240 Chem Unsaturated Hydrocarbons II Alkynes Chapter 4

240 Chem Unsaturated Hydrocarbons II: Alkynes Chapter 4 1
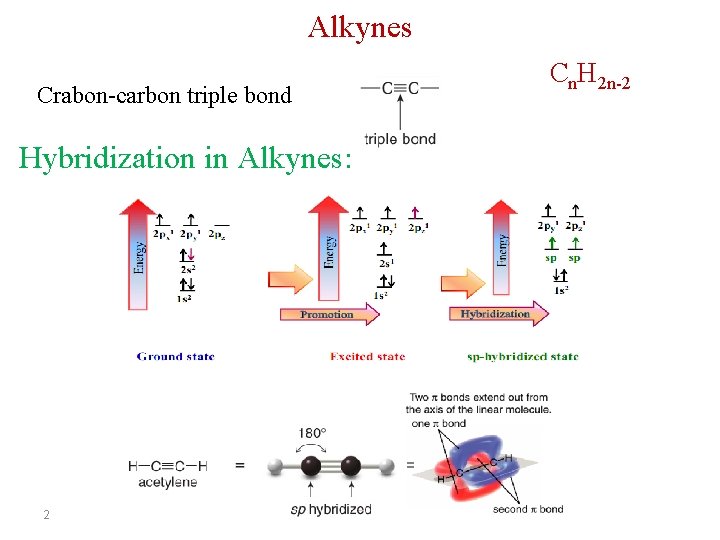
Alkynes Cn. H 2 n-2 Crabon-carbon triple bond Hybridization in Alkynes: = 2
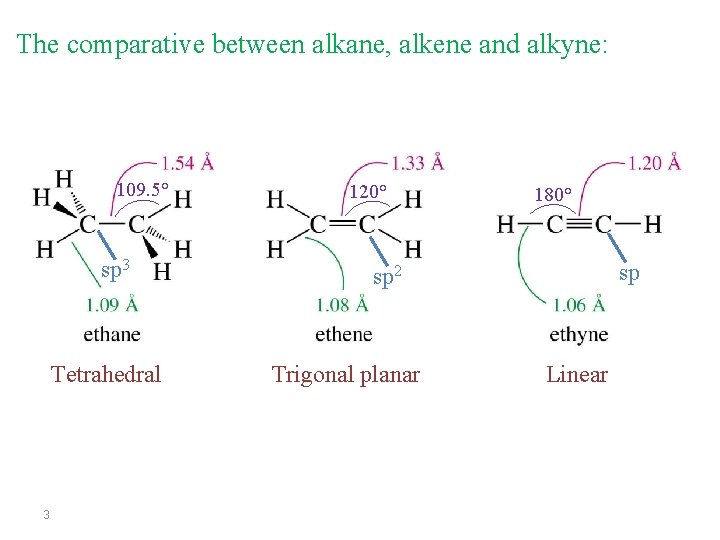
The comparative between alkane, alkene and alkyne: 109. 5° sp 3 Tetrahedral 3 120° 180° sp sp 2 Trigonal planar Linear
