24 6 Sources of Phenols Phenol is an
















- Slides: 34

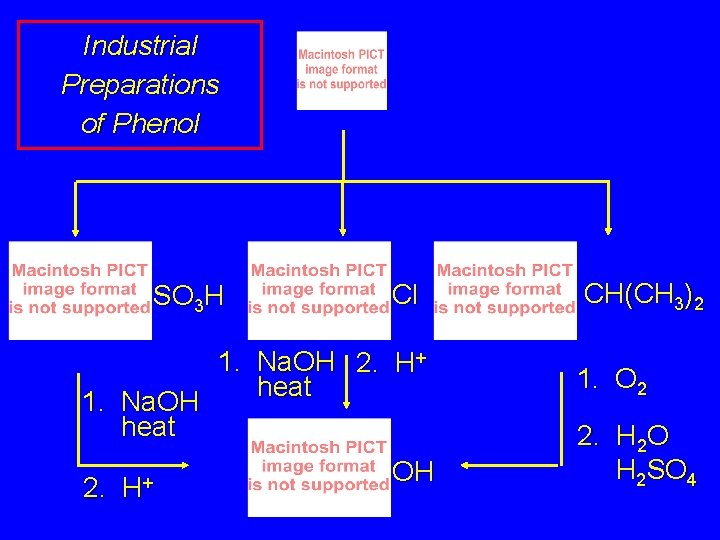
24. 6 Sources of Phenols Phenol is an important industrial chemical. Major use is in phenolic resins for adhesives and plastics. Annual U. S. production is about 4 billion pounds per year.

Industrial Preparations of Phenol SO 3 H Cl 1. Na. OH 2. H+ heat 1. Na. OH heat 2. H+ OH CH(CH 3)2 1. O 2 2. H 2 O H 2 SO 4

Laboratory Synthesis of Phenols from arylamines via diazonium ions O 2 N NH 2 1. Na. NO 2, H 2 SO 4, H 2 O O 2 N OH 2. H 2 O, heat (81 -86%)

24. 7 Naturally Occurring Phenols Many phenols occur naturally

Example: Thymol OH CH 3 CH(CH 3)2 Thymol (major constituent of oil of thyme)

Example: 2, 5 -Dichlorophenol OH Cl Cl 2, 5 -Dichlorophenol (from defensive secretion of a species of grasshopper)

24. 8 Reactions of Phenols: Electrophilic Aromatic Substitution Hydroxyl group strongly activates the ring toward electrophilic aromatic substitution

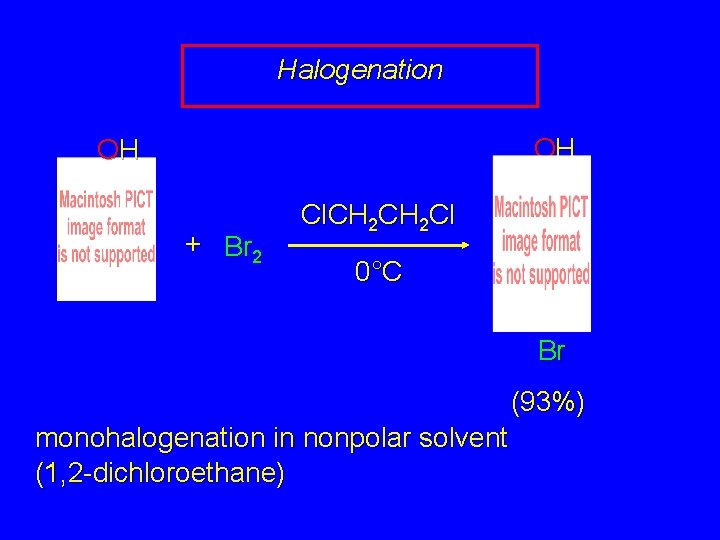
Electrophilic Aromatic Substitution in Phenols Halogenation Nitrosation Sulfonation Friedel-Crafts Alkylation Friedel-Crafts Acylation

