2018 GLOBAL PORTICO PROGRAM UPDATE Portico Transcatheter Aortic

2018 GLOBAL PORTICO PROGRAM UPDATE Portico™ Transcatheter Aortic Valve System Augusto Pichard, MD Medical Director, Abbott Structural Heart Investigational device. Limited by Federal (U. S) law to investigational use only. Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US.

Disclosures A. Pichard. MD Medical Director, Abbott Structural Heart

INTRODUCTION TO THE PORTICO™ VALVE • Smallest TAVI profile when • Unique Nitinol properties allow valve to be fully repositioned* and retrieved* in situ used with the Portico Solo™ Expandable Sheath 1 • 360 degree flexible and trackable delivery system 3 • No rapid pacing required 4 • Low pacemaker rates 5 • Self-expanding, fully • Bovine pericardium leaflets with porcine pericardium sealing cuff re-captureable 6* • Largest cell geometry 2 • Both leaflets and cuff are treated with Linx™ AC technology** Patient Annulus (mm) 19 • Non-flared annulus section of stent frame and optimal valve height 20 21 23 Use. IFU-Table Range (mm) Potico 2 -ARTMT 100092981 H. Patient anatomical specifications. *Until fully deployed. *There is no clinical data currently available that evaluates the long-term impact of anticalcification tissue treatment in humans. ** Portico IFU-Description. ARTMT 100092981. The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. 22 25 23 24 27 25 26 27 29 1. Competitive Profile Comparison_SJM-PTC-0715 -0048. 2. Coronary access report, for cell size measurements #90103707. 3. Søndergaard L. How Portico helps solve challenging cases: Horizontal aorta & calcified annulus. Webinar March 4, 2016. 4. Portico IFU. P 10 -12 Implanting the Valve. 5. Linke A et al. PCR London Valves 2015. 6. Potico IFU-Implanting the Valve-ARTMT 100092981 H. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the 3

TOP REASONS TO RETHINK THE PORTICO SELF-EXPANDING VALVE: • Achieve excellent patient outcomes without compromising your delivery technique 1 • Experience controlled, relaxed deployment, thanks to continuous hemodynamic stability 2 • Tackle every case, even the most tortuous anatomies, with the most deliverable valve on the market 3 • Protect future treatment options by preserving coronary access. 1. Linke A et al. Treatment of aortic stenosis with a self-expanding, resheathable transcatheter valve: One-year results of the international multicenter Portico™ TAVI system study. Circulation: Cardiovascular Interventions. [In press; Accepted for publication Dec 18, 2017] 2. Portico IFU. 3. Søndergaard L. How Portico helps solve challenging cases: Horizontal aorta & calcified annulus. Webinar March 4, 2016. The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 4

EASY CORONARY ACCESS Images courtesy Dr Ganesh Manoharan Royal Victoria Hospital, Belfast, UK The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 5

PORTICO™ DELIVERY SYSTEM Transfemoral System Transaortic or Subclavian System* 18 F delivery system compatible with the 23 and 25 mm valves. 19 F delivery system compatible with the 27 and 29 mm valves The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 6

INSERTION PROFILE DIAMETERS PORTICO IS NO BIGGER THAN EVOLUT R OR SAPIEN 3* 25 22. 5 Diameter (Fr) 20 15 18. 0 14. 0 13. 5 20. 1 19. 0 14. 0 11. 0 10 ID 5 0 ABT: Porticoᵀᴹ Soloᵀᴹ ABT: Porticoᵀᴹ MDT: Evolutᵀᴹ R (In. Lineᵀᴹ Sheath) EW: Sapienᵀᴹ 3 (e. Sheathᵀᴹ) BSX: Lotusᵀᴹ (Lotusᵀᴹ Sheath) *Measurements for all technologies are based on manufacturers’ published specifications and validated by SJM internal measurements on file at WC Report #90237740 The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 7

