2282021 1 Solubility Rules 2282021 2 1 The

2/28/2021 1

Solubility Rules 2/28/2021 2

1. The common acids are soluble in water. Examples: n HCl n H 2 SO 4 2/28/2021 3

2. Most salts of the alkali metal ions (Li +1, Na+1, K+1 and Rb+1) and ammonium ion (NH 4+1) are soluble in water. 2/28/2021 4

Examples: n Li. Cl n Na. Cl n NH 4 I 2/28/2021 5

3. Most nitrate (NO 3 -1), acetate (C 2 H 3 O 2 -1), and chlorate (Cl. O 3 -1) salts are soluble. 2/28/2021 6
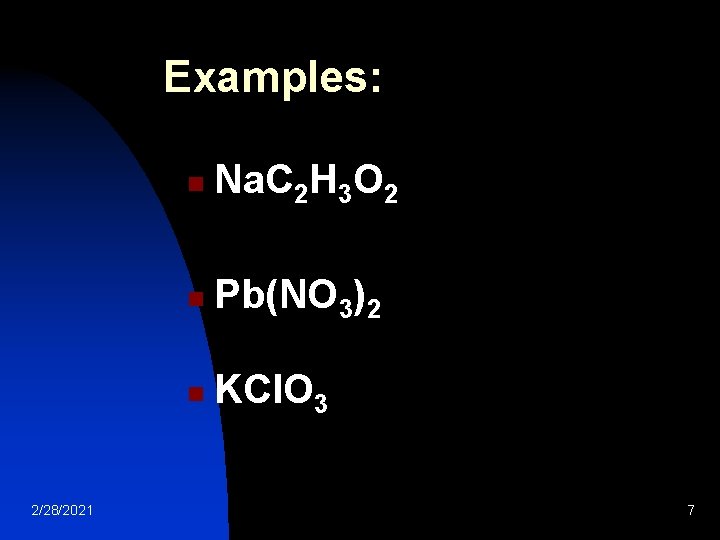
Examples: 2/28/2021 n Na. C 2 H 3 O 2 n Pb(NO 3)2 n KCl. O 3 7

4. Most Cl-1, Br-1, and I-1 salts are soluble. Exceptions are salts with Hg 2+2, Pb+2, Ag+1, and Cu+2. 2/28/2021 8

Examples: Soluble salts: n n KI Na. Br 2/28/2021 Insoluble salts: n Pb. I 2 n Ag. Cl 9

5. Sulfates (SO 4 n n -2) Most salts are soluble Exceptions are salts with Ba+2, Pb+2, and Ca+2 2/28/2021 10

Examples: Soluble salts: Insoluble salts: n Na 2 SO 4 n Ba. SO 4 n (NH 4)2 SO 4 n Ca. SO 4 2/28/2021 11

6. Hydroxides (OH-1) n n 2/28/2021 Most are insoluble Exceptions are those of group 1 cations and group 2 cations from Mg down 12

Examples: Soluble salts: Insoluble salts: n Na. OH n Cu(OH)2 n Ca(OH)2 n Pb(OH)2 2/28/2021 13

7. 3 CO 3 n n -2, -2 S , and PO 4 - Insoluble Except salts of group 1 cations and NH 4+1 2/28/2021 14

Examples: Soluble salts: Insoluble salts: n Na 2 CO 3 n Ca. CO 3 n K 2 CO 3 n Pb. CO 3 2/28/2021 15

Complete self check exercises a-c on page 190 2/28/2021 16

Answers: n a. Ba(NO 3)2 (aq) + 2 Na. Cl (aq) 2 Na. NO 3(aq) + Ba. Cl 2 (aq) No precipitation 2/28/2021 17
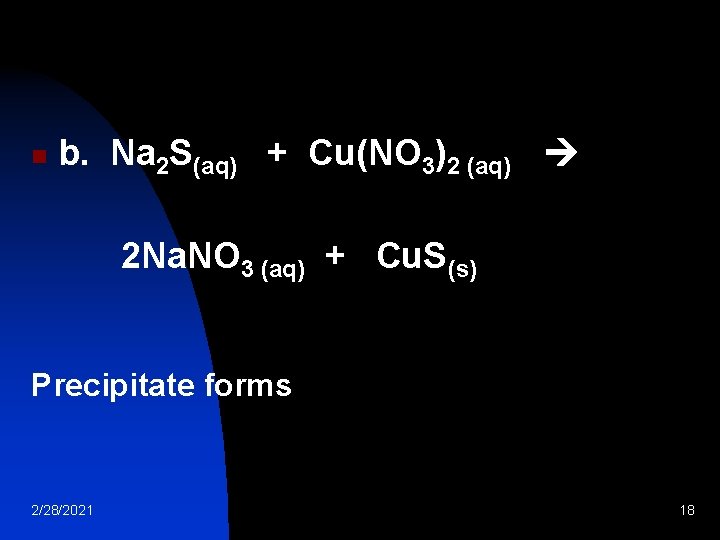
n b. Na 2 S(aq) + Cu(NO 3)2 (aq) 2 Na. NO 3 (aq) + Cu. S(s) Precipitate forms 2/28/2021 18
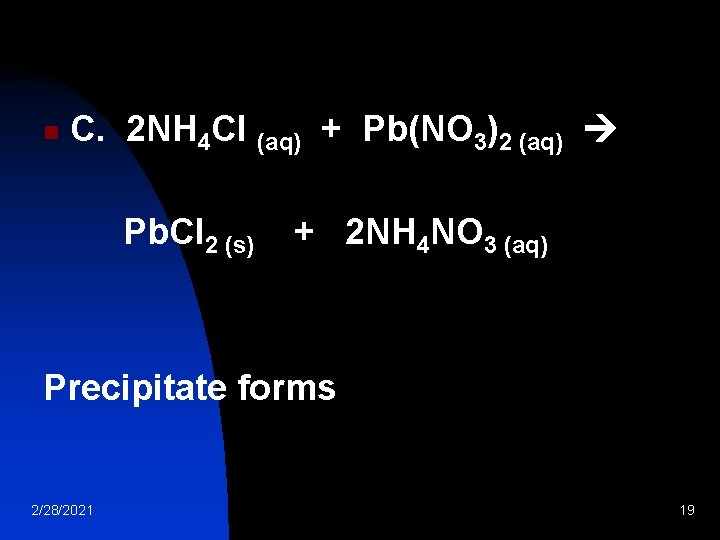
n C. 2 NH 4 Cl (aq) + Pb(NO 3)2 (aq) Pb. Cl 2 (s) + 2 NH 4 NO 3 (aq) Precipitate forms 2/28/2021 19
- Slides: 19