Halogenation OH OH + Br 2 Cl. CH 2 Cl 0°C Br (93%) monohalogenation in nonpolar solvent (1, 2 -dichloroethane)

Halogenation OH OH + 3 Br 2 F H 2 O Br Br 25°C F Br (95%) multiple halogenation in polar solvent (water)

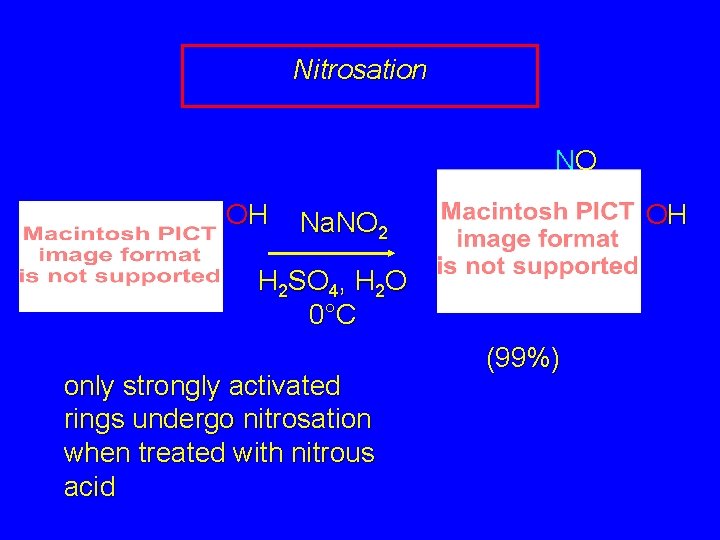
Electrophilic Aromatic Substitution in Phenols Halogenation Nitrosation Sulfonation Friedel-Crafts Alkylation Friedel-Crafts Acylation

Nitration OH OH NO 2 HNO 3 acetic acid 5°C CH 3 OH group controls regiochemistry CH 3 (73 -77%)

Electrophilic Aromatic Substitution in Phenols Halogenation Nitrosation Sulfonation Friedel-Crafts Alkylation Friedel-Crafts Acylation

Nitrosation NO OH OH Na. NO 2 H 2 SO 4, H 2 O 0°C only strongly activated rings undergo nitrosation when treated with nitrous acid (99%)

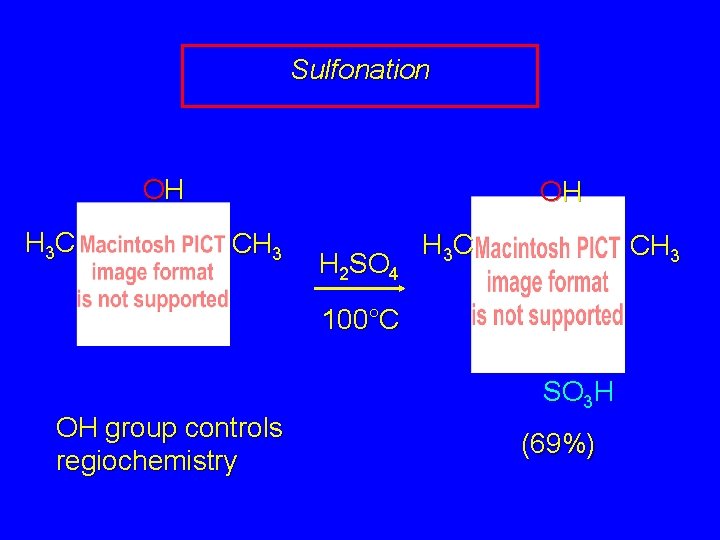
Electrophilic Aromatic Substitution in Phenols Halogenation Nitrosation Sulfonation Friedel-Crafts Alkylation Friedel-Crafts Acylation

Sulfonation OH H 3 C OH CH 3 H 2 SO 4 H 3 C CH 3 100°C OH group controls regiochemistry SO 3 H (69%)

Electrophilic Aromatic Substitution in Phenols Halogenation Nitrosation Sulfonation Friedel-Crafts Alkylation Friedel-Crafts Acylation