NEXTGEN ABBOTT TAVI DELIVERY SYSTEM EASE OF USE • Improved Handle Design • Improved Retainer Release REDUCED INSERTION PROFILE • Sheathless Approach with Integrated Sheath • Compatible with Expandable Sheaths • Hydrophilic Coating IMPROVED PLACEMENT ACCURACY • New Stability Layer • Improved Flexibility and Trackability (360° flexibility) • Passive Alignment The product in this slide is in development and not yet available for sale. Any features or performance information presented are based on the current design goals and may be subjected to revisions to meet the needs of the product development program. Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 8

PORTICO™ VALVE: ADVANCED FEATURES TO CONTROL VALVE DEPLOYMENT § No drop in hemodynamic pressure § Ample time to achieve optimal placement before release § No rapid pacing required Functioning leaflets The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 9

PORTICO™ VALVE: ADVANCED FEATURES TO MINIMIZE PVL • Frame: conformable, adaptable • Sealing cuff • Valve functions in round or elliptical annulus • Large cell geometry allows valve tissue to conform around calcific nodules Calcific Nodules Large cell geometry 1 High tissueto-tissue contact 1. Coronary access report, for cell size measurements #90103707. The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 10

NEXTGEN ABBOTT TAVI VALVE TECHNOLOGY Design Goals 1. IMPROVED PVL PERFORMANCE • New inner and outer cuffs design • Extended landing and sealing zone • Optimized annulus contact and conformability 2. EXCELLENT PERFORMANCE • Durability • Hemodynamics 3. IMPROVED IMPLANT STABLITY • Optimized performance during initial positioning, implantation and after release 4. MINIMIZED PATIENT RISK For illustrative purpose only. • Atraumatic Aortic Cells • Low valve crimp profile allows for small delivery system profile The product in this slide is in development and not yet available for sale. Any features or performance information presented are based on the current design goals and may be subjected to revisions to meet the needs of the product development program. Not to be reproduced, distributed or 11 SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US.

