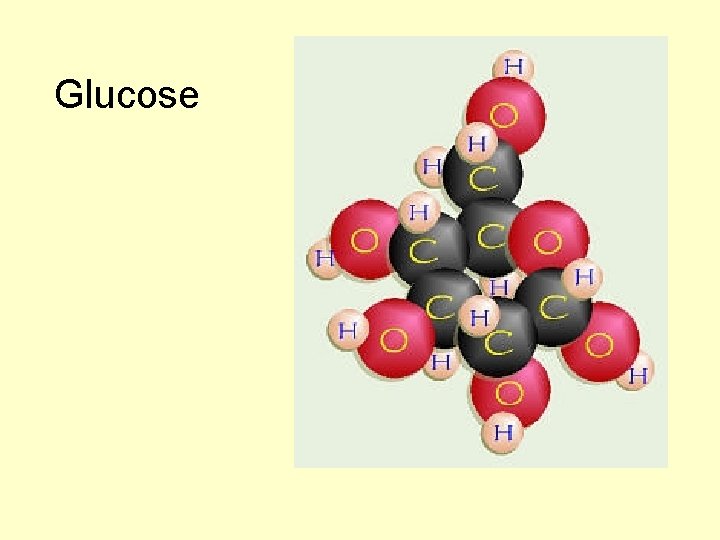
Glucose


Molecules that can accept or release H+ resist change in p. H Are usually weak acid/base pairs