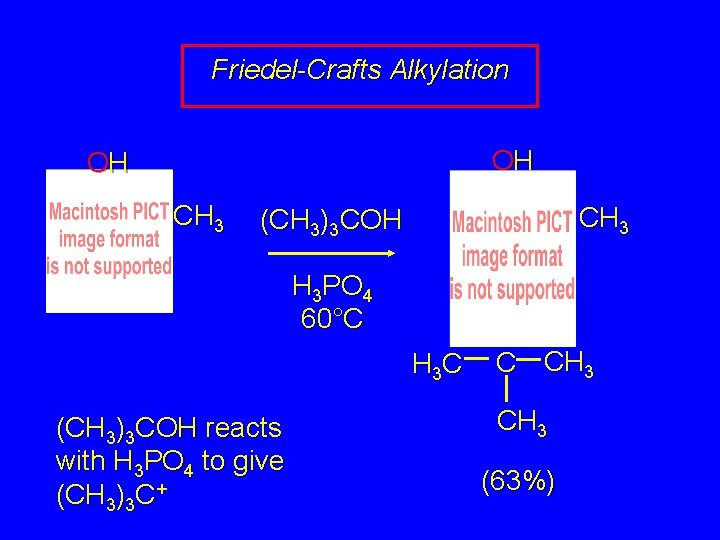
Friedel-Crafts Alkylation OH OH CH 3 (CH 3)3 COH H 3 PO 4 60°C H 3 C (CH 3)3 COH reacts with H 3 PO 4 to give (CH 3)3 C+ C CH 3 (63%)

Electrophilic Aromatic Substitution in Phenols Halogenation Nitrosation Sulfonation Friedel-Crafts Alkylation Friedel-Crafts Acylation

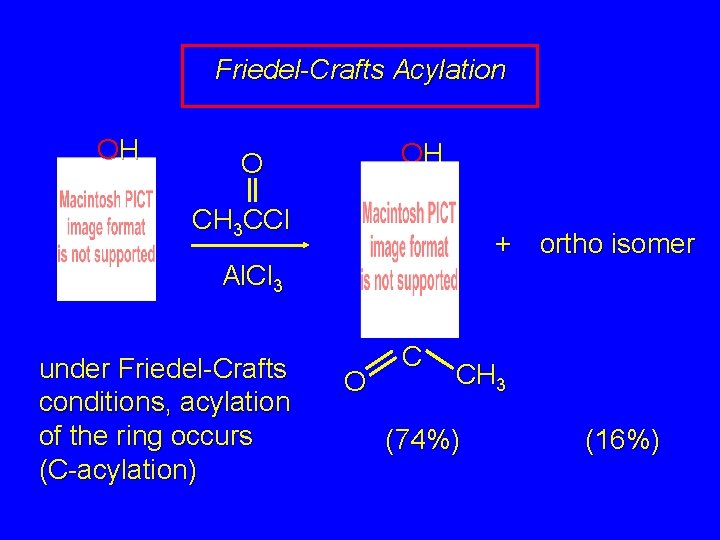
24. 9 Acylation of Phenols Acylation can take place either on the ring by electrophilic aromatic substitution or on oxygen by nucleophilic acyl substitution

Friedel-Crafts Acylation OH OH O CH 3 CCl + ortho isomer Al. Cl 3 under Friedel-Crafts conditions, acylation of the ring occurs (C-acylation) O C CH 3 (74%) (16%)