PORTICO™ VALVE: ADVANCED FEATURES TO MINIMIZE AV BLOCK • Intra-annular placement to minimize extension into the LVOT • Non-flared annular skirt with porcine pericardial cuff • Uniform distribution of radial force to annulus The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 12
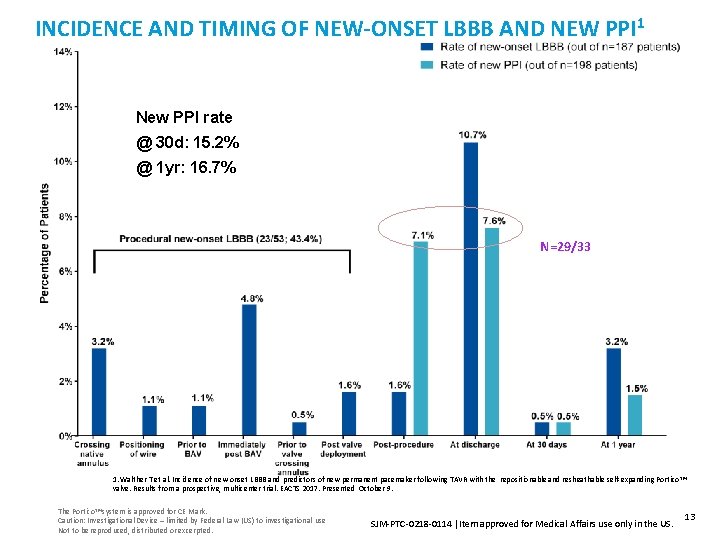
INCIDENCE AND TIMING OF NEW-ONSET LBBB AND NEW PPI 1 New PPI rate @ 30 d: 15. 2% @ 1 yr: 16. 7% N=29/33 1. Walther T et al. Incidence of new-onset LBBB and predictors of new permanent pacemaker following TAVR with the repositionable and resheathable self-expanding Portico ™ valve. Results from a prospective, multicenter trial. EACTS 2017. Presented October 9. The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 13
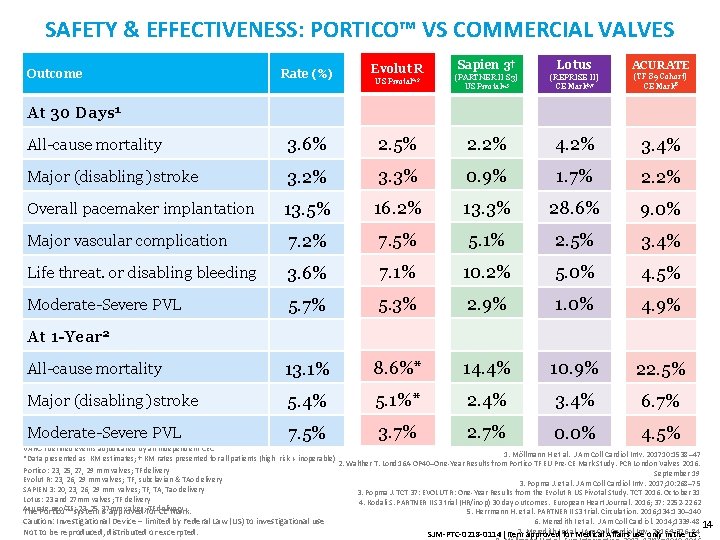
SAFETY & EFFECTIVENESS: PORTICO™ VS COMMERCIAL VALVES Rate (%) Evolut R Sapien 3† Lotus US Pivotal 2, 3 (PARTNER II S 3) US Pivotal 4, 5 (REPRISE II) CE Mark 6, 7 (TF 89 Cohort) CE Mark 8 All-cause mortality 3. 6% 2. 5% 2. 2% 4. 2% 3. 4% Major (disabling) stroke 3. 2% 3. 3% 0. 9% 1. 7% 2. 2% Overall pacemaker implantation 13. 5% 16. 2% 13. 3% 28. 6% 9. 0% Major vascular complication 7. 2% 7. 5% 5. 1% 2. 5% 3. 4% Life threat. or disabling bleeding 3. 6% 7. 1% 10. 2% 5. 0% 4. 5% Moderate-Severe PVL 5. 7% 5. 3% 2. 9% 1. 0% 4. 9% All-cause mortality 13. 1% 8. 6%* 14. 4% 10. 9% 22. 5% Major (disabling) stroke 5. 4% 5. 1%* 2. 4% 3. 4% 6. 7% Moderate-Severe PVL 7. 5% 3. 7% 2. 7% 0. 0% 4. 5% Outcome ACURATE At 30 Days 1 At 1 -Year 2 VARC-I defined events adjudicated by an independent CEC 1. Möllmann H et al. J Am Coll Cardiol Intv. 201710: 1538– 47 *Data presented as KM estimates; † KM rates presented for all patients (high risk + inoperable) 2. Walther T. Lond 16 A-OP 40–One-Year Results from Portico TF EU Pre-CE Mark Study. PCR London Valves 2016. Portico: 23, 25, 27, 29 mm valves; TF delivery September 19 Evolut R: 23, 26, 29 mm valves; TF, subclavian & TAo delivery 3. Popma J. et al. J Am Coll Cardiol Intv. 2017; 10: 268– 75 SAPIEN 3: 20, 23, 26, 29 mm valves; TF, TA, Tao delivery 3. Popma J. TCT 37: EVOLUT R: One-Year Results from the Evolut R US Pivotal Study. TCT 2016. October 31 Lotus: 23 and 27 mm valves; TF delivery 4. Kodali S. PARTNER II S 3 trial (HR/inop) 30 day outcomes. European Heart Journal. 2016; 37: 2252 -2262 Acurate neo/TF: system 23, 25, is 27 mm valves; for TF delivery The Portico™ approved CE Mark. 5. Herrmann H. et al. PARTNER II S 3 trial. Circulation. 2016; 134: 130– 140 Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. 6. Meredith I et al. J Am Coll Cardiol. 2014; 1339 -48 14 7. Meredith J Am Coll. Affairs Cardioluse Intv. only 2016; 9: 376– 84 SJM-PTC-0218 -0114 Item approved. I et foral. Medical in the US.