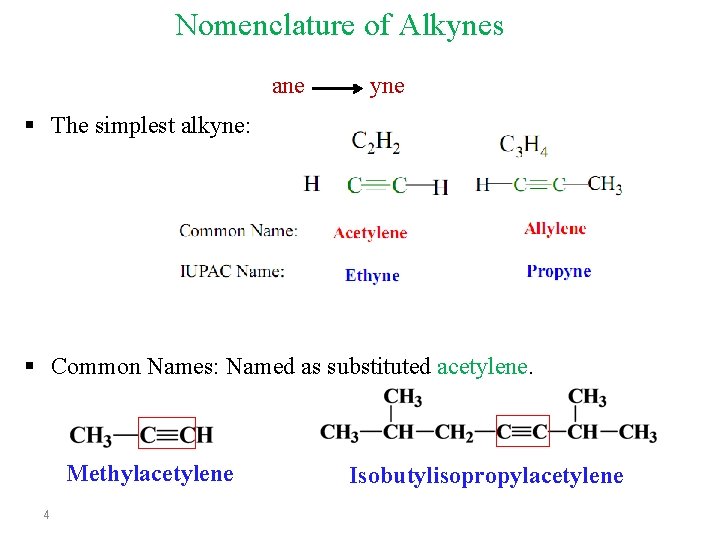
Nomenclature of Alkynes ane yne § The simplest alkyne: § Common Names: Named as substituted acetylene. Methylacetylene 4 Isobutylisopropylacetylene
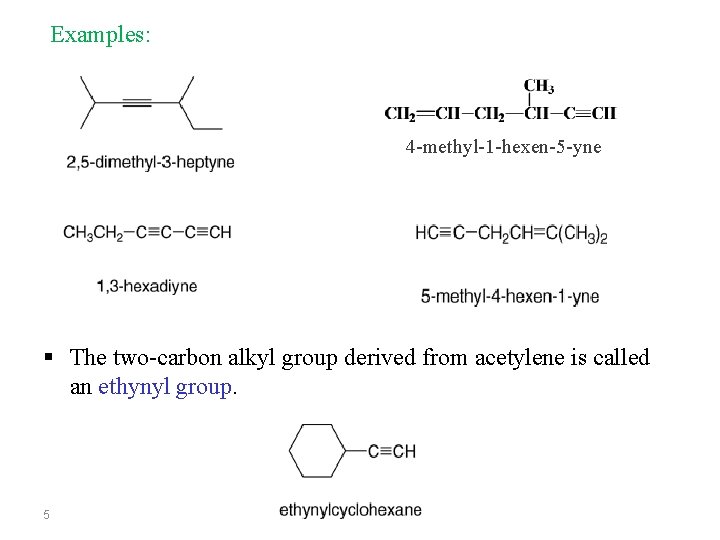
Examples: 4 -methyl-1 -hexen-5 -yne § The two-carbon alkyl group derived from acetylene is called an ethynyl group. 5