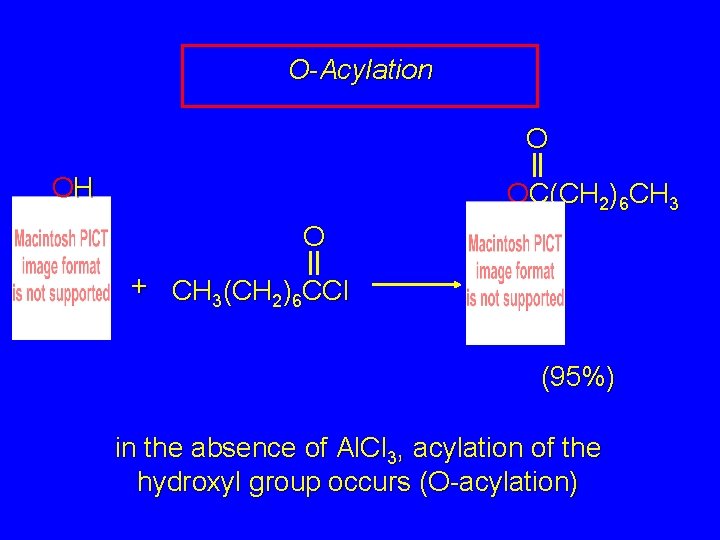
O-Acylation O OH OC(CH 2)6 CH 3 O + CH 3(CH 2)6 CCl (95%) in the absence of Al. Cl 3, acylation of the hydroxyl group occurs (O-acylation)

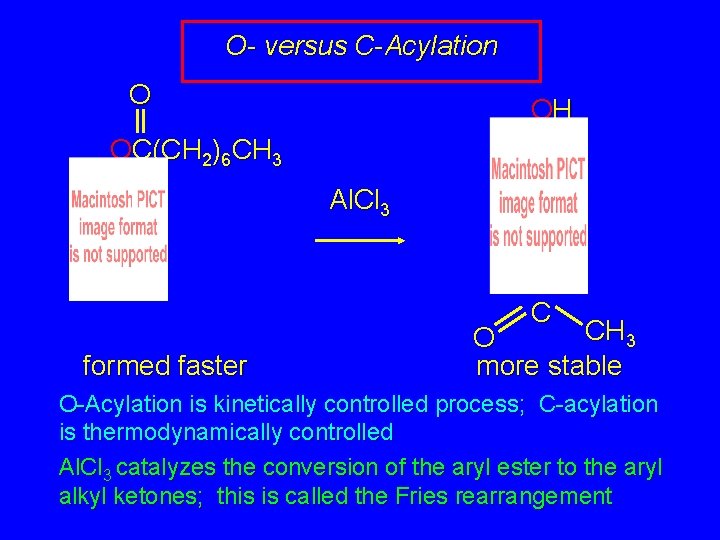
O- versus C-Acylation O OH OC(CH 2)6 CH 3 Al. Cl 3 C formed faster CH 3 O more stable O-Acylation is kinetically controlled process; C-acylation is thermodynamically controlled Al. Cl 3 catalyzes the conversion of the aryl ester to the aryl alkyl ketones; this is called the Fries rearrangement

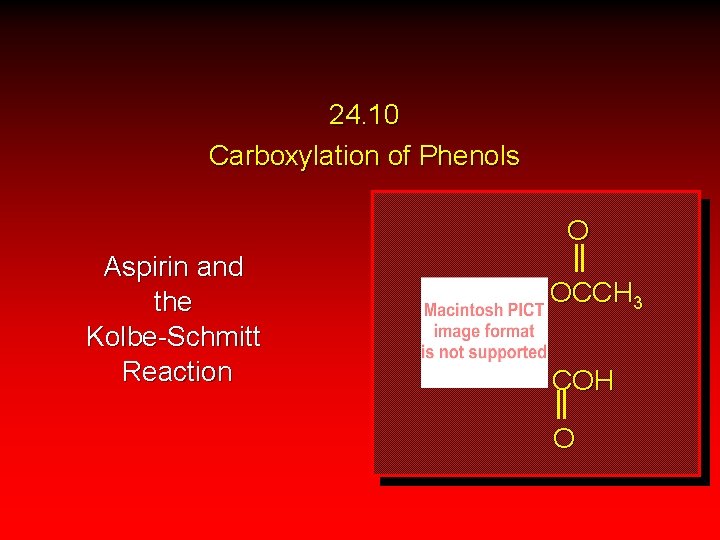
24. 10 Carboxylation of Phenols O Aspirin and the Kolbe-Schmitt Reaction OCCH 3 COH O