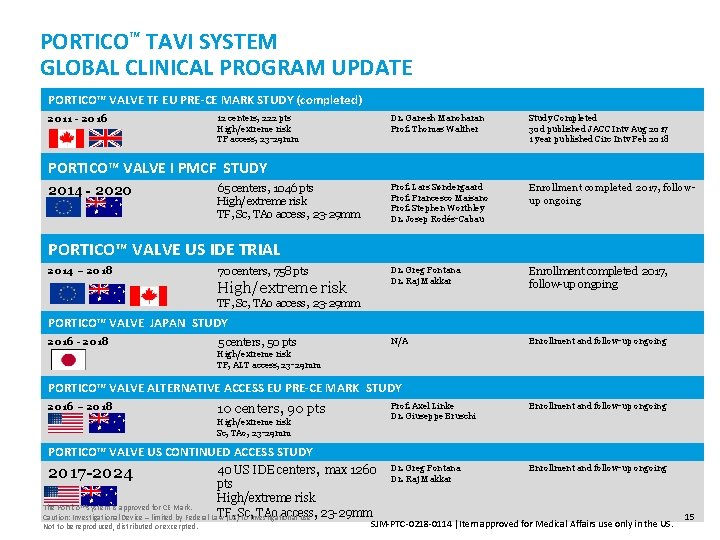
PORTICO™ TAVI SYSTEM GLOBAL CLINICAL PROGRAM UPDATE PORTICO™ VALVE TF EU PRE-CE MARK STUDY (completed) 2011 - 2016 12 centers, 222 pts High/extreme risk TF access, 23 -29 mm Dr. Ganesh Manoharan Prof. Thomas Walther Study Completed 30 d published JACC Intv Aug 2017 1 year published Circ Intv Feb 2018 Prof. Lars Søndergaard Prof. Francesco Maisano Prof. Stephen Worthley Dr. Josep Rodés-Cabau Enrollment completed 2017, followup ongoing Dr. Greg Fontana Dr. Raj Makkar Enrollment completed 2017, follow-up ongoing N/A Enrollment and follow-up ongoing PORTICO™ VALVE I PMCF STUDY 2014 - 2020 65 centers, 1046 pts High/extreme risk TF, Sc, TAo access, 23 -29 mm PORTICO™ VALVE US IDE TRIAL 2014 – 2018 70 centers, 758 pts High/extreme risk TF, Sc, TAo access, 23 -29 mm PORTICO™ VALVE JAPAN STUDY 2016 - 2018 5 centers, 50 pts High/extreme risk TF, ALT access, 23 -29 mm PORTICO™ VALVE ALTERNATIVE ACCESS EU PRE-CE MARK STUDY 2016 – 2018 10 centers, 90 pts High/extreme risk Sc, TAo, 23 -29 mm Prof. Axel Linke Dr. Giuseppe Bruschi Enrollment and follow-up ongoing Dr. Greg Fontana Dr. Raj Makkar Enrollment and follow-up ongoing PORTICO™ VALVE US CONTINUED ACCESS STUDY 40 US IDE centers, max 1260 pts High/extreme risk The Portico™ system is approved for CE Mark. TF, TAo access, Caution: Investigational Device – limited by Federal Law (US)Sc, to investigational use 23 -29 mm 2017 -2024 Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 15
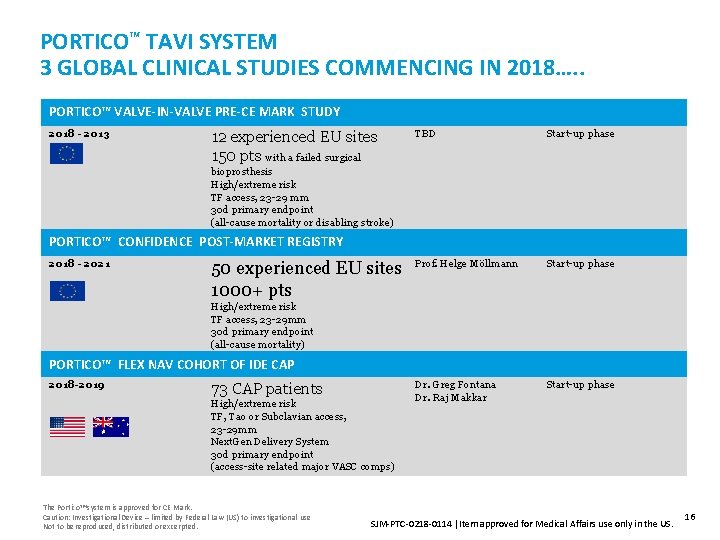
PORTICO™ TAVI SYSTEM 3 GLOBAL CLINICAL STUDIES COMMENCING IN 2018…. . PORTICO™ VALVE-IN-VALVE PRE-CE MARK STUDY 2018 - 2013 12 experienced EU sites 150 pts with a failed surgical TBD Start-up phase Prof. Helge Möllmann Start-up phase Dr. Greg Fontana Dr. Raj Makkar Start-up phase bioprosthesis High/extreme risk TF access, 23 -29 mm 30 d primary endpoint (all-cause mortality or disabling stroke) PORTICO™ CONFIDENCE POST-MARKET REGISTRY 2018 - 2021 50 experienced EU sites 1000+ pts High/extreme risk TF access, 23 -29 mm 30 d primary endpoint (all-cause mortality) PORTICO™ FLEX NAV COHORT OF IDE CAP 2018 -2019 73 CAP patients High/extreme risk TF, Tao or Subclavian access, 23 -29 mm Next. Gen Delivery System 30 d primary endpoint (access-site related major VASC comps) The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 16

