25 6 Cyclic Forms of Carbohydrates Furanose Forms

25. 6 Cyclic Forms of Carbohydrates: Furanose Forms
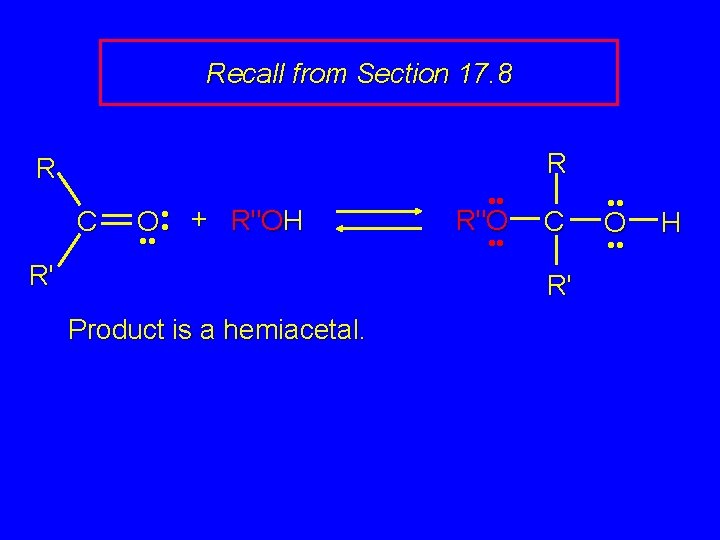
Recall from Section 17. 8 R R C O • • + R"OH • • R' • • R"O • • C R' Product is a hemiacetal. • • O • • H
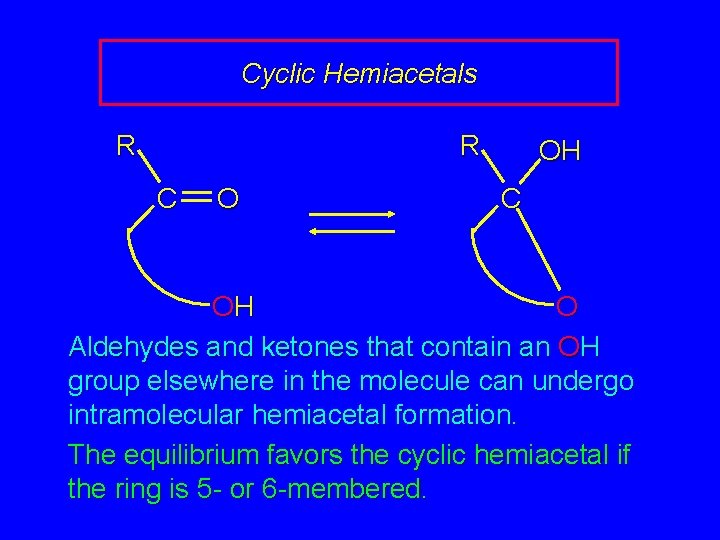
Cyclic Hemiacetals R R C O OH C OH O Aldehydes and ketones that contain an OH group elsewhere in the molecule can undergo intramolecular hemiacetal formation. The equilibrium favors the cyclic hemiacetal if the ring is 5 - or 6 -membered.
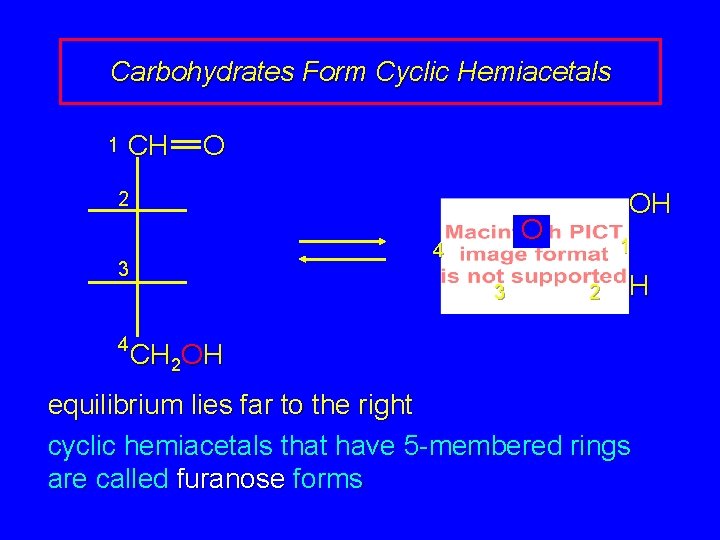
Carbohydrates Form Cyclic Hemiacetals 1 CH O OH 2 4 3 4 O 3 1 2 H CH 2 OH equilibrium lies far to the right cyclic hemiacetals that have 5 -membered rings are called furanose forms
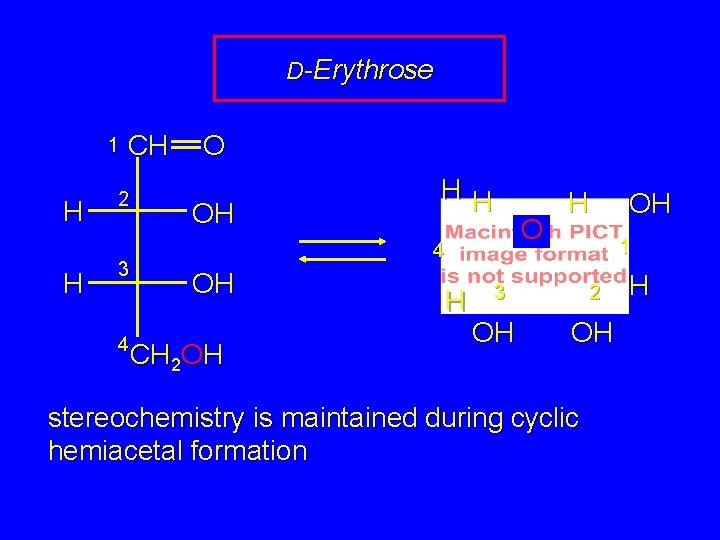
D-Erythrose 1 H H CH 2 3 4 O OH HH O 4 OH CH 2 OH H H 1 3 OH OH 2 OH stereochemistry is maintained during cyclic hemiacetal formation H