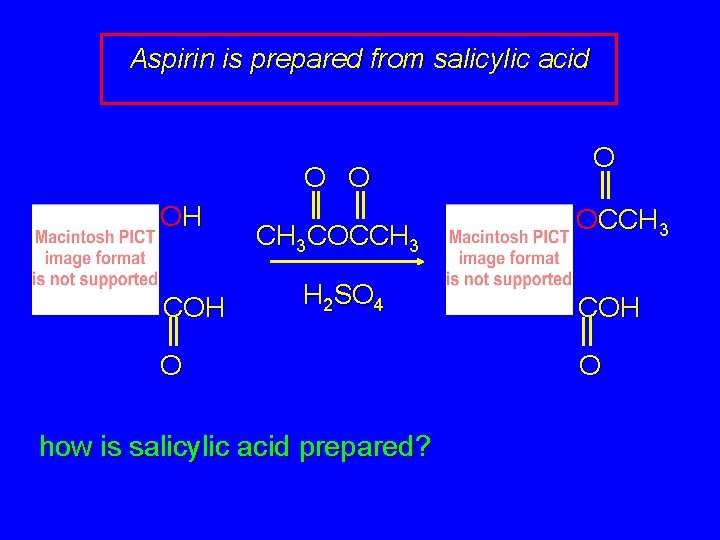
Aspirin is prepared from salicylic acid O O OH CH 3 COCCH 3 H 2 SO 4 O how is salicylic acid prepared? O OCCH 3 COH O

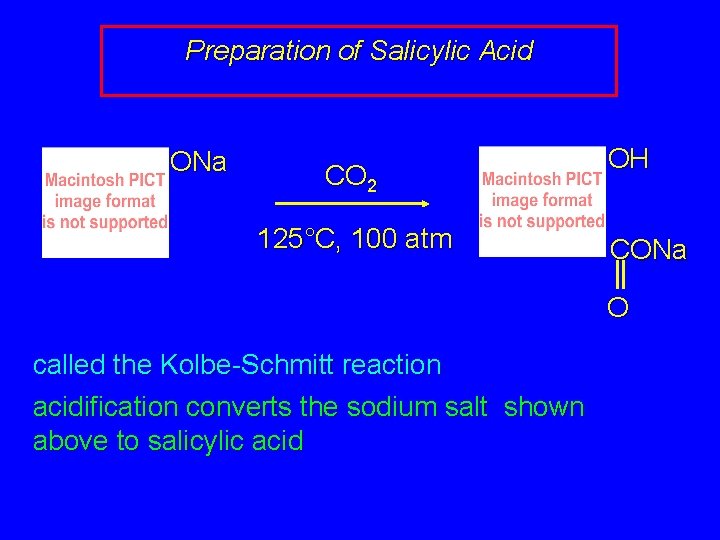
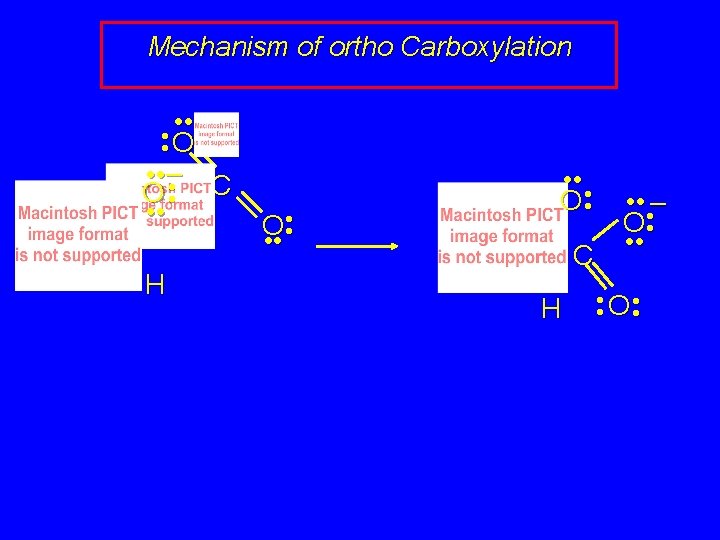
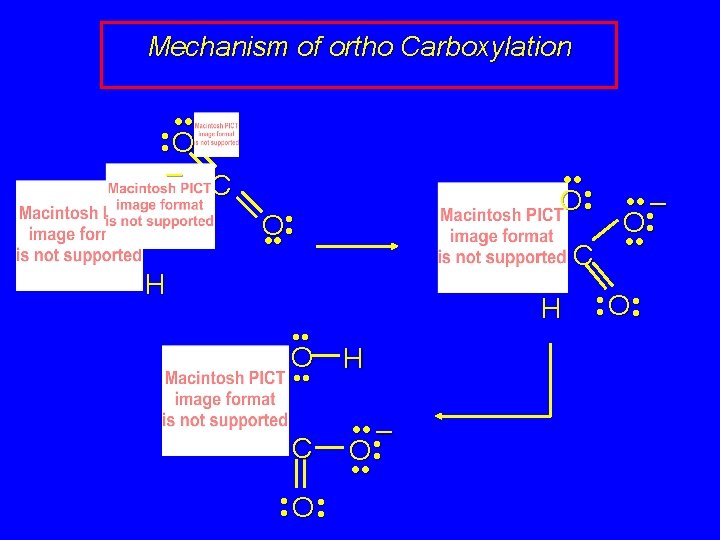
Preparation of Salicylic Acid ONa CO 2 125°C, 100 atm OH CONa O called the Kolbe-Schmitt reaction acidification converts the sodium salt shown above to salicylic acid

