25 December 2021 1 Last Lesson 2 Last
25 December 2021 1. Last Lesson 2. Last week Describe the different energy changes when making & breaking intermolecular bonds 5. Challenge • Give an example of a physical change. • Describe what happens to the particles in this change • What is ultrasound? 3. Last topic Explain in your own words why light bends when it travels from air into a glass block • Give 2 uses of ultrasound. 4. Last Year • Name the different types of energy. • Group these into; o ‘stored energy’ o ‘energy in action’. Success is the result of preparation, hard work and learning from failure

Understand write word equations. • • Products Reactions Compound Balanced Complete simple word equations. Write word equations based on information and work out the products formed. Write balanced equations.

Explain what has happened when magnesium metal was burnt in oxygen. Identify the reactants and the products. You must write a minimum of 2 full sentences and include a minimum of 2 key words. Reaction - Magnesium and Oxygen | Think. Tac You. Tube

Word Equations • All equations take the general form: Reactants Products Word equations simply replace “reactants and products” with the names of the actual reactants and products. E. g Reactants Products Magnesium + oxygen Magnesium oxide Sodium + water Sodium hydroxide + hydrogen Magnesium + lead nitrate Magnesium nitrate + lead Water + calcium nitrate Nitric acid + calcium hydroxide

25/12/2021 • • Hydrochloric acid = Chloride salts Sulfuric acid = Sulphate salts Nitric acid = Nitrate salts Metals + Acid Salt + Hydrogen. For example, Lithium + Sulphuric Acid Lithium Sulphate + Hydrogen
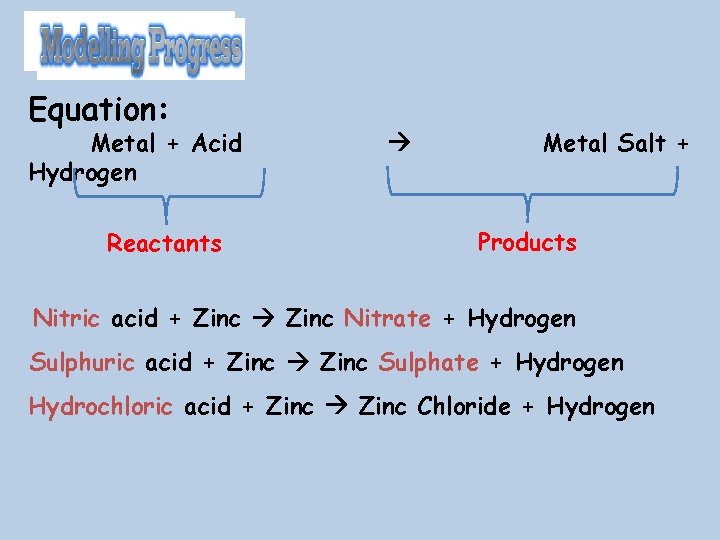
Equation: Metal + Acid Hydrogen Reactants Metal Salt + Products Nitric acid + Zinc Nitrate + Hydrogen Sulphuric acid + Zinc Sulphate + Hydrogen Hydrochloric acid + Zinc Chloride + Hydrogen
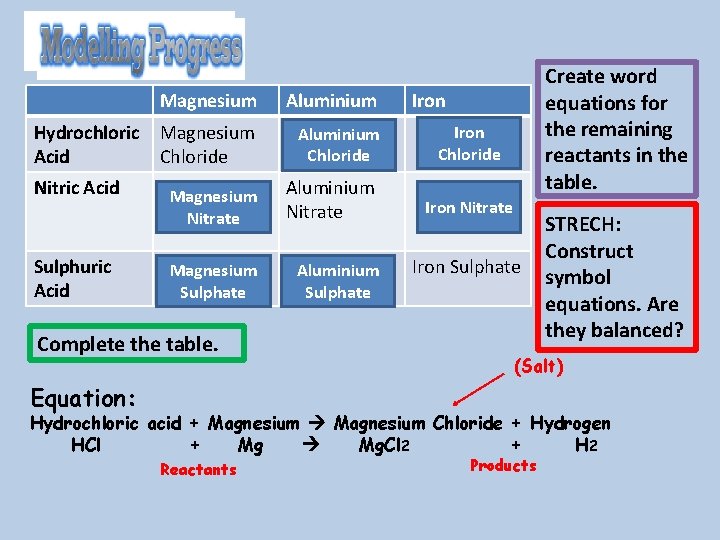
Hydrochloric Acid Nitric Acid Sulphuric Acid Magnesium Aluminium Magnesium Chloride Aluminium Chloride Magnesium Nitrate Aluminium Nitrate Magnesium Sulphate Aluminium Sulphate Complete the table. Create word equations for the remaining reactants in the table. Iron Chloride Iron Nitrate Iron Sulphate STRECH: Construct symbol equations. Are they balanced? (Salt) Equation: Hydrochloric acid + Magnesium Chloride + Hydrogen HCl + Mg Mg. Cl 2 + H 2 Reactants Products

• Write the word equations for the descriptions below. 1. The copper oxide was added to hot sulphuric acid and it reacted to give a blue solution of copper sulphate and water. Copper oxide + sulphuric acid copper sulphate + water 2. The magnesium was added to hot sulphuric acid and it reacted to give colourless magnesium sulphate solution plus hydrogen Magnesium + sulphuric acid Magnesium + sulphate hydrogen

