22 4 Measures of Amine Basicity Measures of

22. 4 Measures of Amine Basicity

Measures of Basicity The basicity of amines may be measured by: 1) Kb 2) p. Kb 3) Ka of conjugate acid 4) p. Ka of conjugate acid

Basicity Constant (Kb) and p. Kb Kb is the equilibrium constant for the reaction: R 3 N • • + H + R 3 N • • OH • • Kb = [R 3 NH+][HO–] [R 3 N] and p. Kb = - log Kb – • • H + • • OH • •

Ka and p. Ka of Conjugate Acid Ka is the equilibrium constant for the dissociation of the conjugate acid of the amine: + R 3 N • • + H+ H Ka = [R 3 N][H+] [R 3 NH+] and p. Ka = - log Ka

Relationships between acidity and basicity constants Ka Kb = 10 -14 p. Ka + p. Kb = 14

22. 5 Basicity of Amines

Effect of Structure on Basicity 1. Alkylamines are slightly stronger bases than ammonia.
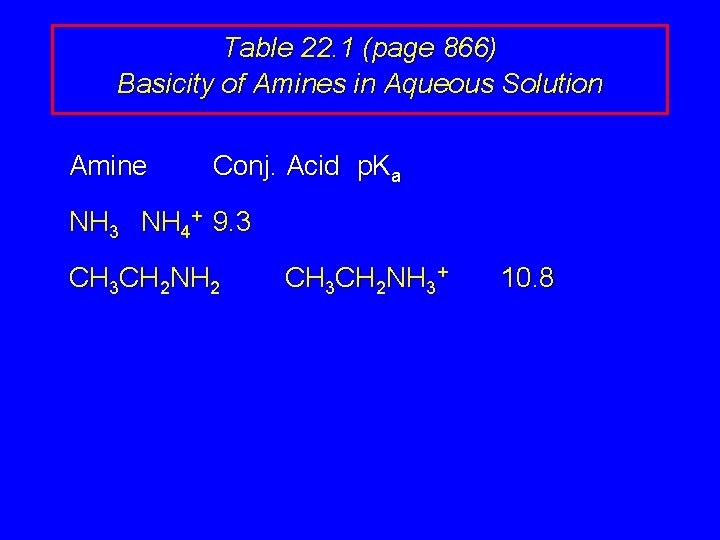
Table 22. 1 (page 866) Basicity of Amines in Aqueous Solution Amine Conj. Acid p. Ka NH 3 NH 4+ 9. 3 CH 3 CH 2 NH 2 CH 3 CH 2 NH 3+ 10. 8
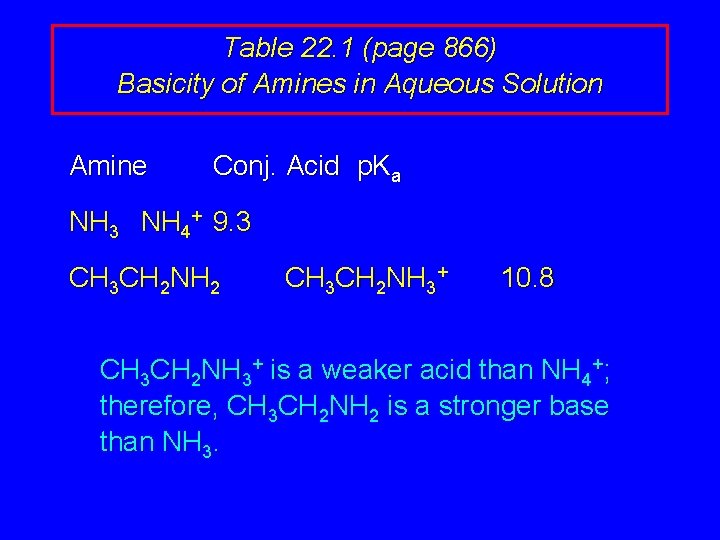
Table 22. 1 (page 866) Basicity of Amines in Aqueous Solution Amine Conj. Acid p. Ka NH 3 NH 4+ 9. 3 CH 3 CH 2 NH 2 CH 3 CH 2 NH 3+ 10. 8 CH 3 CH 2 NH 3+ is a weaker acid than NH 4+; therefore, CH 3 CH 2 NH 2 is a stronger base than NH 3.