PORTICO™ VALVE MOMENTUM…. • Commercial approval of four valve sizes for the last 24+ Months • 32 countries implant Portico™ Valves • Japan trial will complete enrollment in 2018 • ALT Access will complete enrollment in 2018 • Portico I enrollment completed (total 1, 032 patients). Primary results to be presented in 2018 • US IDE randomized trial 1 yr follow-up to be completed in 2018 • US IDE Continued Access study started in 2017 • Two new trials starting in 2018 • Flex. NAV Delivery System clinical study starting in 2018 • Next generation developments on track The Portico™ system is approved for CE Mark. Caution: Investigational Device – limited by Federal Law (US) to investigational use Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 17

ABBOTT STRUCTURAL HEART: 9 GLOBAL LAUNCHES IN 2019…. PIPELINE AORTIC MITRAL 2018 2019 Pediatric Mechanical Valve Portico US Pediatric Mechanical Mitra. Clip Japan V 3. 0 Valve Trifecta Portico DS GT V 1. 5 Portico V 2. 0 Tendyne V 1. 0 Mitra. Clip V 5. 0 Mitra. Clip V 4. 0 TRICUSPID STRUCTURAL INTERVENTIONS 2020 Tri. Clip EU PFO DS ASD DS LAA SS DISCLAIMER: Products in development. Not commercially available. Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 18

Examples Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 19
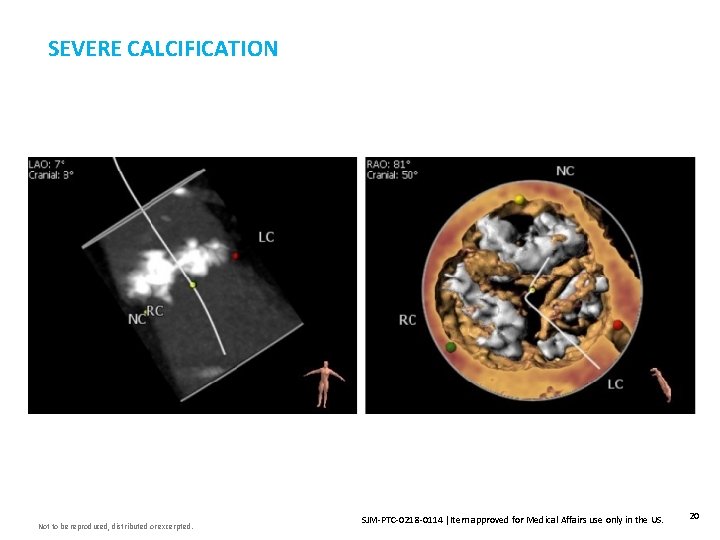
SEVERE CALCIFICATION Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 20
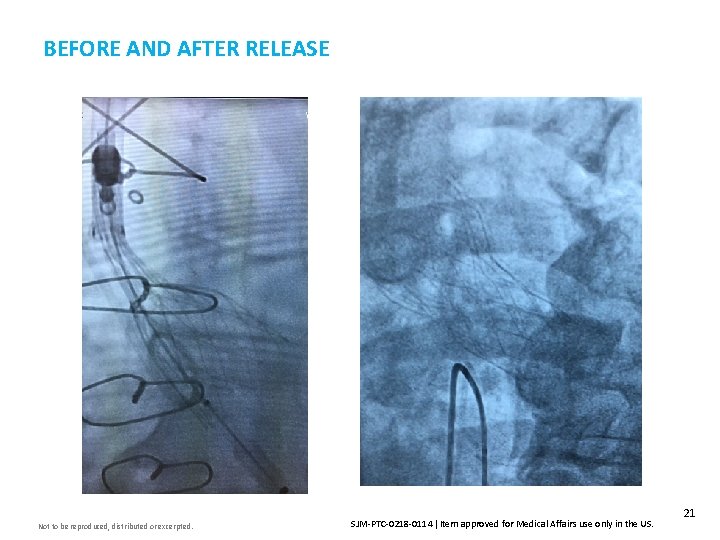
BEFORE AND AFTER RELEASE Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 21