D-Erythrose 1 2 3 4

D-Erythrose 1 2 3 4 1 4 turn 90° 3 2

D-Erythrose move O into position by rotating about bond between carbon-3 and carbon-4 1 4 3 2

D-Erythrose 1 4 3 2

D-Erythrose 1 4 3 2 close ring by hemiacetal formation between OH at C-4 and carbonyl group

D-Erythrose 1 4 3 2
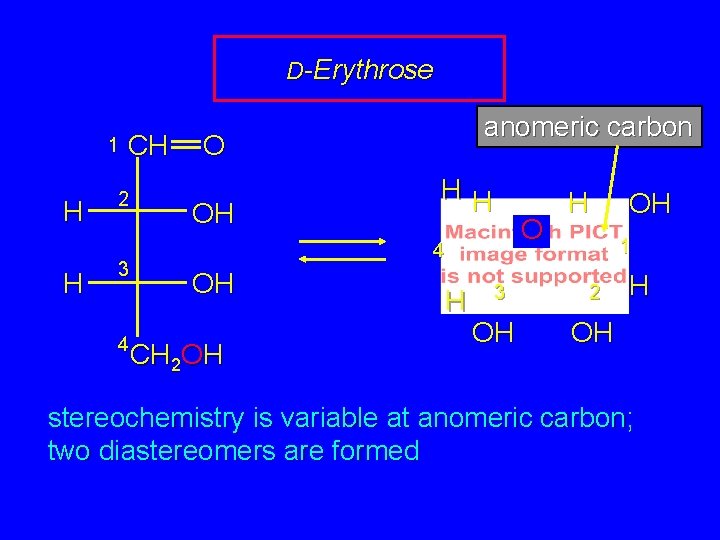
D-Erythrose 1 H H CH 2 3 4 anomeric carbon O OH HH O 4 OH CH 2 OH H 3 OH H OH 1 2 H OH stereochemistry is variable at anomeric carbon; two diastereomers are formed
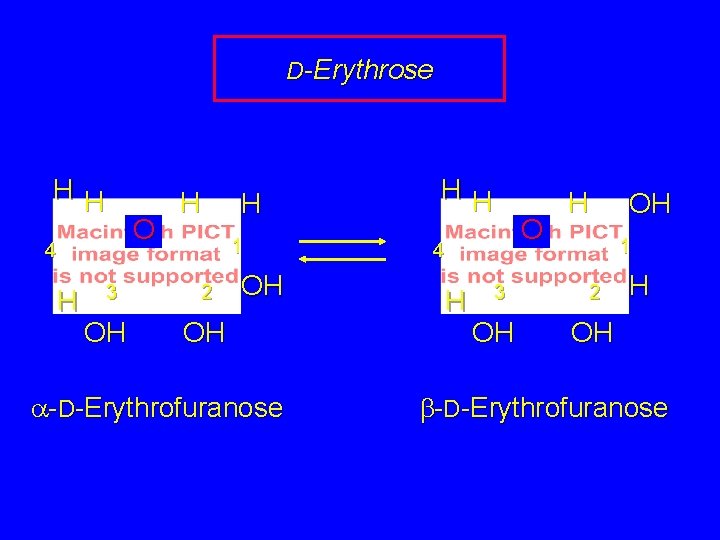
D-Erythrose HH O 4 H 3 OH H H 1 2 OH OH a-D-Erythrofuranose HH O 4 H 3 OH H OH 1 2 H OH b-D-Erythrofuranose
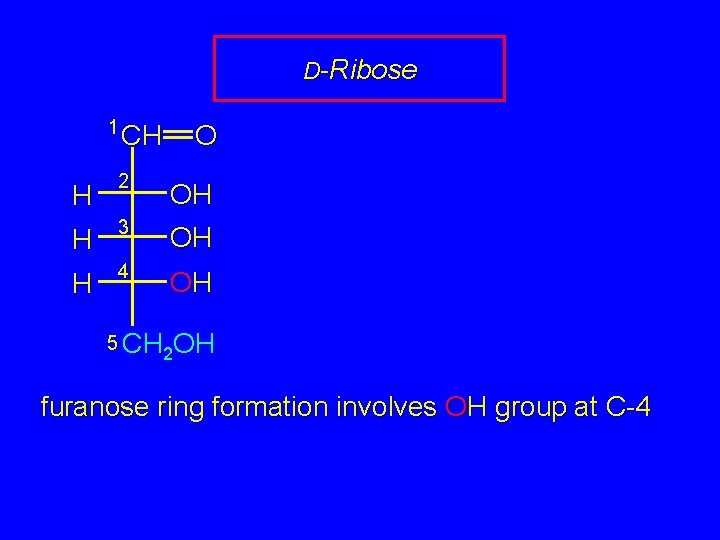
D-Ribose 1 CH O CH 2 OH H 3 OH H 4 OH H 5 CH OH 2 2 furanose ring formation involves OH group at C-4
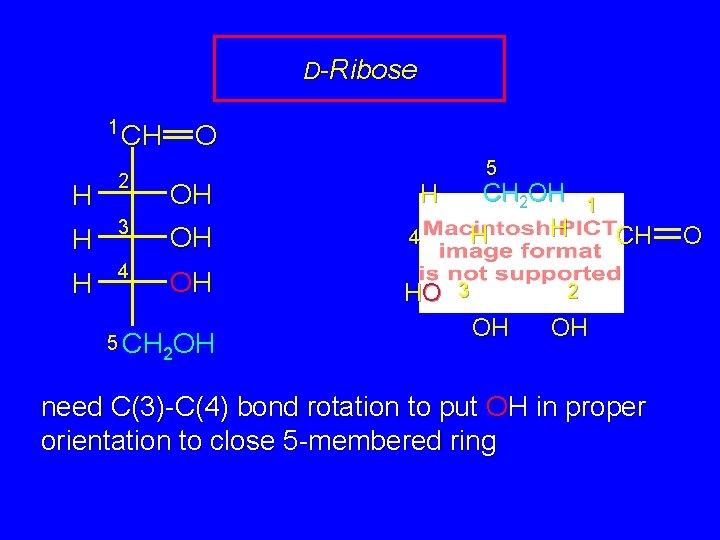
D-Ribose 1 CH O CH H 2 OH CH 2 OH H H 3 OH 4 OH HO 5 CH OH 2 5 3 1 CH 2 OH OH 2 need C(3)-C(4) bond rotation to put OH in proper orientation to close 5 -membered ring O

D-Ribose 5 HOCH 2 OH H 4 H 3 OH H 2 OH CH 2 OH H 1 CH 5 O 4 HO 3 1 2 OH OH CH O

D-Ribose 5 HOCH 2 OH H 4 H 3 OH H 1 CH O 2 OH CH 2 OH group becomes a substituent on ring
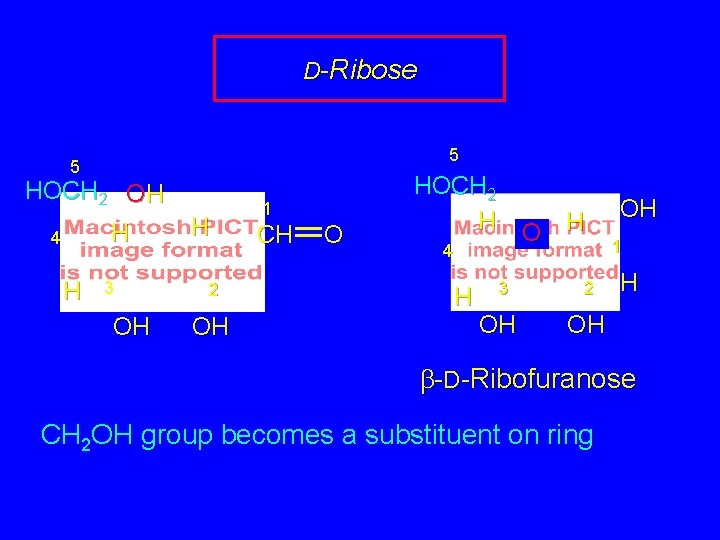
D-Ribose 5 5 HOCH 2 OH H 4 H 3 OH H 2 OH 1 CH O HOCH 2 H O H 4 H 3 OH 2 OH 1 H OH b-D-Ribofuranose CH 2 OH group becomes a substituent on ring