Terminal and Internal alkynes § Terminal alkynes are more acidic than other hydrocarbons. Physical Properties C 1 -C 4 gases C 5 -C 18 liquids More than C 18 solids § Alkynes are nonpolar compounds. Thus alkynes are soluble in the nonpolar solvents such as carbon tetrachloride (CCl 4) and benzene (C 6 H 6), but they are insoluble in polar solvents such as water. 6

Preparation of Alkynes 1 - Industrial From Calcium carbide 7
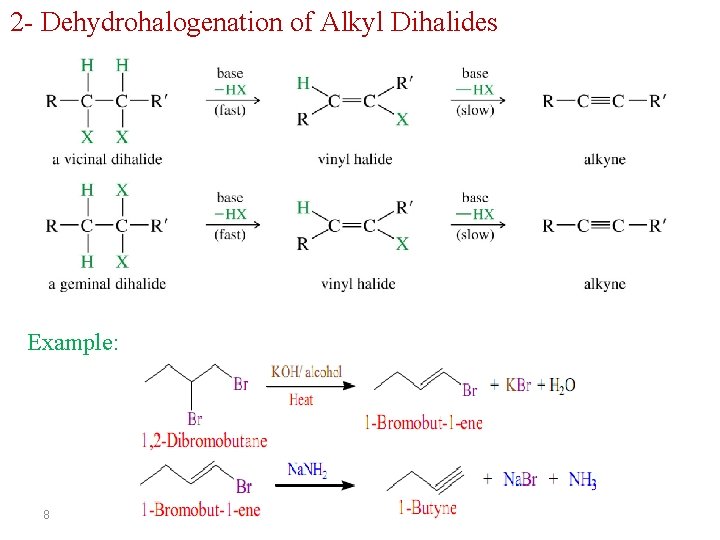
2 - Dehydrohalogenation of Alkyl Dihalides Example: 8
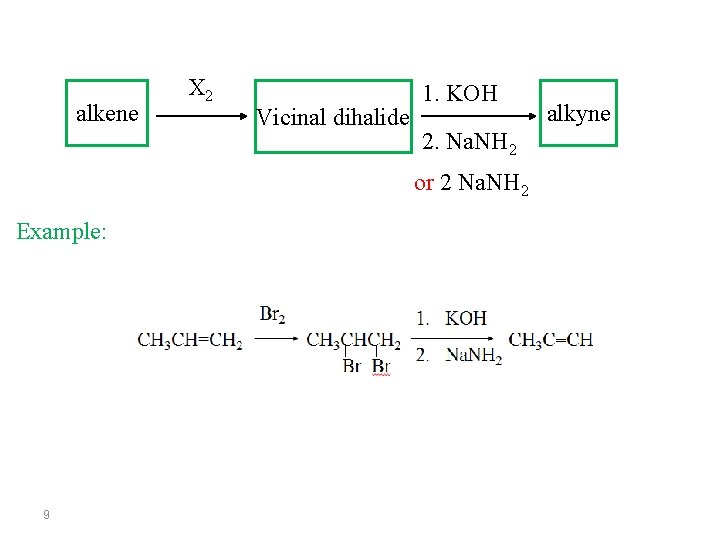
alkene X 2 Vicinal dihalide 1. KOH 2. Na. NH 2 or 2 Na. NH 2 Example: 9 alkyne
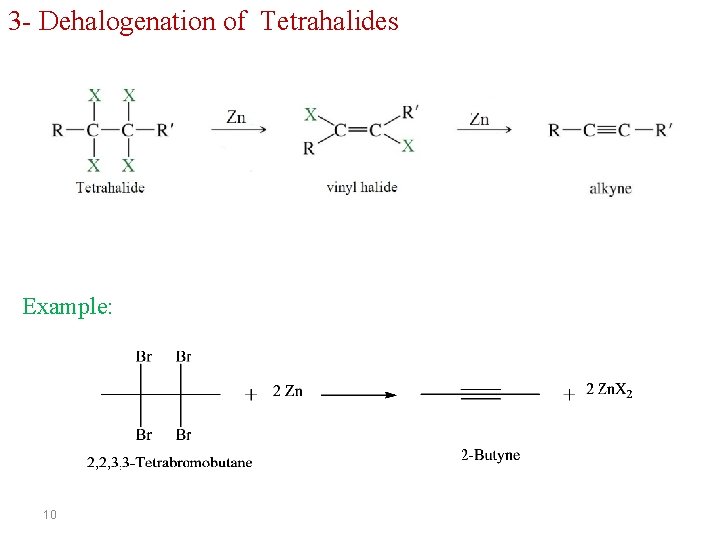
3 - Dehalogenation of Tetrahalides Example: 10
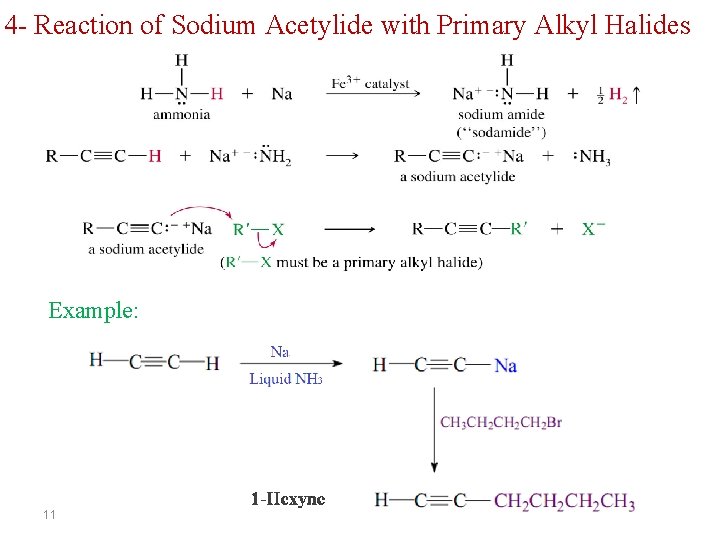
4 - Reaction of Sodium Acetylide with Primary Alkyl Halides Example: 11
