22 4 Hydrocarbon Rings Chapter 22 Hydrocarbon Compounds

22. 4 Hydrocarbon Rings> Chapter 22 Hydrocarbon Compounds 22. 1 Hydrocarbons 22. 2 Unsaturated Hydrocarbons 22. 3 Isomers 22. 4 Hydrocarbon Rings 22. 5 Hydrocarbons from Earth’s Crust 1 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> CHEMISTRY & YOU Does a compound have to be smelly in order to be classified as aromatic? When you hear the word aromatic, you may think of perfume or flowers. 2 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Cyclic Hydrocarbons What is the general structure of a cyclic hydrocarbon? 3 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Cyclic Hydrocarbons • Not all hydrocarbons are straight chains or branched chains. • In some hydrocarbon compounds, the carbon chain is in the form of a ring. 4 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
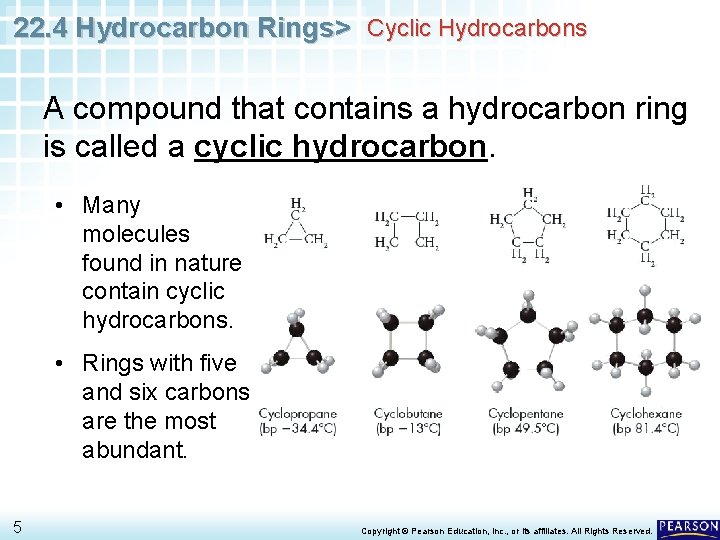
22. 4 Hydrocarbon Rings> Cyclic Hydrocarbons A compound that contains a hydrocarbon ring is called a cyclic hydrocarbon. • Many molecules found in nature contain cyclic hydrocarbons. • Rings with five and six carbons are the most abundant. 5 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Cyclic Hydrocarbons • Just as straight-chain and branched-chain alkanes can be either saturated or unsaturated, so can cyclic hydrocarbons. • A cyclic hydrocarbon that contains only single bonds, and is therefore saturated is called a cycloalkane. 6 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
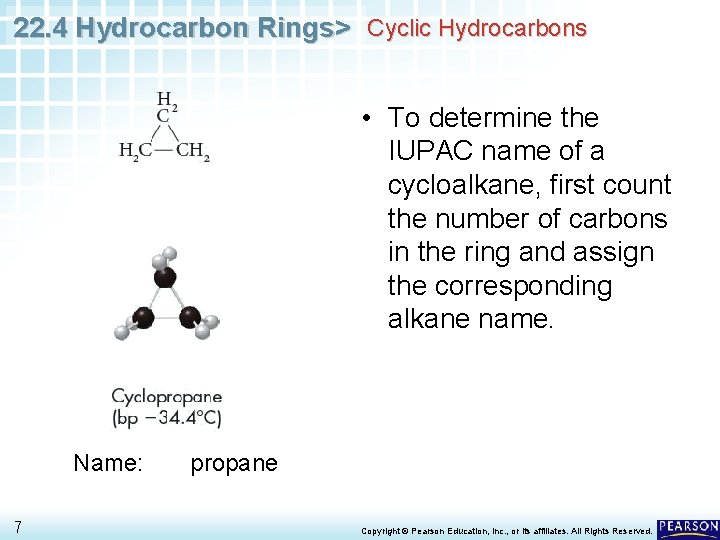
22. 4 Hydrocarbon Rings> Cyclic Hydrocarbons • To determine the IUPAC name of a cycloalkane, first count the number of carbons in the ring and assign the corresponding alkane name. Name: 7 propane Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Cyclic Hydrocarbons • To determine the IUPAC name of a cycloalkane, first count the number of carbons in the ring and assign the corresponding alkane name. Name: cyclopropane 8 • Then simply add the prefix cyclo- to the alkane name. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> How many hydrogen atoms will a cycloalkane contain in comparison to a straight chain alkane with the same number of carbons? 