25. 7 Cyclic Forms of Carbohydrates: Pyranose Forms
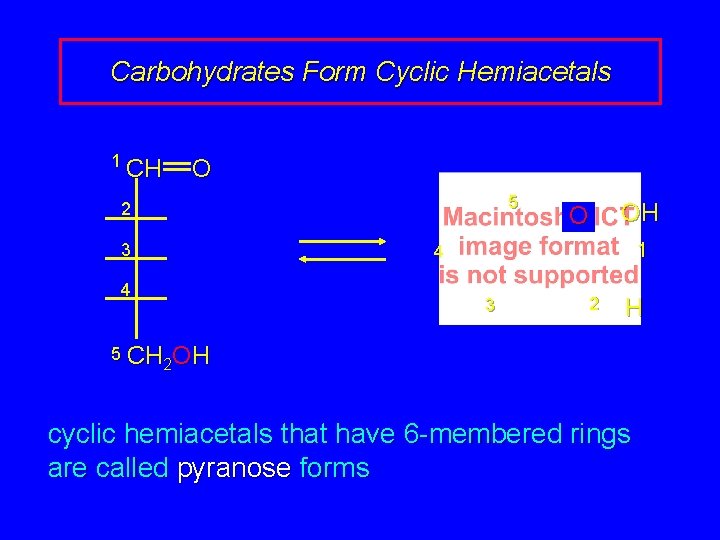
Carbohydrates Form Cyclic Hemiacetals 1 CH O 5 2 3 OH O 1 4 4 3 2 H 5 CH OH 2 2 cyclic hemiacetals that have 6 -membered rings are called pyranose forms
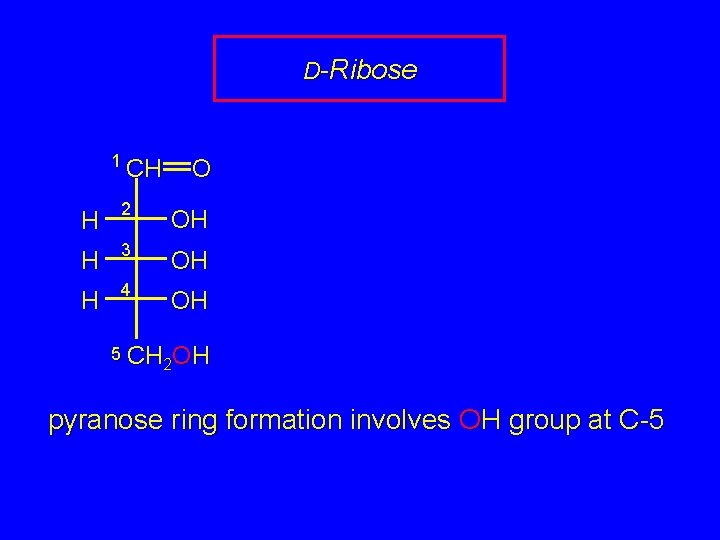
D-Ribose 1 CH O H 2 OH H 3 OH H 4 OH 5 CH OH 2 2 pyranose ring formation involves OH group at C-5
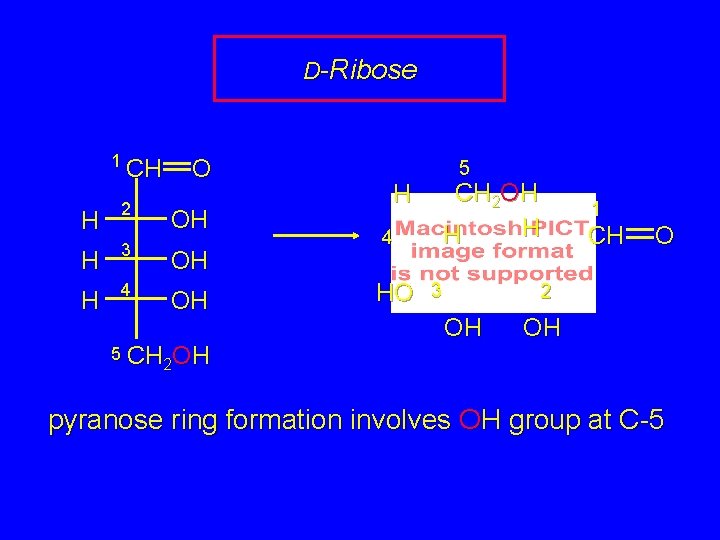
D-Ribose 1 CH O H 2 H 3 OH H 4 OH OH 5 CH OH 2 5 CH 2 OH H 4 HO 3 1 CH O 2 OH OH 2 pyranose ring formation involves OH group at C-5

