2017 ESMO CONGRESS 8 12 September 2017 Madrid

2017 ESMO CONGRESS 8– 12 September 2017 | Madrid, Spain Supported by Eli Lilly and Company has not influenced the content of this publication

Letter from Prof Jean Yves-Blay Dear Colleagues It is my pleasure to present this WSN slide set which has been designed to highlight and summarise key findings in sarcoma from the major congresses in 2017. This slide set specifically focuses on the ESMO 2017 Congress. The area of clinical research in oncology is a challenging and ever changing environment. Within this environment we all value access to scientific data and research that helps to educate and inspire further advancements in our roles as scientists, clinicians and educators. I hope you find this review of the latest developments in sarcoma of benefit to you in your practice. I would like to thank our WSN members Drs Piotr Rutkowski and Claudia Valverde for their roles as Editors – for prioritising abstracts and reviewing slide content. The slide set you see before you would not be possible without their commitment and hard work. And finally, we are also very grateful to Lilly Oncology for their financial, administrial and logistical support in the realisation of this complex yet rewarding activity. Yours sincerely, Jean Yves-Blay WSN Chairman of the Board

WSN Medical Oncology Slide Deck Editors 2017 Piotr Rutkowski Maria Sklodowska-Curie Institute – Oncology Center, Warsaw, Poland Claudia Valverde Vall d’Hebron University Hospital, Barcelona, Spain

Contents • Gastrointestinal stromal tumours • Locally advanced sarcoma 5 17 • Advanced/metastatic sarcoma: chemotherapy, targeted therapy and immunotherapy 34

Gastrointestinal stromal tumours
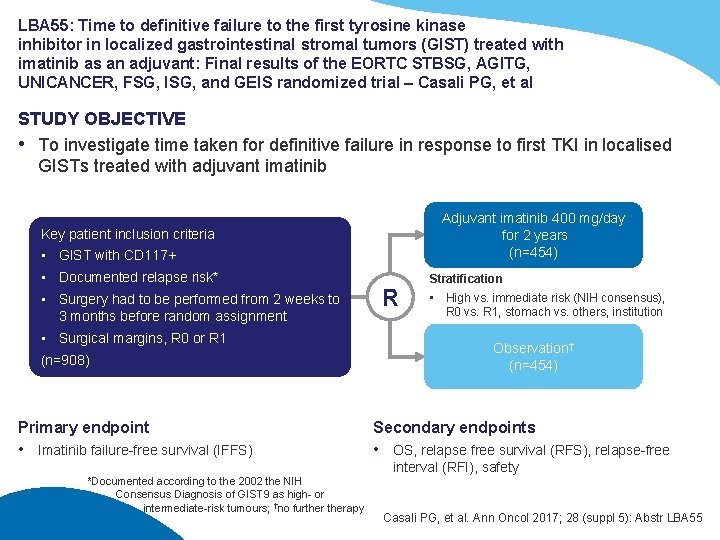
LBA 55: Time to definitive failure to the first tyrosine kinase inhibitor in localized gastrointestinal stromal tumors (GIST) treated with imatinib as an adjuvant: Final results of the EORTC STBSG, AGITG, UNICANCER, FSG, ISG, and GEIS randomized trial – Casali PG, et al STUDY OBJECTIVE • To investigate time taken for definitive failure in response to first TKI in localised GISTs treated with adjuvant imatinib Adjuvant imatinib 400 mg/day for 2 years (n=454) Key patient