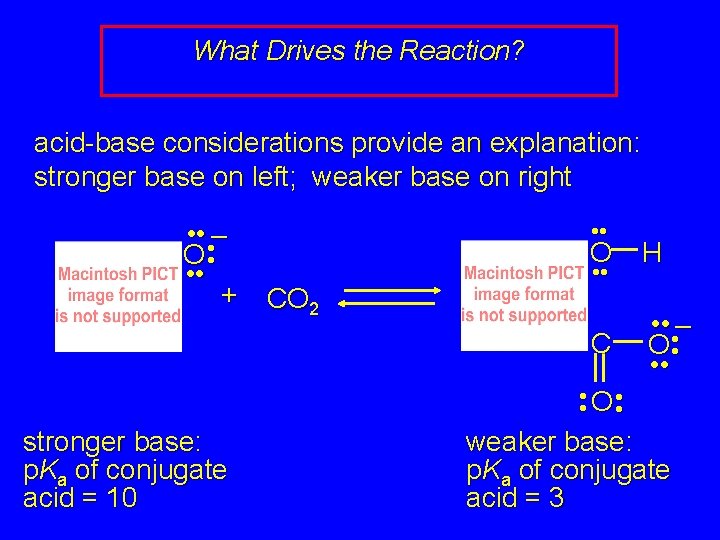
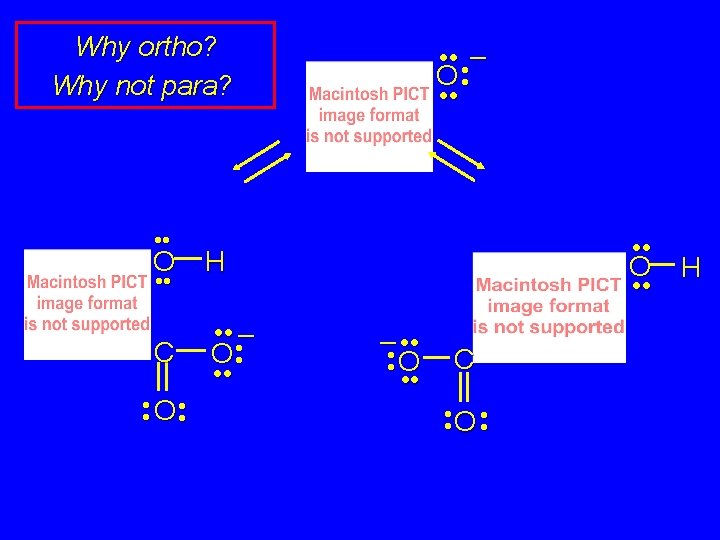
What Drives the Reaction? acid-base considerations provide an explanation: stronger base on left; weaker base on right • • • – O • • • + • • CO 2 O H C • • • – O • • • O • • stronger base: p. Ka of conjugate acid = 10 weaker base: p. Ka of conjugate acid = 3