D-Ribose 5 CH 2 OH H 4 HO 3 1 CH O 2 OH OH pyranose ring formation involves OH group at C-5
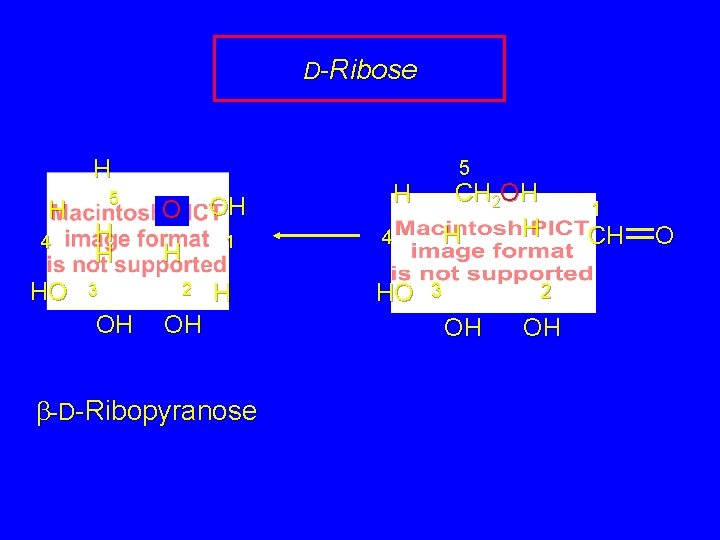
D-Ribose H H 4 HO 5 H H 3 OH O OH H 1 2 H OH b-D-Ribopyranose 5 CH 2 OH H 4 HO 3 2 OH OH 1 CH O