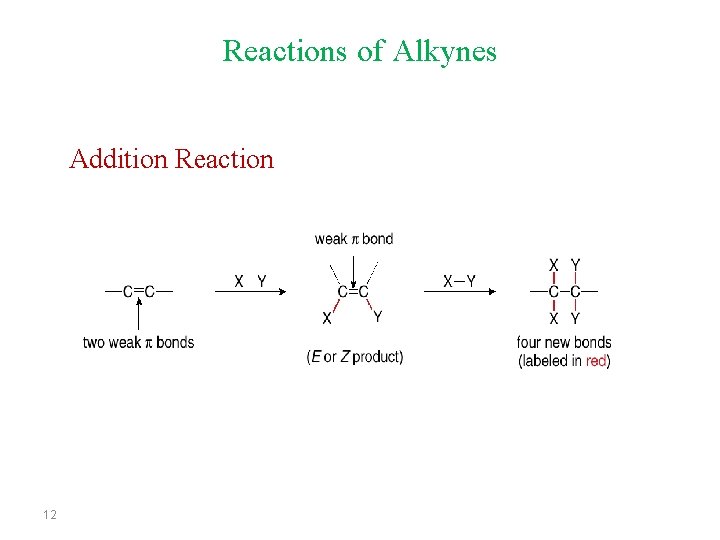
Reactions of Alkynes Addition Reaction 12
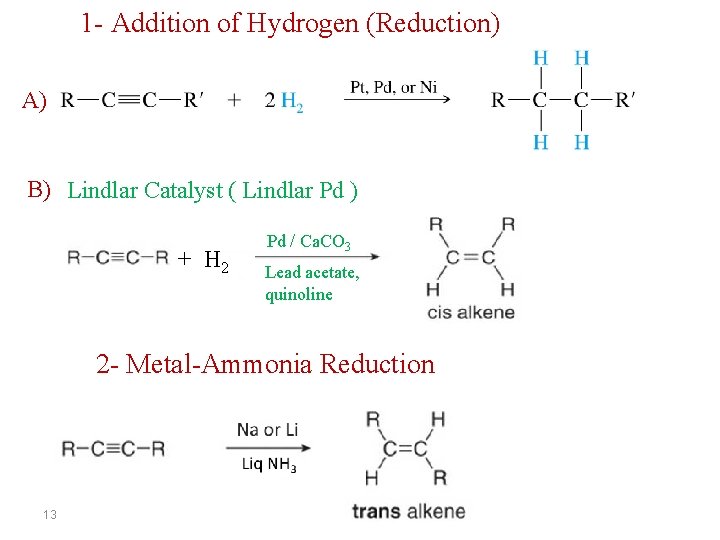
1 - Addition of Hydrogen (Reduction) A) B) Lindlar Catalyst ( Lindlar Pd ) + H 2 Pd / Ca. CO 3 Lead acetate, quinoline 2 - Metal-Ammonia Reduction 13
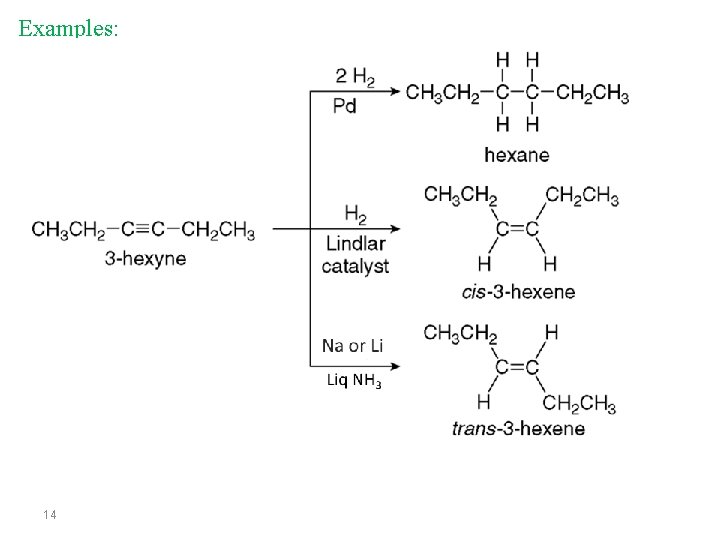
Examples: 14
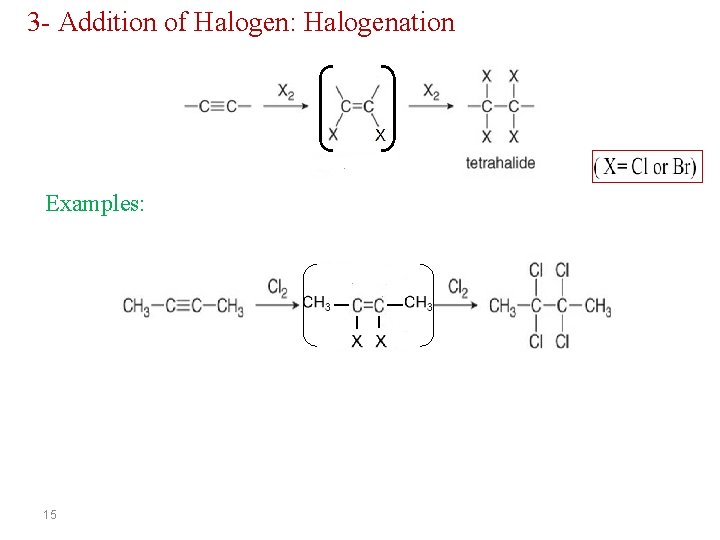