inclusion criteria • GIST with CD 117+ • Documented relapse risk* • Surgery had to be performed from 2 weeks to 3 months before random assignment • Surgical margins, R 0 or R 1 (n=908) Primary endpoint • Imatinib failure-free survival (IFFS) R Stratification • High vs. immediate risk (NIH consensus), R 0 vs. R 1, stomach vs. others, institution Observation† (n=454) Secondary endpoints • OS, relapse free survival (RFS), relapse-free interval (RFI), safety *Documented according to the 2002 the NIH Consensus Diagnosis of GIST 9 as high- or intermediate-risk tumours; †no furtherapy Casali PG, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 55
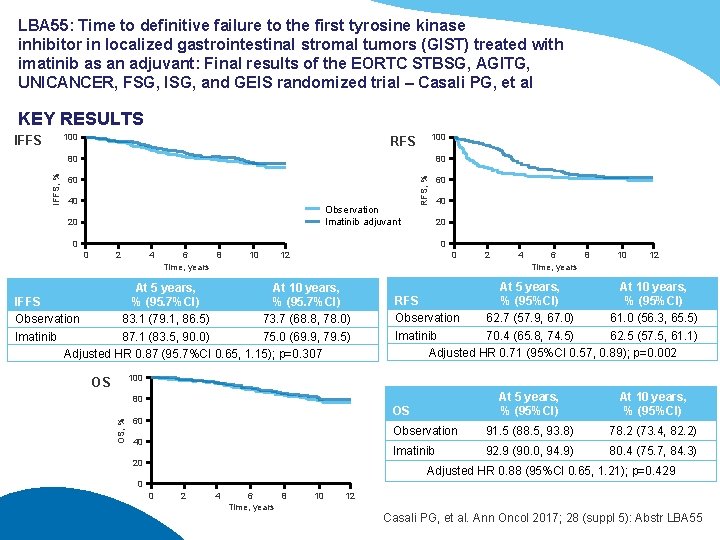
LBA 55: Time to definitive failure to the first tyrosine kinase inhibitor in localized gastrointestinal stromal tumors (GIST) treated with imatinib as an adjuvant: Final results of the EORTC STBSG, AGITG, UNICANCER, FSG, ISG, and GEIS randomized trial – Casali PG, et al KEY RESULTS 100 RFS 80 80 60 60 40 Observation Imatinib adjuvant 20 RFS, % IFFS 40 20 0 2 4 6 8 Time, years 10 12 0 At 5 years, At 10 years, IFFS % (95. 7%CI) Observation 83. 1 (79. 1, 86. 5) 73. 7 (68. 8, 78. 0) Imatinib 87. 1 (83. 5, 90. 0) 75. 0 (69. 9, 79. 5) Adjusted HR 0. 87 (95. 7%CI 0. 65, 1. 15); p=0. 307 2 4 6 8 Time, years 10 12 At 5 years, At 10 years, RFS % (95%CI) Observation 62. 7 (57. 9, 67. 0) 61. 0 (56. 3, 65. 5) Imatinib 70. 4 (65. 8, 74. 5) 62. 5 (57. 5, 61. 1) Adjusted HR 0. 71 (95%CI 0. 57, 0. 89); p=0. 002 100 OS At 5 years, % (95%CI) At 10 years, % (95%CI) Observation 91. 5 (88. 5, 93. 8) 78. 2 (73. 4, 82. 2) Imatinib 92. 9 (90. 0, 94. 9) 80. 4 (75. 7, 84. 3) OS, % 80 OS 60 40 20 Adjusted HR 0. 88 (95%CI 0. 65, 1. 21); p=0. 429 0 0 2 4 6 8 Time, years 10 12 Casali PG, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 55
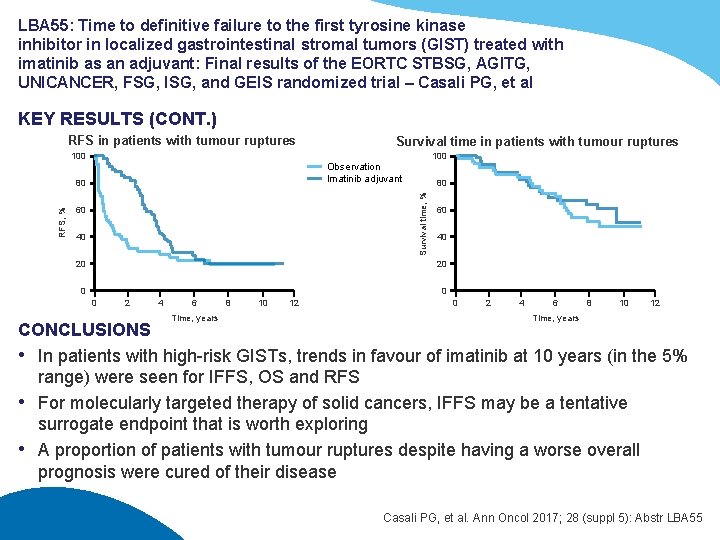
LBA 55: Time to definitive failure to the first tyrosine kinase inhibitor in localized gastrointestinal stromal tumors (GIST) treated with imatinib as an adjuvant: Final results of the EORTC STBSG, AGITG, UNICANCER, FSG, ISG, and GEIS randomized trial – Casali PG, et al KEY RESULTS (CONT. ) RFS in patients with tumour ruptures Survival time in patients with