D-Ribose H H 4 HO 5 H H 3 OH H O OH H 1 2 OH H H 4 HO 5 H H 3 OH O H 2 H 1 OH OH a-D-Ribopyranose
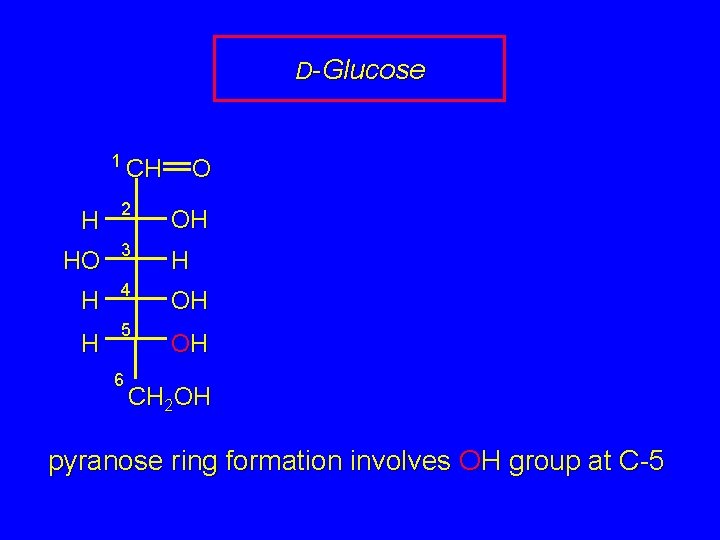
D-Glucose 1 CH O H 2 OH HO 3 H H 4 OH H 5 6 OH CH 2 OH pyranose ring formation involves OH group at C-5
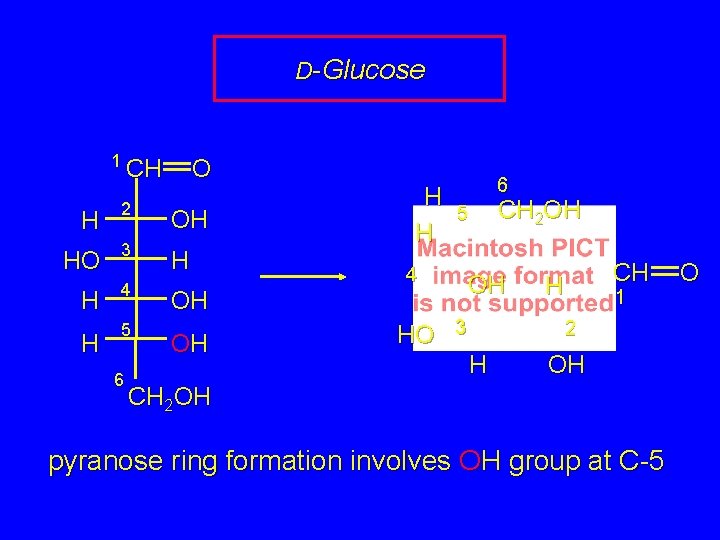
D-Glucose 1 CH O H 2 OH HO 3 H H 4 H 5 6 H H 4 OH OH HO 6 CH 2 OH 5 OH OH 3 CH H 1 2 H OH CH 2 OH pyranose ring formation involves OH group at C-5 O
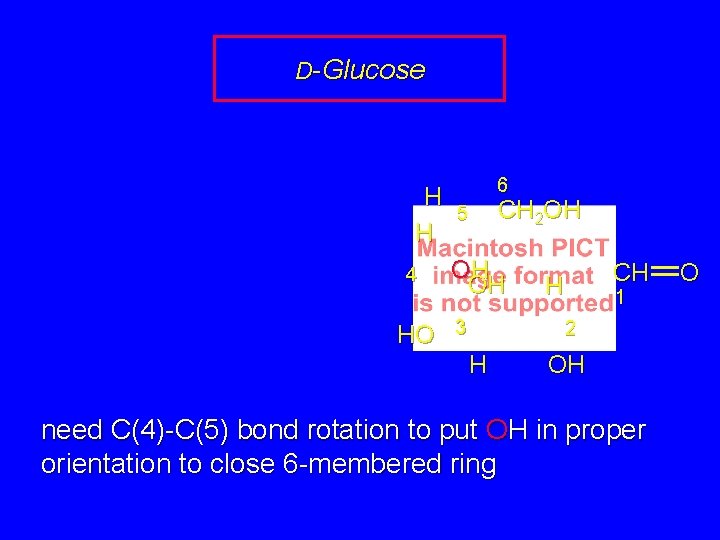
D-Glucose H H 4 HO 6 CH 2 OH 5 OH OH 3 CH H 1 2 H OH need C(4)-C(5) bond rotation to put OH in proper orientation to close 6 -membered ring O
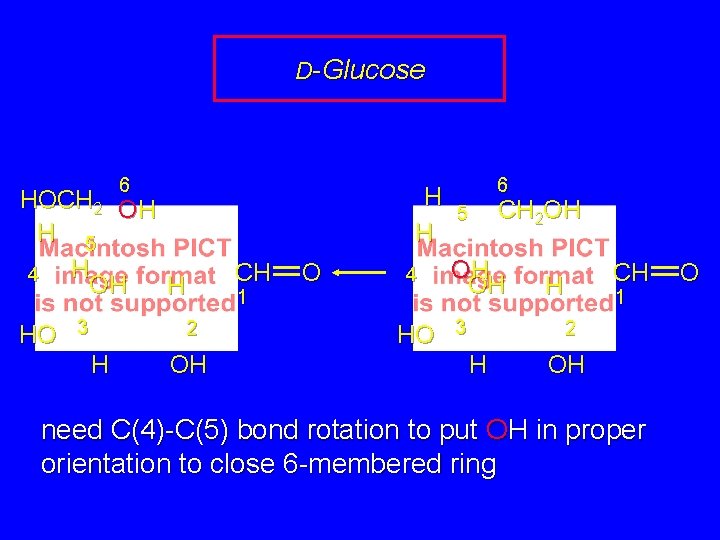