3 - Addition of Halogen: Halogenation Examples: 15
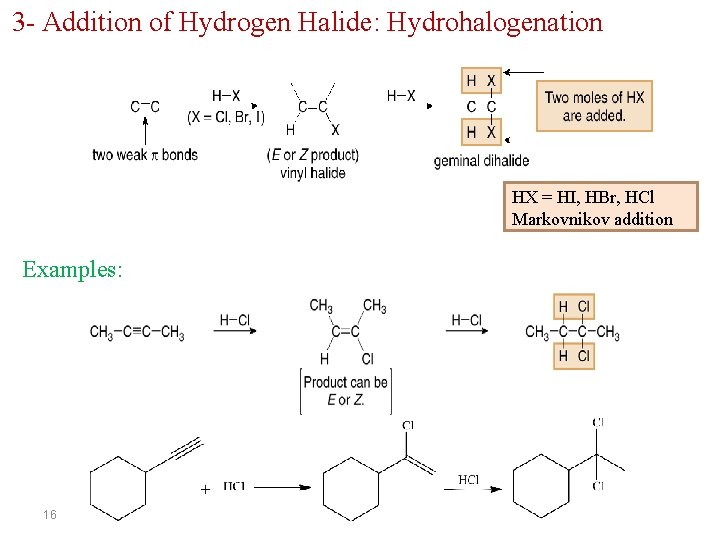
3 - Addition of Hydrogen Halide: Hydrohalogenation HX = HI, HBr, HCl Markovnikov addition Examples: 16
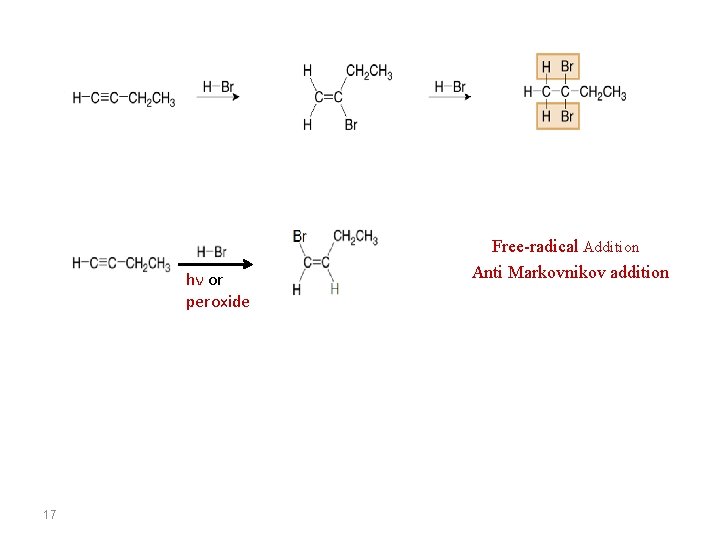
hν or peroxide 17 Free-radical Addition Anti Markovnikov addition
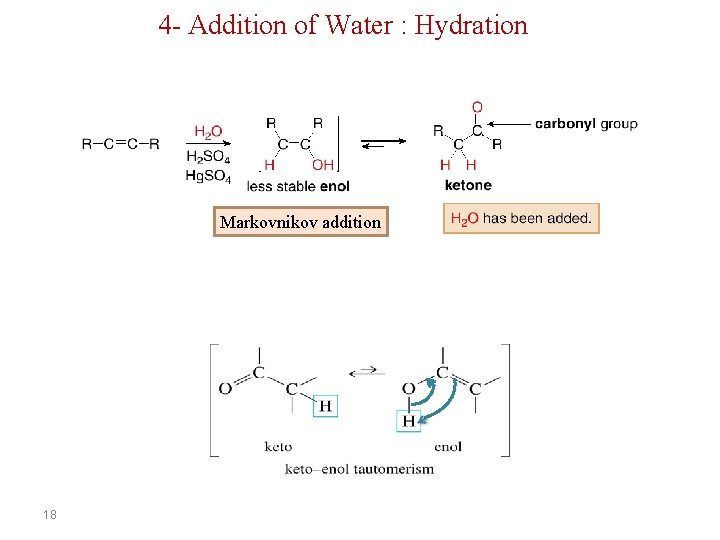
4 - Addition of Water : Hydration Markovnikov addition 18