9 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> How many hydrogen atoms will a cycloalkane contain in comparison to a straight chain alkane with the same number of carbons? A cycloalkane will contain two fewer hydrogen atoms than a straight chain alkane with the same number of carbons because there is one additional carbon-carbon bond in the cycloalkane. 10 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons • What is the most accurate description of bonding in benzene? 11 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons • There is a class of unsaturated cyclic hydrocarbons that are responsible for the aromas of spices such as vanilla, cinnamon, cloves, and ginger. • These compounds were originally called aromatic compounds because they have distinct, pleasant aromas. • However not all compounds classified has aromatic have pleasant odors, or any odor at all. 12 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
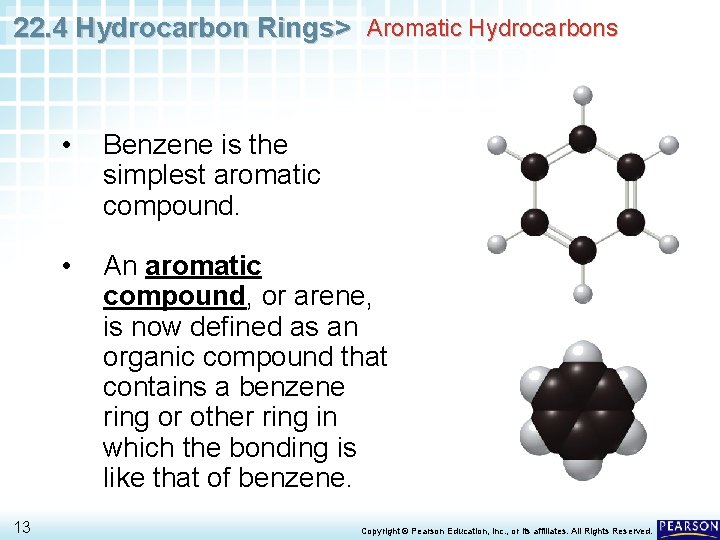
22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons 13 • Benzene is the simplest aromatic compound. • An aromatic compound, or arene, is now defined as an organic compound that contains a benzene ring or other ring in which the bonding is like that of benzene. Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons • Any compound not classified as an aromatic compound is an aliphatic compound. • Alkanes, alkenes, alkynes, and cycloalkanes are aliphatic compounds. • The properties of aromatic compounds are quite different from those of aliphatic compounds. 14 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> CHEMISTRY & YOU Can you explain what it means if a compound is aromatic? 15 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> CHEMISTRY & YOU Can you explain what it means if a compound is aromatic? An aromatic compound is defined as an organic compound that contains a benzene ring or other ring in which the bonding is like that of benzene. Some aromatic compounds do have a smell, but not all do. 16 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
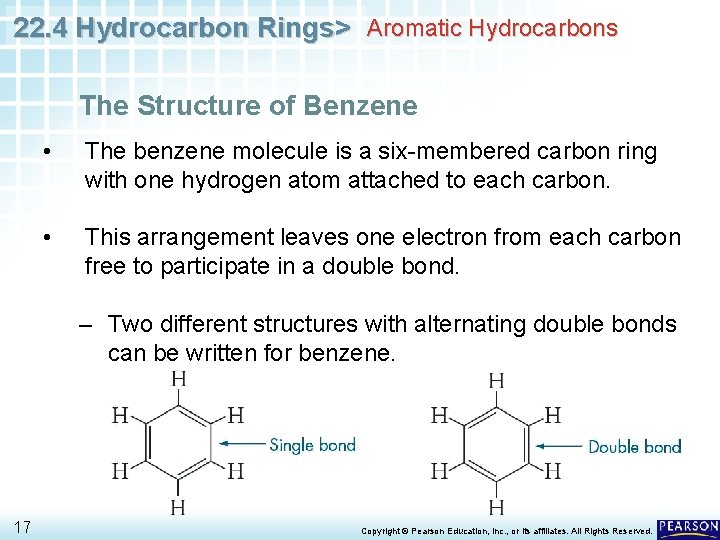
22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons The Structure of Benzene • The benzene molecule is a six-membered carbon ring with one hydrogen atom attached to each carbon. • This arrangement leaves one electron from each carbon free to participate in a double bond. – Two different structures with alternating double bonds can be written for benzene. 17 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