D-Glucose 6 H H HOCH 2 OH H 5 H 4 OH H HO 3 CH 1 2 H OH O 4 HO 6 CH 2 OH 5 OH OH 3 CH H 1 2 H OH need C(4)-C(5) bond rotation to put OH in proper orientation to close 6 -membered ring O
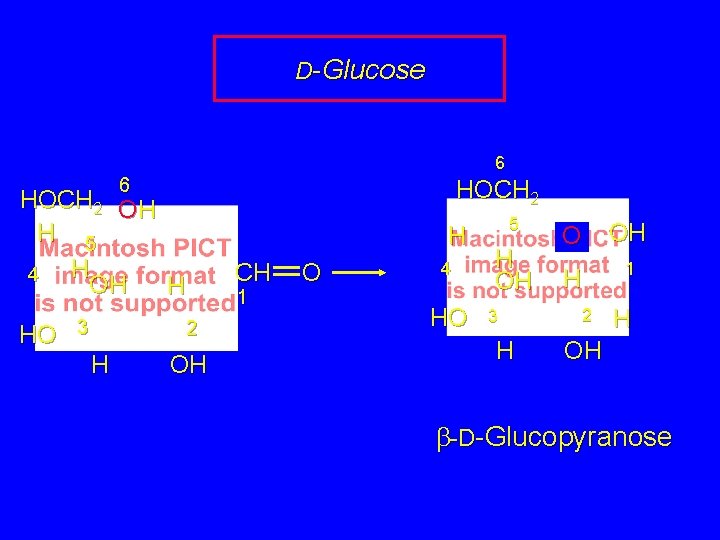
D-Glucose 6 6 HOCH 2 OH H 5 H 4 OH H HO 3 H CH 1 2 H OH O 4 HO 5 H OH 3 H O OH H 1 2 H OH b-D-Glucopyranose
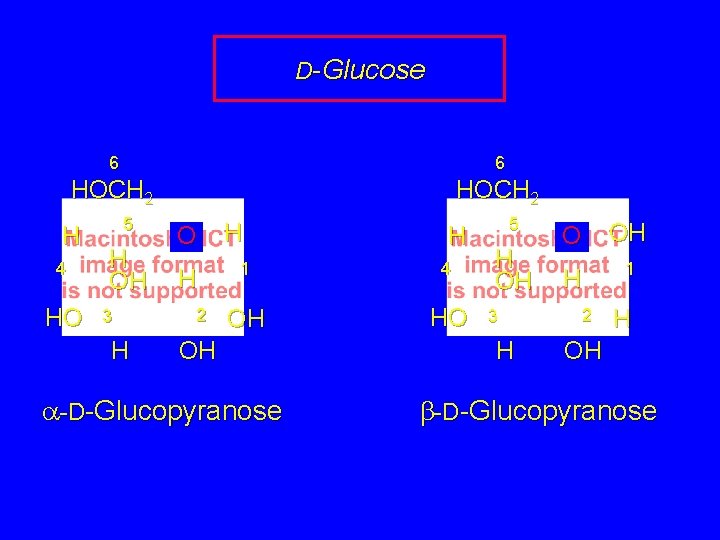
D-Glucose 6 6 HOCH 2 H 4 HO 5 H OH 3 H O H 2 H H 1 4 OH HO OH a-D-Glucopyranose 5 H OH 3 H O OH H 1 2 H OH b-D-Glucopyranose
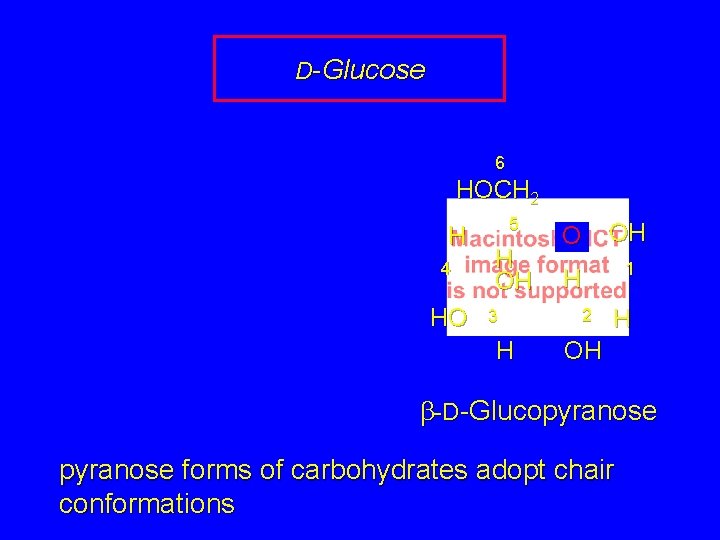
D-Glucose 6 HOCH 2 H 4 HO 5 H OH 3 H O OH H 1 2 H OH b-D-Glucopyranose forms of carbohydrates adopt chair conformations