Preparation of Salicylic Acid ONa CO 2 125°C, 100 atm OH CONa O how does carbon-carbon bond form? recall electron delocalization in phenoxide ion negative charge shared by oxygen and by the ring carbons that are ortho and para to oxygen





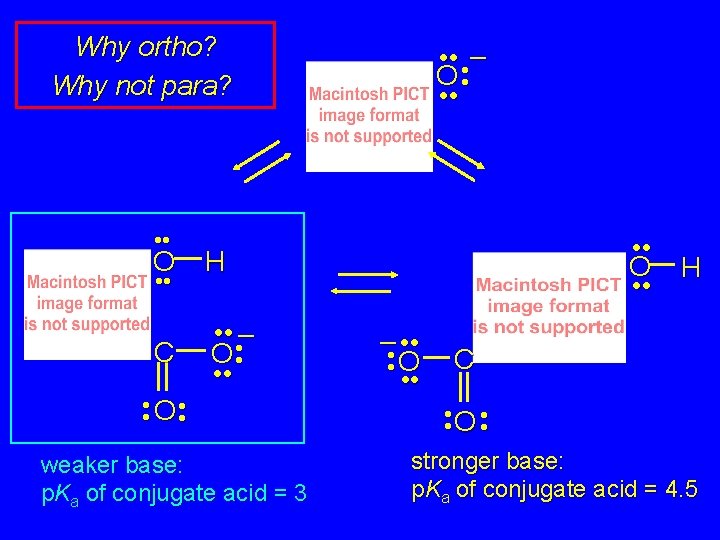
Why ortho? Why not para? • • O H C • • • – O • • • O • • weaker base: p. Ka of conjugate acid = 3 • • • – O • • • O • • – • • O • • H C • • O • • stronger base: p. Ka of conjugate acid = 4. 5

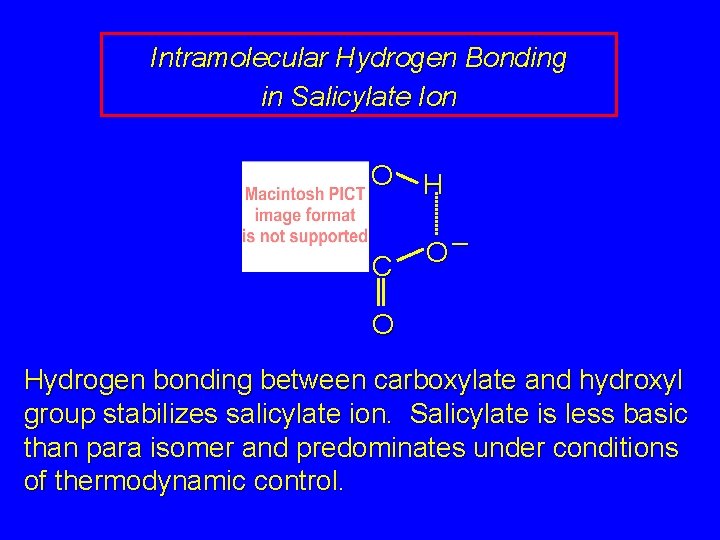
Intramolecular Hydrogen Bonding in Salicylate Ion O H C O– O Hydrogen bonding between carboxylate and hydroxyl group stabilizes salicylate ion. Salicylate is less basic than para isomer and predominates under conditions of thermodynamic control.