Effect of Structure on Basicity 1. Alkylamines are slightly stronger bases than ammonia. 2. Alkylamines differ very little in basicity.
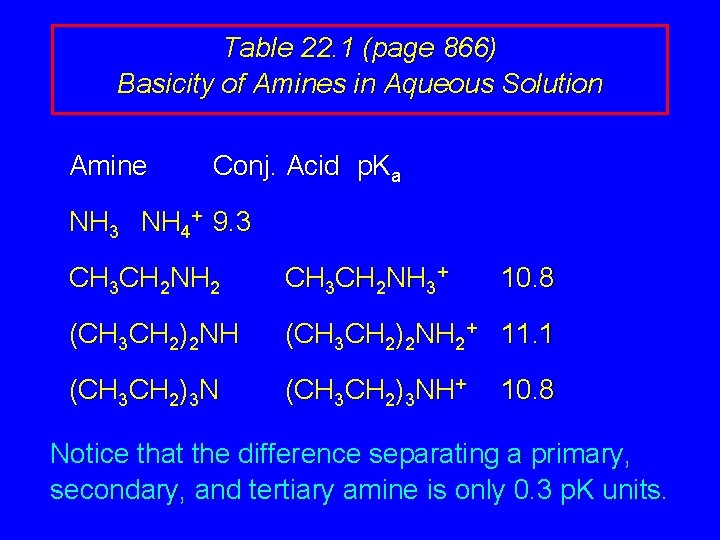
Table 22. 1 (page 866) Basicity of Amines in Aqueous Solution Amine Conj. Acid p. Ka NH 3 NH 4+ 9. 3 CH 3 CH 2 NH 2 CH 3 CH 2 NH 3+ 10. 8 (CH 3 CH 2)2 NH 2+ 11. 1 (CH 3 CH 2)3 NH+ 10. 8 Notice that the difference separating a primary, secondary, and tertiary amine is only 0. 3 p. K units.