D-Glucose 6 HOCH 2 H 4 HO HO 5 3 H HOCH 2 2 H H O OH 1 H OH 4 HO 5 H OH 3 H O OH H 1 2 H OH b-D-Glucopyranose all substituents are equatorial in b-D-glucopyranose

D-Glucose H HOCH 2 H HO HO H HOCH 2 H O OH 1 OH H b-D-Glucopyranose HO HO H H O OH 1 OH a-D-Glucopyranose OH group at anomeric carbon is axial in a-D-glucopyranose H
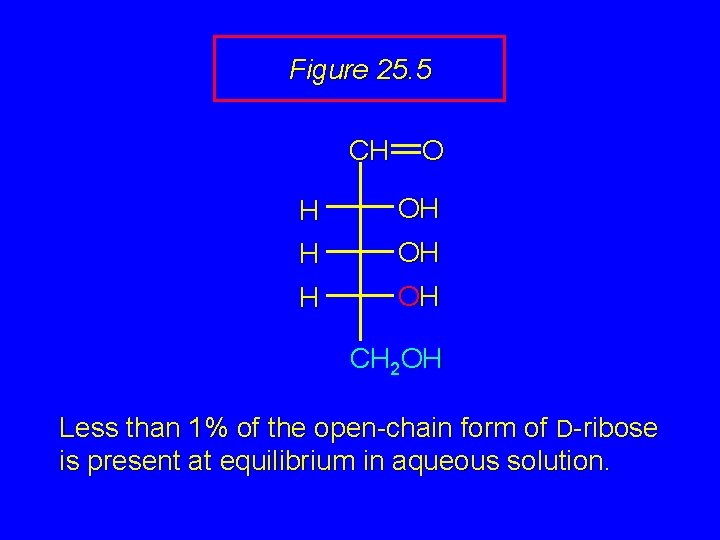
Figure 25. 5 CH O H OH CH 2 OH Less than 1% of the open-chain form of D-ribose is present at equilibrium in aqueous solution.