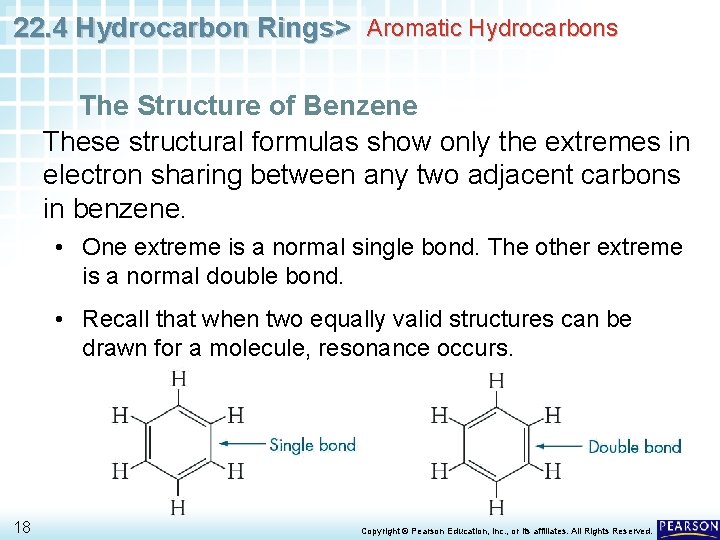
22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons The Structure of Benzene These structural formulas show only the extremes in electron sharing between any two adjacent carbons in benzene. • One extreme is a normal single bond. The other extreme is a normal double bond. • Recall that when two equally valid structures can be drawn for a molecule, resonance occurs. 18 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons The Structure of Benzene • In a benzene molecule, the bonding electrons between carbon atoms are shared evenly around the ring. 19 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons The Structure of Benzene • Benzene and other molecules that exhibit resonance are more stable than similar molecules that do not exhibit resonance. • Thus, benzene is not as reactive as six carbon alkenes. 20 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
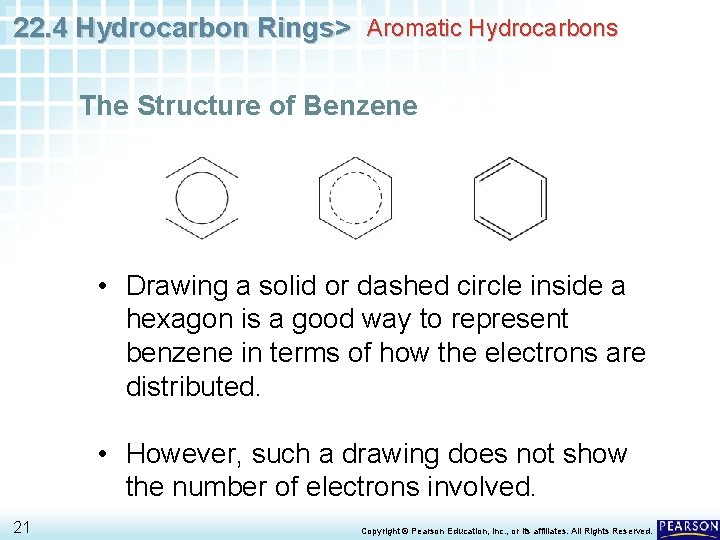
22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons The Structure of Benzene • Drawing a solid or dashed circle inside a hexagon is a good way to represent benzene in terms of how the electrons are distributed. • However, such a drawing does not show the number of electrons involved. 21 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons The Structure of Benzene • For this reason, the traditional structure, shown to the right in the series above, is used in this textbook. • Remember, though, that each bond in the ring is identical. 22 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons Substituted Aromatic Rings • Many dyes used to produce the intense colors of your clothing, such as the blue shown here are substituted aromatic compounds. 23 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
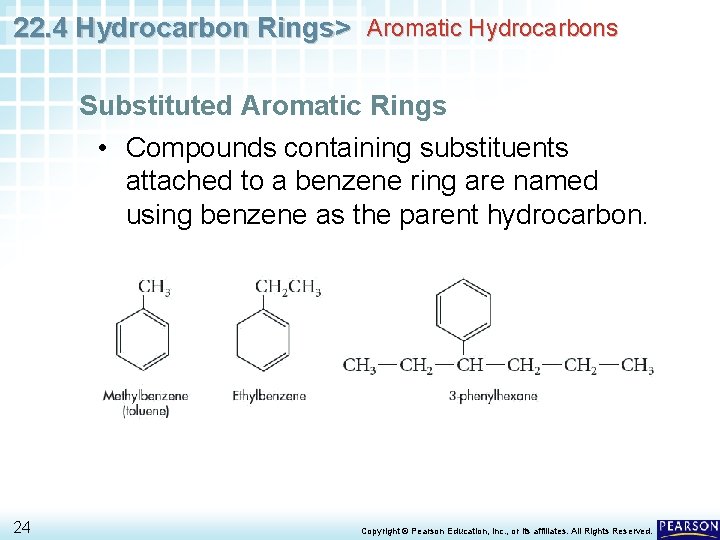
22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons Substituted Aromatic Rings • Compounds containing substituents attached to a benzene ring are named using benzene as the parent hydrocarbon. 24 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
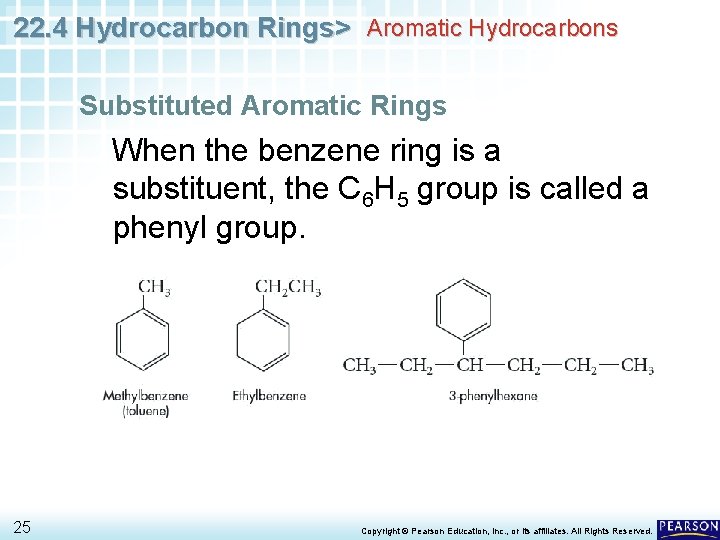