Effect of Structure on Basicity 1. Alkylamines are slightly stronger bases than ammonia. 2. Alkylamines differ very little in basicity. 3. Arylamines are much weaker bases than ammonia.
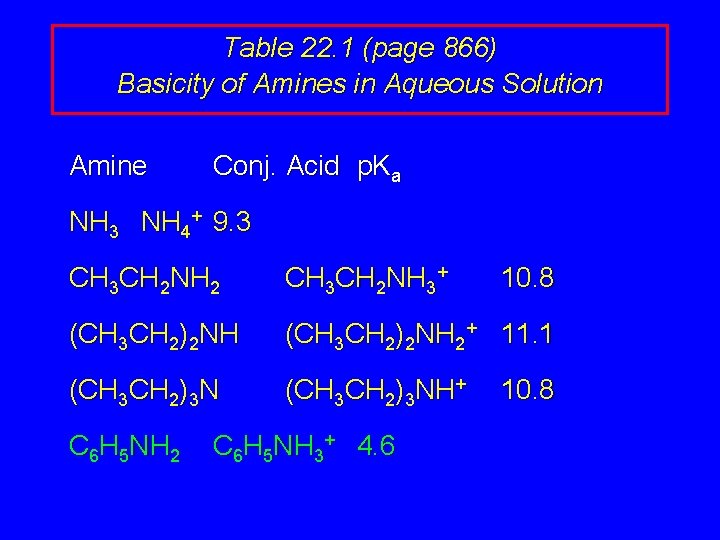
Table 22. 1 (page 866) Basicity of Amines in Aqueous Solution Amine Conj. Acid p. Ka NH 3 NH 4+ 9. 3 CH 3 CH 2 NH 2 CH 3 CH 2 NH 3+ (CH 3 CH 2)2 NH 2+ 11. 1 (CH 3 CH 2)3 NH+ C 6 H 5 NH 2 C 6 H 5 NH 3+ 4. 6 10. 8
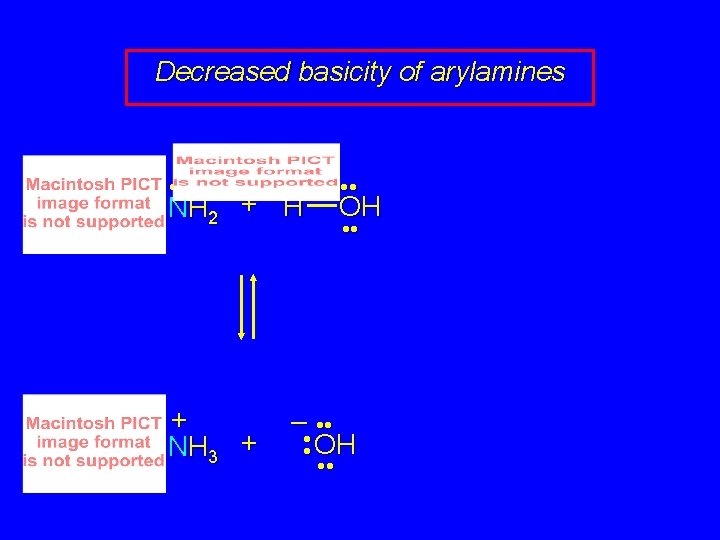
Decreased basicity of arylamines • • NH 2 + H + NH 3 + OH • • – • • OH • •
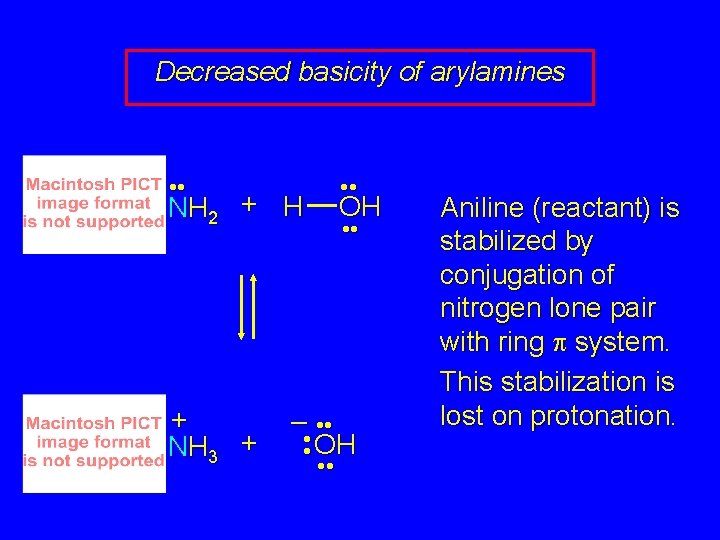
Decreased basicity of arylamines • • NH 2 + H + NH 3 + OH • • – • • OH • • Aniline (reactant) is stabilized by conjugation of nitrogen lone pair with ring p system. This stabilization is lost on protonation.

Decreased basicity of arylamines Increasing delocalization makes diphenylamine a weaker base than aniline, and triphenylamine a weaker base than diphenylamine. Kb C 6 H 5 NH 2 (C 6 H 5)2 NH (C 6 H 5)3 N 3. 8 x 10 -10 6 x 10 -14 ~10 -19

Effect of Substituents on Basicity of Arylamines 1. Alkyl groups on the ring increase basicity, but only slightly (less than 1 p. K unit).

Basicity of Arylamines X X H CH 3 p. Kb 9. 4 8. 7 NH 2 p. Ka 4. 6 5. 3 X NH 3+

Effect of Substituents on Basicity of Arylamines 1. Alkyl groups on the ring increase basicity, but only slightly (less than 1 p. K unit). 2. Electron withdrawing groups, especially ortho and/or para to amine group, decrease basicity and can have a large effect.

Basicity of Arylamines X X H CH 3 CF 3 O 2 N p. Kb 9. 4 8. 7 11. 5 13. 0 NH 2 p. Ka 4. 6 5. 3 2. 5 1. 0 X NH 3+


p-Nitroaniline – • • O • • + N • • O • • – • • NH 2 + NH 2 • • O • • – • • Lone pair on amine nitrogen is conjugated with p -nitro group—more delocalized than in aniline itself. Delocalization lost on protonation.