Figure 25. 5 76% of the D-ribose is a mixture of the a and bpyranose forms, with the b-form predominating H H O HO OH OH H b-D-Ribopyranose (56%) H O HO H H OH OH 1 H OH a-D-Ribopyranose (20%)

Figure 25. 5 The a and b-furanose forms comprise 24% of the mixture. HOCH 2 H O H H OH OH b-D-Ribofuranose (18%) HOCH 2 H O H H H OH OH OH a-D-Ribofuranose (6%)

25. 8 Mutarotation

Mutarotation is a term given to the change in the observed optical rotation of a substance with time. Glucose, for example, can be obtained in either its a or b-pyranose form. The two forms have different physical properties such as melting point and optical rotation. When either form is dissolved in water, its initial rotation changes with time. Eventually both solutions have the same rotation.

Mutarotation of D-Glucose H HOCH 2 H HO HO H HOCH 2 H O OH 1 OH H b-D-Glucopyranose Initial: [a]D +18. 7° HO HO H H O OH 1 OH a-D-Glucopyranose Initial: [a]D +112. 2° H
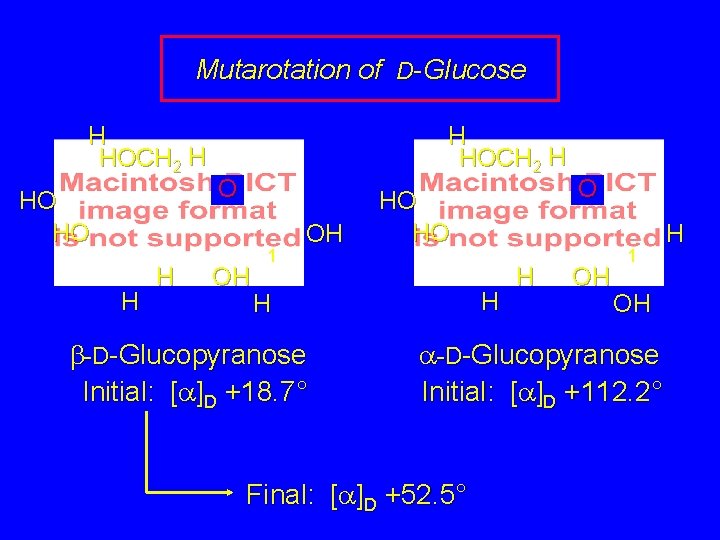
Mutarotation of D-Glucose H HOCH 2 H HO HO H HOCH 2 H O OH 1 OH HO HO H H b-D-Glucopyranose Initial: [a]D +18. 7° H O OH 1 OH a-D-Glucopyranose Initial: [a]D +112. 2° Final: [a]D +52. 5° H
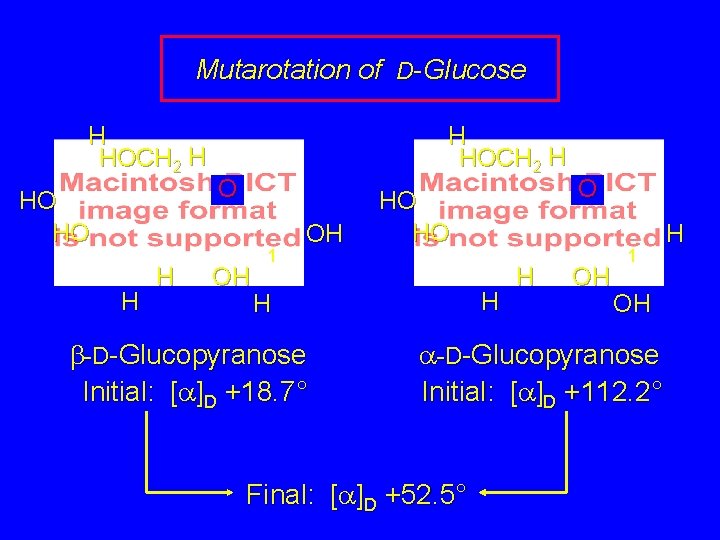
Mutarotation of D-Glucose H HOCH 2 H HO HO H HOCH 2 H O OH 1 OH HO HO H H b-D-Glucopyranose Initial: [a]D +18. 7° H O OH 1 OH a-D-Glucopyranose Initial: [a]D +112. 2° Final: [a]D +52. 5° H

Mutarotation of D-Glucose H HOCH 2 H HO HO H HOCH 2 H O OH 1 OH H b-D-Glucopyranose HO HO H H O OH 1 OH a-D-Glucopyranose Explanation: After being dissolved in water, the a and b forms slowly interconvert via the openchain form. An equilibrium state is reached that contains 64% b and 36% a. H
- Slides: 43