FINAL OUTCOME • Trace PVL at the end • 4 mm Hg gradient • Final AVA >2 cm 2 Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 22
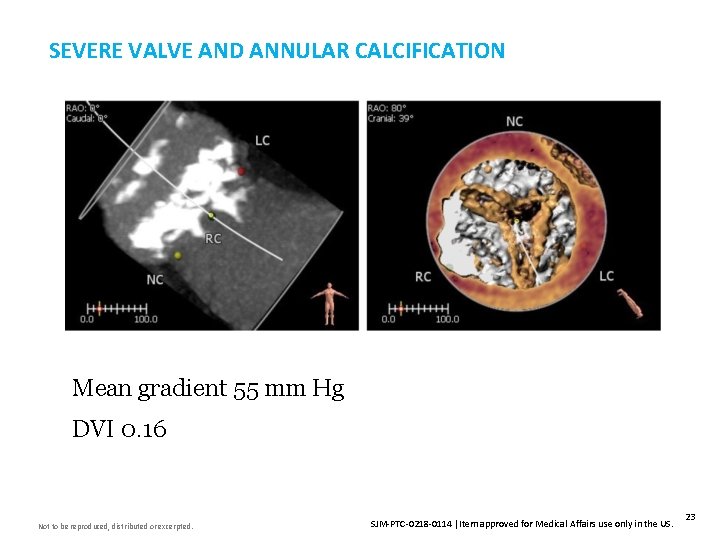
SEVERE VALVE AND ANNULAR CALCIFICATION Insert Image Here Mean gradient 55 mm Hg DVI 0. 16 Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 23
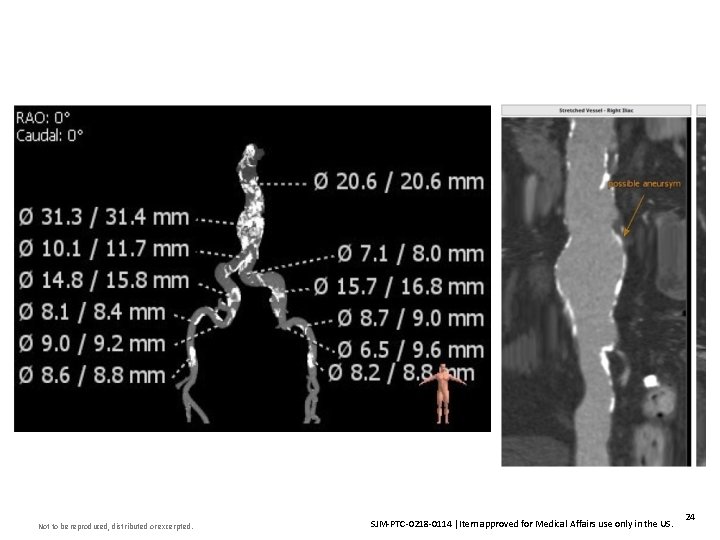
Insert Image Here Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 24