Effect is Cumulative Aniline is 3800 times more basic than p-nitroaniline. Aniline is ~1, 000, 000 times more basic than 2, 4 -dinitroaniline.
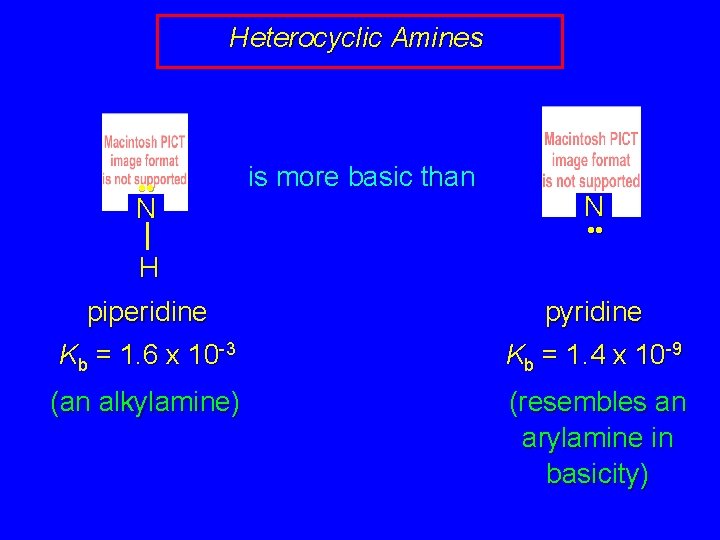
Heterocyclic Amines • • N is more basic than N • • H piperidine Kb = 1. 6 x 10 -3 pyridine Kb = 1. 4 x 10 -9 (an alkylamine) (resembles an arylamine in basicity)
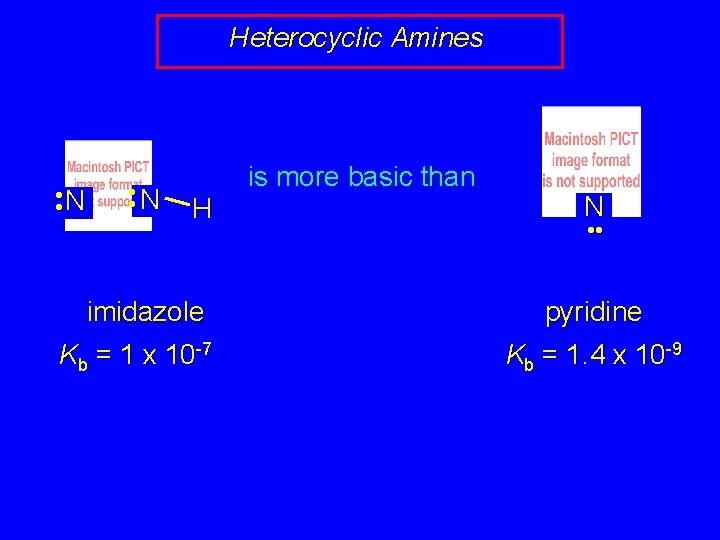
Heterocyclic Amines • • N H imidazole Kb = 1 x 10 -7 is more basic than N • • pyridine Kb = 1. 4 x 10 -9