• Write the word equations for the descriptions below. 3. The methane burned in oxygen and it reacted to give carbon dioxide and water. methane + oxygen Carbon dioxide + water 4. The copper metal was placed in the silver nitrate solution. The copper slowly disappeared forming blue copper nitrate solution and needles of silver metal seemed to grow from the surface of the copper + Silver nitrate Copper nitrate + silver

25/12/2021 Write the word equations for the reactions below. 1. Calcium + Hydrochloric Acid ______ + _____ 2. Magnesium + Hydrochloric Acid ______ + _____ 3. Copper + Copper Chloride + _____ 4. Lead + Nitric Acid ______ + _____ 5. Iron + _________ Iron Nitrate + _____ 6. Zinc + Sulfuric Acid ________ + Hydrogen 7. Tin + _________ Tin Sulphate + Hydrogen 8. Calcium______ + Sulfuric Acid ______ 9. Nickel + _________ Nickel Chloride + _____ Create your own equation based on your knowledge so far

25/12/2021 1. Calcium + Hydrochloric Acid Calcium Chloride + Hydrogen 2. Magnesium + Hydrochloric Acid Magnesium Chloride + Hydrogen 3. Copper + Hydrochloric Acid Copper Chloride + Hydrogen 4. Lead + Nitric Acid Lead Nitrate + Hydrogen 5. Iron + Nitric Acid Iron Nitrate + Hydrogen 6. Zinc + Sulfuric Acid Zinc Sulphate + Hydrogen 7. Tin + Sulfuric Acid Tin Sulphate + Hydrogen 8. Calcium + Sulfuric Acid Calcium Sulphate + Hydrogen 9. Nickel + Hydrochloric acid Nickel Chloride + Hydrogen

Chemical Symbol Equations EASY • Step 1: Write down the word equation. • Step 2: Replace words with the chemical formula. • Step 3: Check that there are equal numbers of each type of atom on both sides of the equation. If not, then balance the equation by using more than one. Reactants magnesium + oxygen. Mg + O 2 Products magnesium oxide Mg. O Oxygen doesn’t balance. Need 2 Mg. O and so need 2 Mg 2 Mg +2 Mg O +O 2 2 2 Mg. O

Chemical Equations MEDIUM • • • Step 1: Write down the word equation. Step 2: Replace words with the chemical formula. Step 3: Check that there are equal numbers of each type of atom on both sides of the equation. If not, then balance the equation by using more than one. Reactants sodium + water Na + H 2 O Products hydrogen + sodium hydroxide Hydrogen doesn’t balance. + 2 Na 2 H 2 O + H 2 Na. OH Use 2 H 2 O, Na. OH, 2 Na H 2 + + 2 Na. OH

Chemical Equations HARD • • • Step 1: Write down the word equation. Step 2: Replace words with the chemical formula. Step 3: Check that there are equal numbers of each type of atom on both sides of the equation. If not, then balance the equation by using more than one. Reactants magnesium + lead nitrate Mg + Pb(NO 3)2 Already balances. + Mg Pb(NO 3)2 Products magnesium nitrate + lead Mg(NO 3)2 + Pb Just add state symbols Mg(NO 3)2 + Pb

• Below are some chemical equations where the formulae are correct but the balancing step has not been done. Write in appropriate coefficients (numbers) to make them balance. Reactants 2 Ag. NO 3(aq) + CH 4(g) + Mg(s) 2 Na. OH Products Ca. Cl 2(aq) 2 O 2(g) + Ag 2 O(s) + H 2 SO 4(aq) + 2 Ag. Cl(s) Ca(NO 3)2(aq) CO 2(g) + 2 H 2 O(g) Mg. O(s) + 2 Ag(s) Na 2 SO 4(aq) + 2 H 2 O(l)
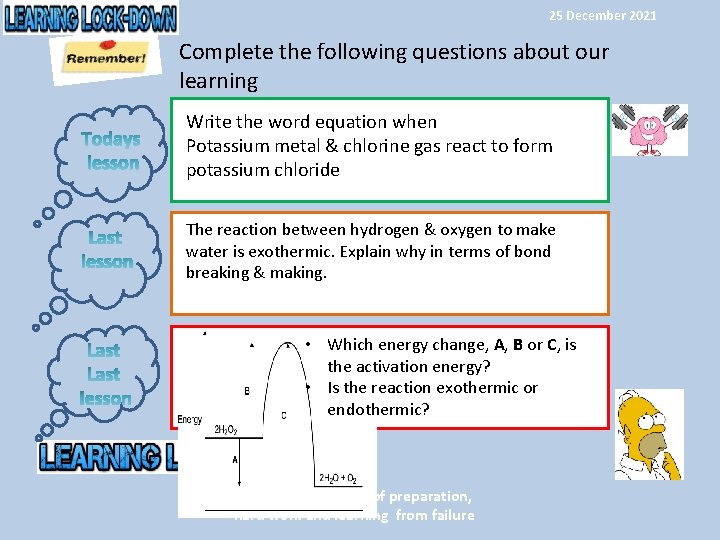
25 December 2021 Complete the following questions about our learning Write the word equation when Potassium metal & chlorine gas react to form potassium chloride The reaction between hydrogen & oxygen to make water is exothermic. Explain why in terms of bond breaking & making. • Which energy change, A, B or C, is the activation energy? • Is the reaction exothermic or endothermic? Success is the result of preparation, hard work and learning from failure
- Slides: 16