PROCEDURE • Predil 22 mm Zmed • Final depth 4 -5 mm • Mild PVL on TTE and angio → Post dil with 24 mm balloon → Trace to mild PVL Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 25
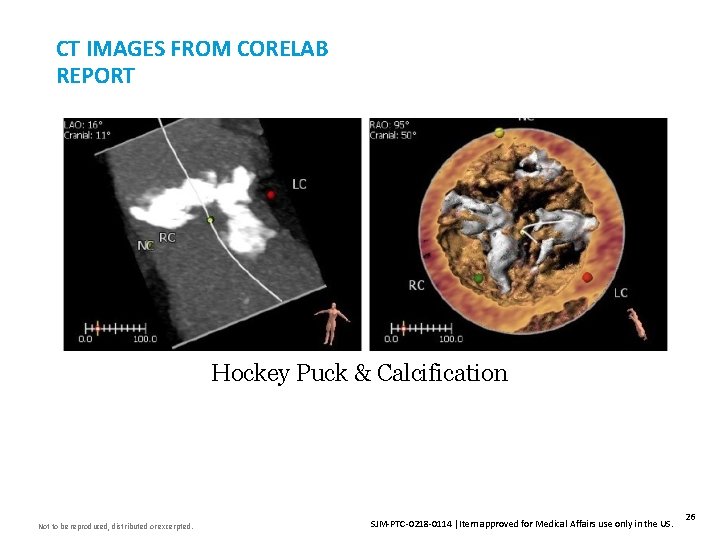
CT IMAGES FROM CORELAB REPORT Insert Image Here Hockey Puck & Calcification Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 26
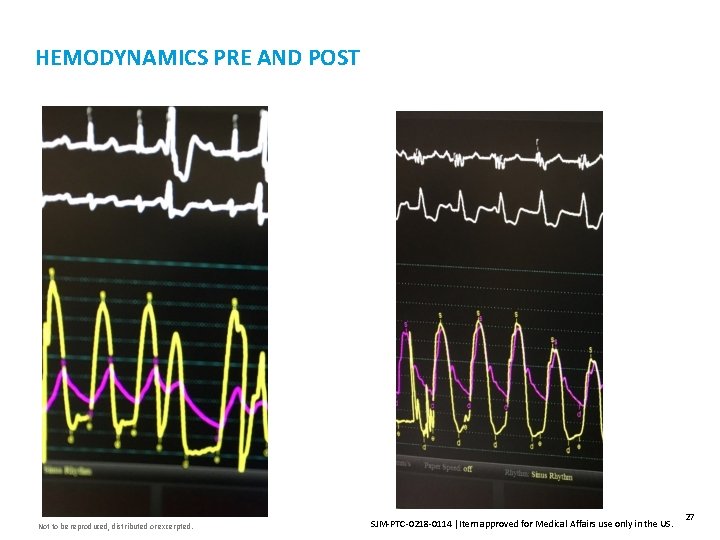
HEMODYNAMICS PRE AND POST Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 27
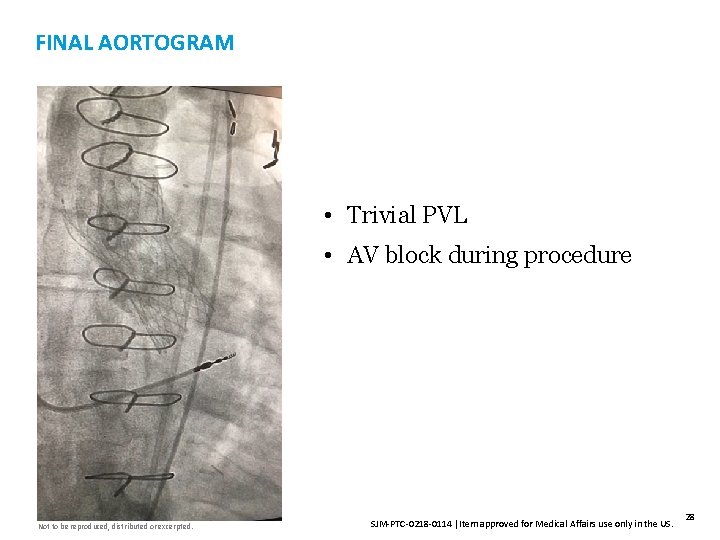
FINAL AORTOGRAM • Trivial PVL • AV block during procedure Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 28