22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons Substituted Aromatic Rings When the benzene ring is a substituent, the C 6 H 5 group is called a phenyl group. 25 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons Substituted Aromatic Rings • Derivatives of benzene with two substituents are called disubstituted benzenes. • Dimethylbenzene, also called xylene, is an example of a disubstituted benzene. 26 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
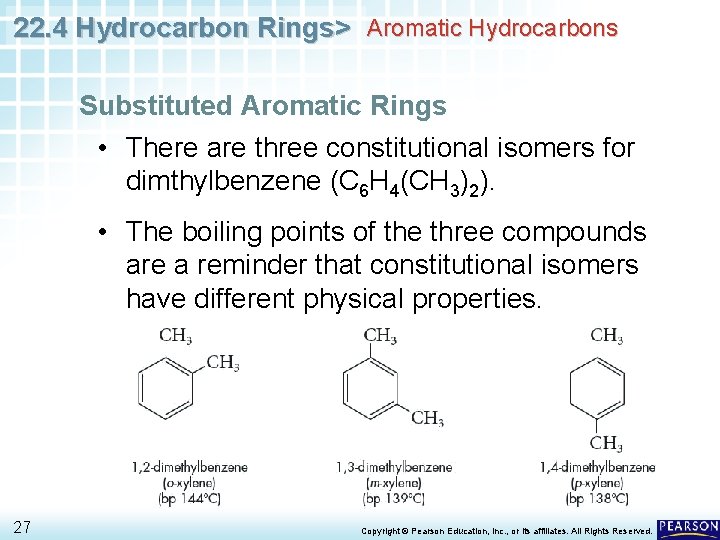
22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons Substituted Aromatic Rings • There are three constitutional isomers for dimthylbenzene (C 6 H 4(CH 3)2). • The boiling points of the three compounds are a reminder that constitutional isomers have different physical properties. 27 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Aromatic Hydrocarbons Substituted Aromatic Rings • In the IUPAC naming system, the possible positions of two substituents in disubstituted benzene are designated as 1, 2; 1, 3; or 1, 4. • Common names for disubstituted benzenes use the terms ortho, meta, and para (abbreviated as o, m, and p) in place of numbers. 28 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> How does the reactivity of benzene compare to the reactivity of sixcarbon alkenes? 29 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> How does the reactivity of benzene compare to the reactivity of sixcarbon alkenes? Benezene is not as reactive as six-carbon alkenes because molecules that exhibit resonance are more stable than similar molecules that do not exhibit resonance. 30 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Key Concepts In some hydrocarbon compounds, the carbon chain is in the form of a ring. In a benzene molecule, the bonding electrons between carbon atoms are shared evenly around the ring. 31 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> Glossary Terms • cyclic hydrocarbon: an organic compound that contains a hydrocarbon ring • cycloalkane: a cyclic hydrocarbon that contains only single bonds • aromatic compound: an organic compound that contains a benzene ring or other ring in which the bonding is like that of benzene; aromatic compounds are also known as arenes • aliphatic compound: any compound not classified as an aromatic compound 32 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.

22. 4 Hydrocarbon Rings> END OF 22. 4 33 Copyright © Pearson Education, Inc. , or its affiliates. All Rights Reserved.
- Slides: 33