tumour ruptures 100 Observation Imatinib adjuvant 80 Survival time, % RFS, % 80 60 40 20 0 2 4 6 Time, years 8 10 12 0 2 4 6 8 10 12 Time, years CONCLUSIONS • In patients with high-risk GISTs, trends in favour of imatinib at 10 years (in the 5% range) were seen for IFFS, OS and RFS • For molecularly targeted therapy of solid cancers, IFFS may be a tentative surrogate endpoint that is worth exploring • A proportion of patients with tumour ruptures despite having a worse overall prognosis were cured of their disease Casali PG, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 55
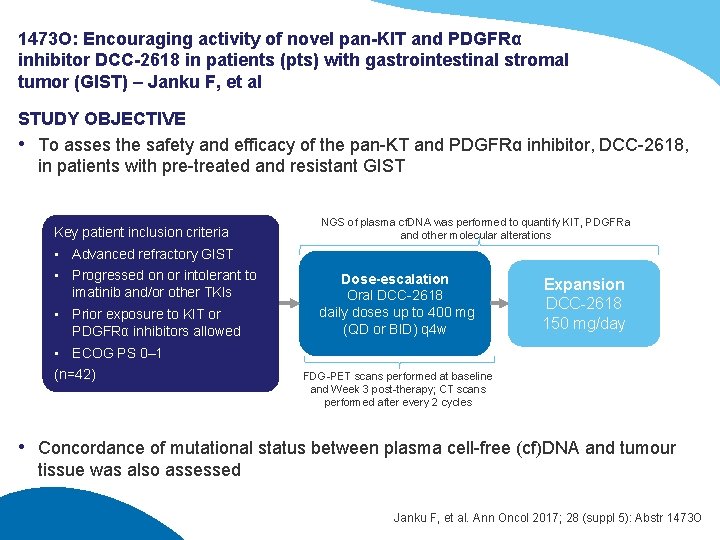
1473 O: Encouraging activity of novel pan-KIT and PDGFRα inhibitor DCC-2618 in patients (pts) with gastrointestinal stromal tumor (GIST) – Janku F, et al STUDY OBJECTIVE • To asses the safety and efficacy of the pan-KT and PDGFRɑ inhibitor, DCC-2618, in patients with pre-treated and resistant GIST Key patient inclusion criteria NGS of plasma cf. DNA was performed to quantify KIT, PDGFRa and other molecular alterations • Advanced refractory GIST • Progressed on or intolerant to imatinib and/or other TKIs • Prior exposure to KIT or PDGFRα inhibitors allowed Dose-escalation Oral DCC-2618 daily doses up to 400 mg (QD or BID) q 4 w Expansion DCC-2618 150 mg/day • ECOG PS 0– 1 (n=42) FDG-PET scans performed at baseline and Week 3 post-therapy; CT scans performed after every 2 cycles • Concordance of mutational status between plasma cell-free (cf)DNA and tumour tissue was also assessed Janku F, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1473 O
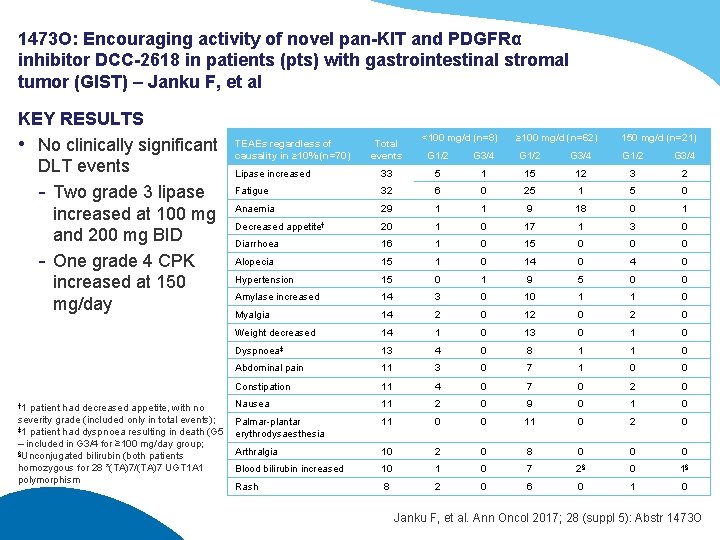
1473 O: Encouraging activity of novel pan-KIT and PDGFRα inhibitor DCC-2618 in patients (pts) with gastrointestinal stromal tumor (GIST) – Janku F, et al KEY RESULTS • No clinically significant DLT events - Two grade 3 lipase increased at 100 mg and 200 mg BID - One grade 4 CPK increased at 150 mg/day † 1 patient had decreased appetite, with no severity grade (included only in total events); ‡ 1 patient had dyspnoea resulting in death (G 5 – included in G 3/4 for ≥ 100 mg/day group; §Unconjugated bilirubin (both patients homozygous for 28 *(TA)7/(TA)7 UGT 1 A 1 polymorphism TEAEs regardless of