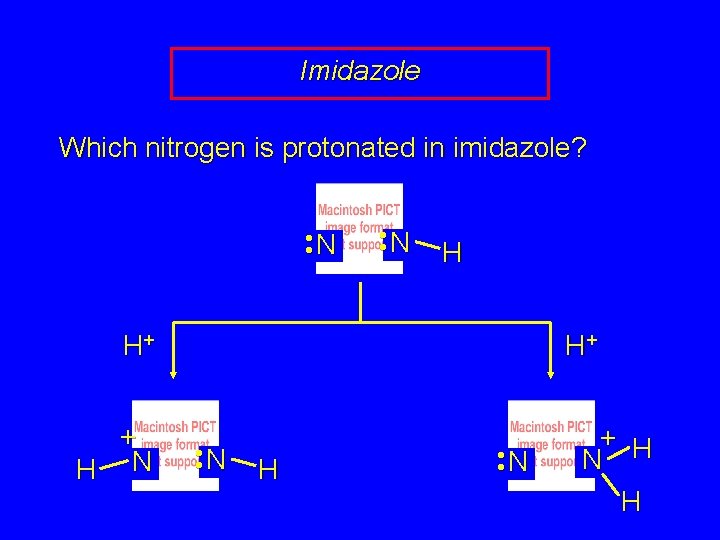
Imidazole Which nitrogen is protonated in imidazole? • • N H H+ + H N H+ • • N H • • N + H N H
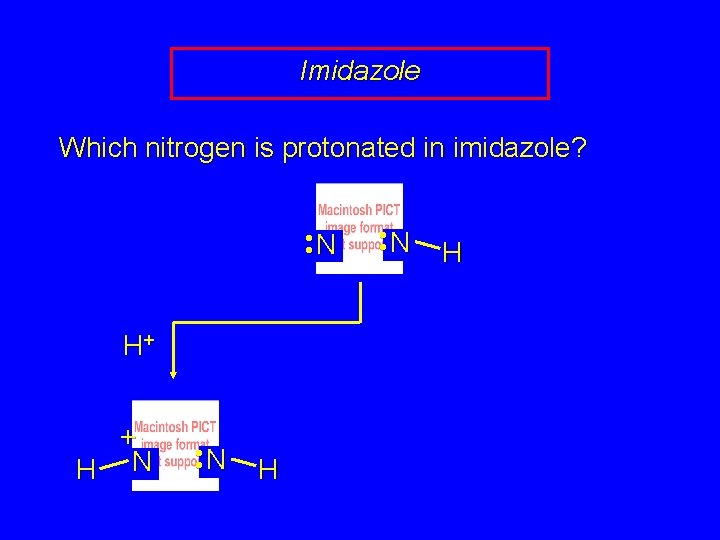
Imidazole Which nitrogen is protonated in imidazole? • • N H+ + H N • • N H
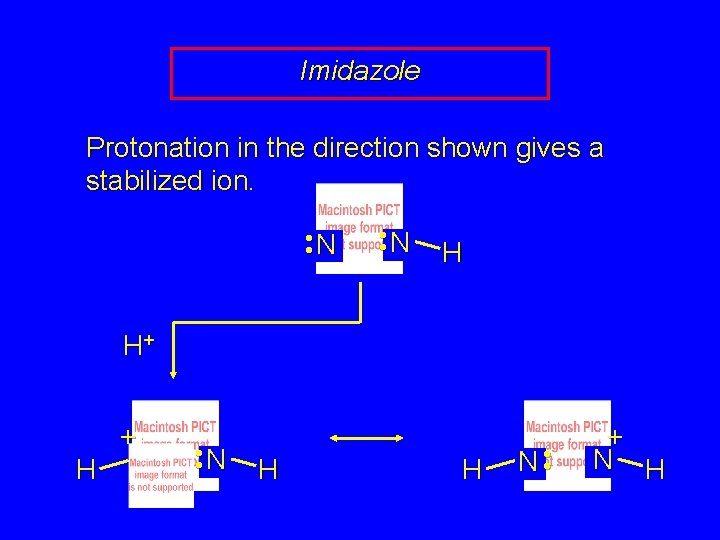
Imidazole Protonation in the direction shown gives a stabilized ion. • • N H H+ + H N • • N H H N • • + N H