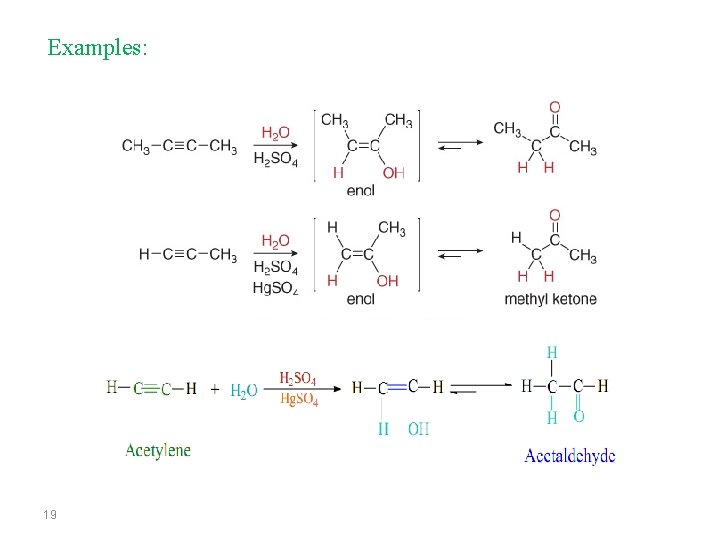
Examples: 19
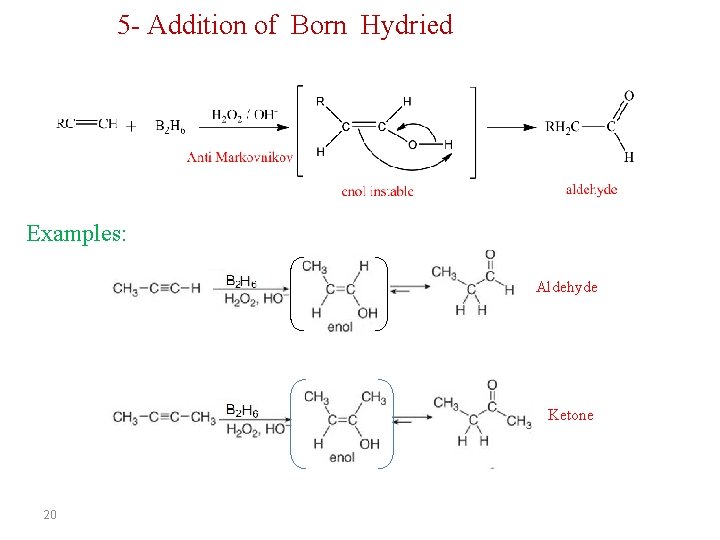
5 - Addition of Born Hydried Examples: Aldehyde Ketone 20
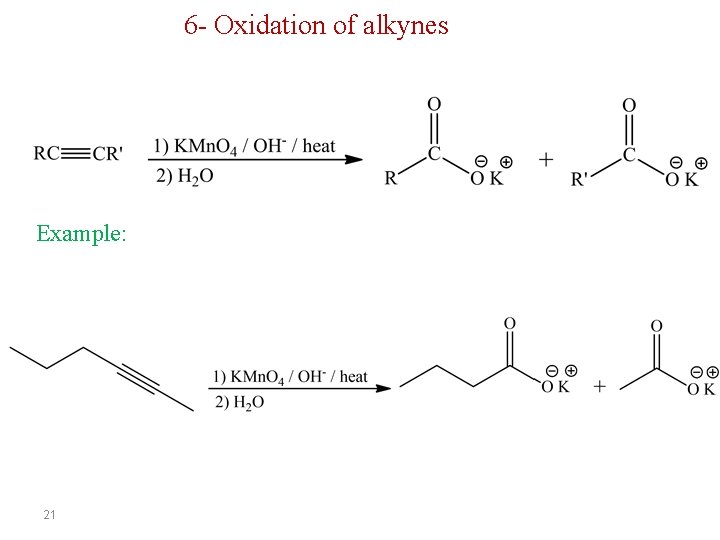
6 - Oxidation of alkynes Example: 21
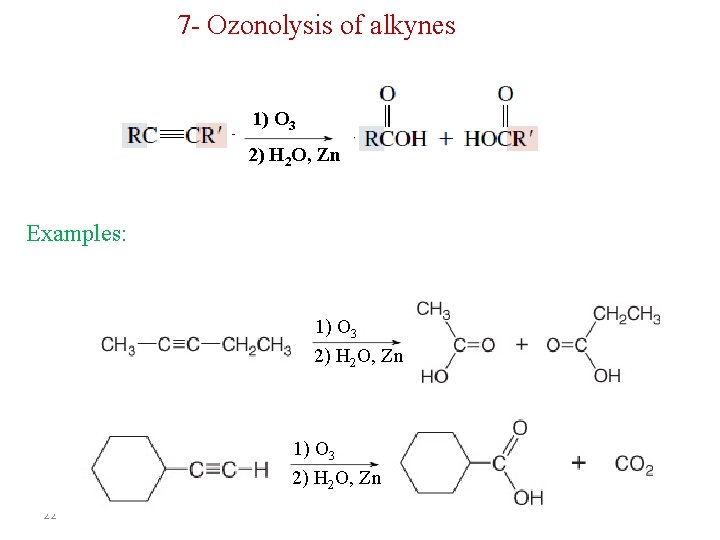
7 - Ozonolysis of alkynes 1) O 3 2) H 2 O, Zn Examples: 1) O 3 2) H 2 O, Zn 22
- Slides: 22