BAD LV FUNCTION • 90 year old • EF 35% → 20% in Cath Lab • Mean gradient 52 mm Hg • DVI 0. 2 • 2 vessel CAD Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 29
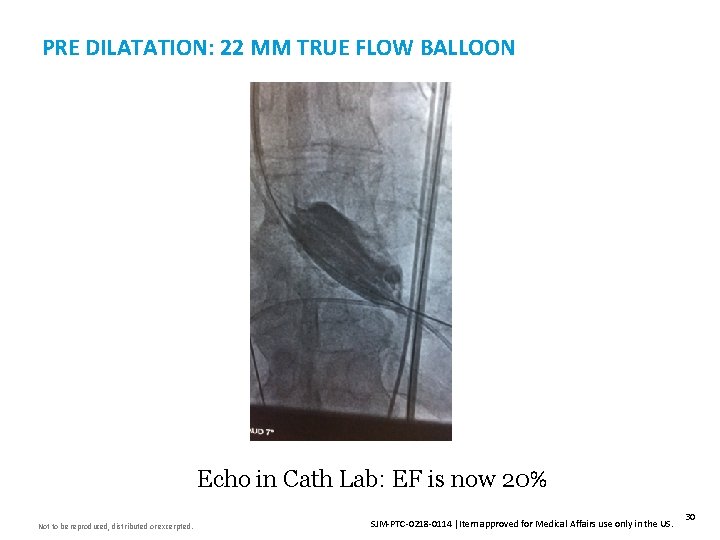
PRE DILATATION: 22 MM TRUE FLOW BALLOON Echo in Cath Lab: EF is now 20% Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 30
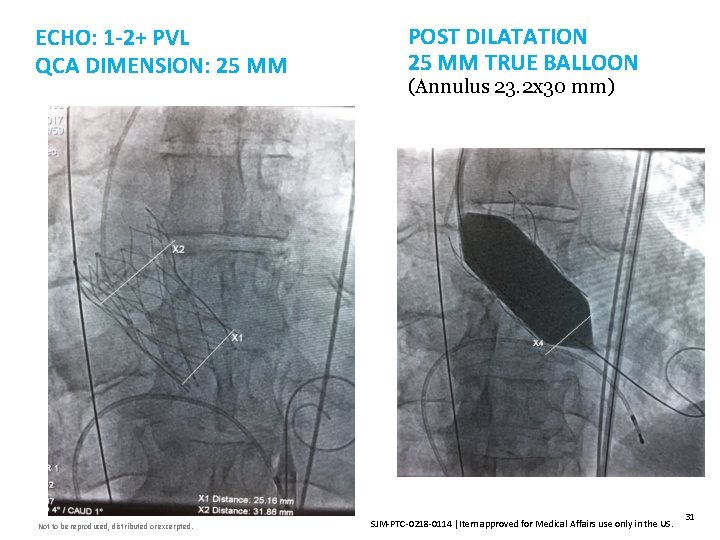
ECHO: 1 -2+ PVL QCA DIMENSION: 25 MM Not to be reproduced, distributed or excerpted. POST DILATATION 25 MM TRUE BALLOON (Annulus 23. 2 x 30 mm) SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 31

FINAL ECHO • Trace PVL • LV function improved from 20% → 40% • AVA >2. 0 cm 2 by echo • No conduction change. Temp pacer pulled out • Femoral closed with 2 proglides Not to be reproduced, distributed or excerpted. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US. 32

Abbott One St. Jude Medical Dr. , St. Paul, MN 55117 USA Tel: 1 651 756 2000 SJM. com St. Jude Medical is now Abbott CAUTION: This product is intended for use by or under the direction of a physician. Prior to use, reference the Instructions for Use, inside the product carton (when available) or at manuals. sjm. com for more detailed information on Indications, Contraindications, Warnings, Precautions and Adverse Events. ™Indicates a trademark of the Abbott group of companies ®Indicates a third-party trademark, which is property of its respective owner. © 2018 Abbott. All Rights Reserved. SJM-PTC-0218 -0114 Item approved for Medical Affairs use only in the US.
- Slides: 33