22. 6 Tetraalkylammonium Salts as Phase-Transfer Catalysts

Phase-Transfer Catalysis Phase-transfer agents promote the solubility of ionic substances in nonpolar solvents. They transfer the ionic substance from an aqueous phase to a non-aqueous one. Phase-transfer agents increase the rates of reactions involving anions. The anion is relatively unsolvated and very reactive in nonpolar media compared to water or alcohols.
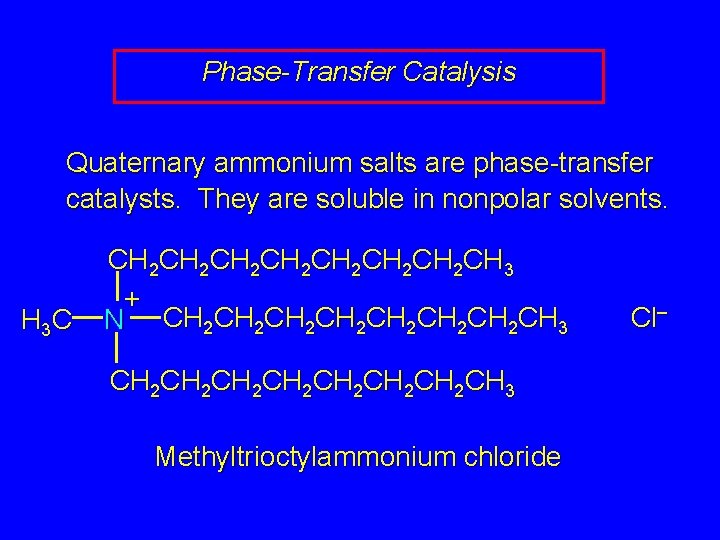
Phase-Transfer Catalysis Quaternary ammonium salts are phase-transfer catalysts. They are soluble in nonpolar solvents. H 3 C CH 2 CH 2 CH 3 + N CH 2 CH 2 CH 2 CH 2 CH 3 Methyltrioctylammonium chloride Cl–
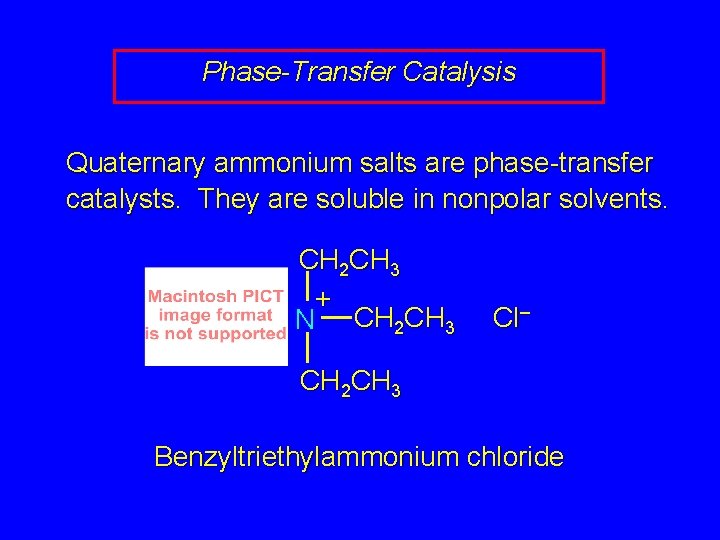
Phase-Transfer Catalysis Quaternary ammonium salts are phase-transfer catalysts. They are soluble in nonpolar solvents. CH 2 CH 3 + N CH 2 CH 3 Cl– CH 2 CH 3 Benzyltriethylammonium chloride

Example The SN 2 reaction of sodium cyanide with butyl bromide occurs much faster when benzyltriethylammonium chloride is present than when it is not. CH 3 CH 2 CH 2 Br + Na. CN benzyltriethylammonium chloride CH 3 CH 2 CH 2 CN + Na. Br
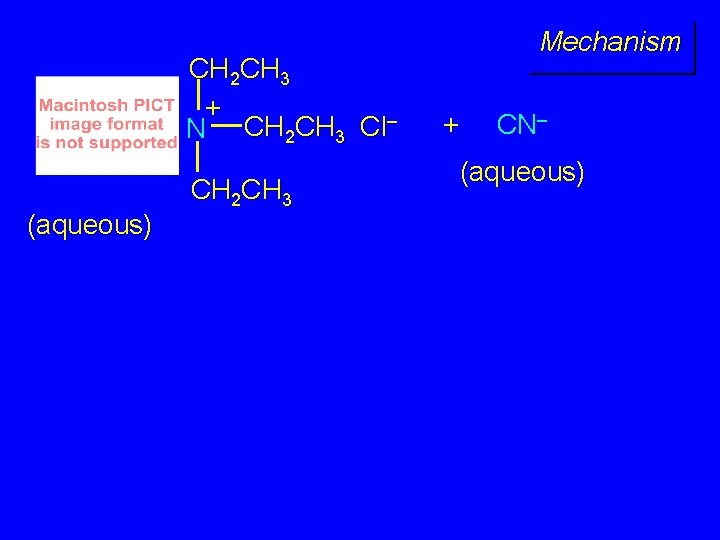
CH 2 CH 3 + N CH 2 CH 3 Cl– (aqueous) CH 2 CH 3 Mechanism + CN– (aqueous)
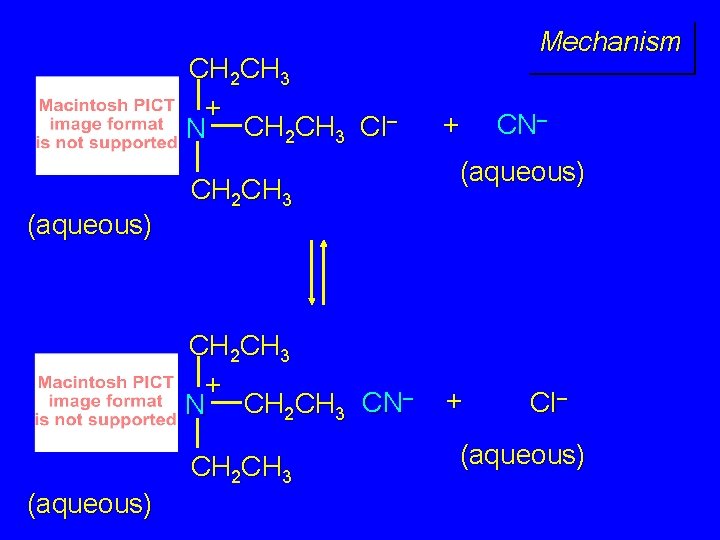
CH 2 CH 3 + N CH 2 CH 3 Cl– (aqueous) CH 2 CH 3 + N CH 2 CH 3 CN– CH 2 CH 3 (aqueous) Mechanism + CN– (aqueous) + Cl– (aqueous)
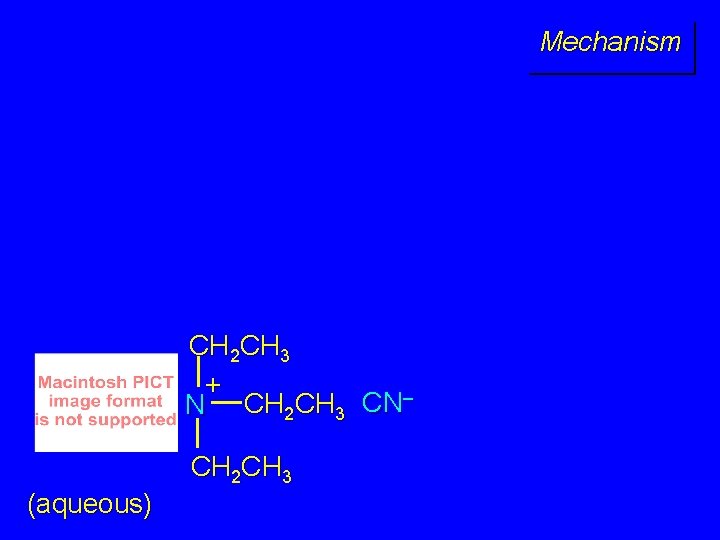
Mechanism CH 2 CH 3 + N CH 2 CH 3 CN– CH 2 CH 3 (aqueous)
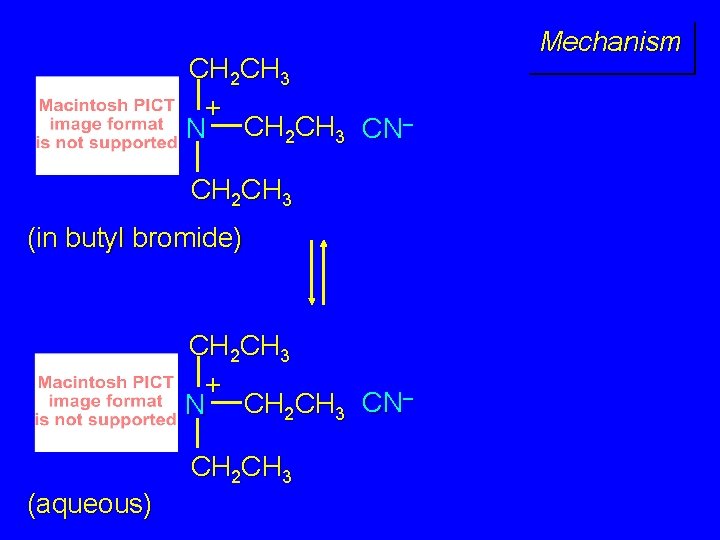
CH 2 CH 3 + N CH 2 CH 3 CN– CH 2 CH 3 (in butyl bromide) CH 2 CH 3 + N CH 2 CH 3 CN– CH 2 CH 3 (aqueous) Mechanism
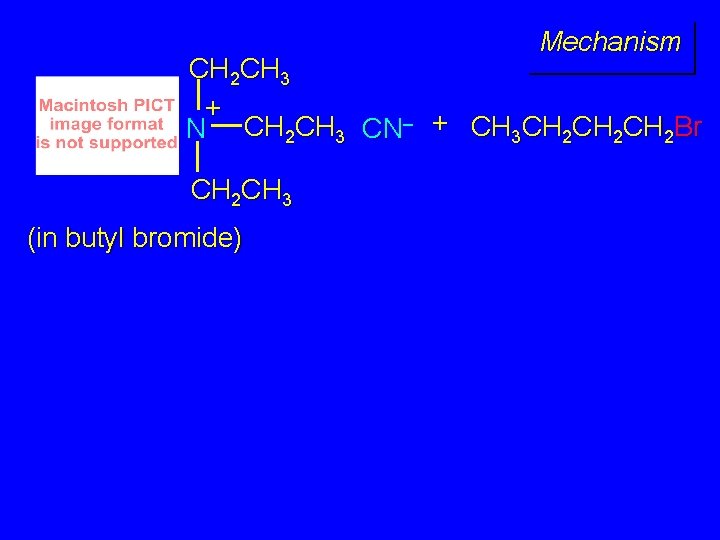
Mechanism CH 2 CH 3 + N CH 2 CH 3 CN– + CH 3 CH 2 CH 2 Br CH 2 CH 3 (in butyl bromide)
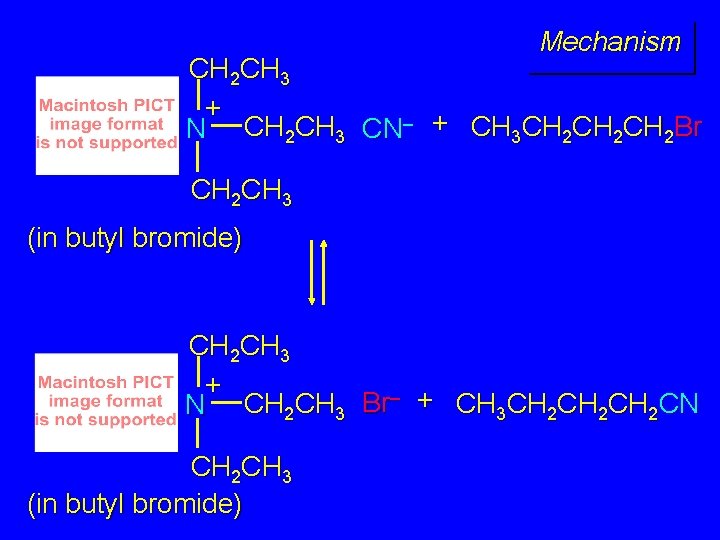
Mechanism CH 2 CH 3 + N CH 2 CH 3 CN– + CH 3 CH 2 CH 2 Br CH 2 CH 3 (in butyl bromide) CH 2 CH 3 + N CH 2 CH 3 Br– + CH 3 CH 2 CH 2 CN CH 2 CH 3 (in butyl bromide)
- Slides: 39