causality in ≥ 10% (n=70) Total events <100 mg/d (n=8) ≥ 100 mg/d (n=62) 150 mg/d (n=21) G 1/2 G 3/4 Lipase increased 33 5 1 15 12 3 2 Fatigue 32 6 0 25 1 5 0 29 1 1 9 18 0 1 20 1 0 17 1 3 0 Diarrhoea 16 1 0 15 0 0 0 Alopecia 15 1 0 14 0 Hypertension 15 0 1 9 5 0 0 Amylase increased 14 3 0 10 1 1 0 Myalgia 14 2 0 12 0 Weight decreased 14 1 0 13 0 1 0 Dyspnoea‡ 13 4 0 8 1 1 0 Abdominal pain 11 3 0 7 1 0 0 Constipation 11 4 0 7 0 2 0 Nausea 11 2 0 9 0 1 0 Palmar-plantar erythrodysaesthesia 11 0 0 11 0 2 0 Arthralgia 10 2 0 8 0 0 0 Blood bilirubin increased 10 1 0 7 2§ 0 1§ Rash 8 2 0 6 0 1 0 Anaemia Decreased appetite† Janku F, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1473 O

1473 O: Encouraging activity of novel pan-KIT and PDGFRα inhibitor DCC-2618 in patients (pts) with gastrointestinal stromal tumor (GIST) – Janku F, et al KEY RESULTS (CONT. ) Partial metabolic response Stable metabolic response Progressive metabolic response <100 md/day (n=1) 1 (100) 0 (0) ≥ 100 mg/day (n=32) 22 (69) 150 mg/day (n=8) 3 (38) n (%) 9 (22) 5 (63) 1 (3) 0 (0) Change from baseline in target tumour size, % FDG-PET scans (n=33) 40 DCC-2618 dose assigned 50 mg BID 200 mg BID 100 mg QD 150 mg BID 150 mg QD 30 20 12 -week DCR*, n/N (%) 19/25 (76) 24 -week DCR*, n/N (%) 12/21 (57) *PR + SD per RECIST * PD 10 0 * -10 -20 -30 PR -40 -50% Screening 4 KIT- and PDGFRα GIST cohorts (daily dose ≥ 100 mg) * * 8 12 16 20 24 28 32 36 40 44 48 52 56 60 64 Weeks on DCC-2618 • DCC-2618 shows durable disease control in heavily pretreated patients with KIT and PDGFRα mutant GIST • In tissue and plasma there was a broad diversity of KIT mutations and more resistance mutations in plasma cf. DNA than in tumour Janku F, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1473 O
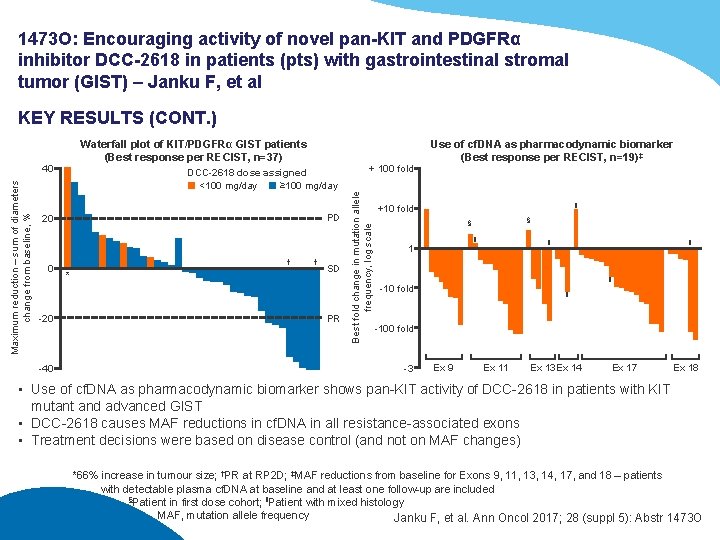
1473 O: Encouraging activity of novel pan-KIT and PDGFRα inhibitor DCC-2618 in patients (pts) with gastrointestinal stromal tumor (GIST) – Janku F, et al KEY RESULTS (CONT. ) Maximum reduction – sum of diameters change from baseline, % 40 DCC-2618 dose assigned <100 mg/day ≥ 100 mg/day PD 20 0 -20 -40 † * † SD PR + 100 fold Best fold change in mutation allele frequency, log scale Waterfall plot of KIT/PDGFRα GIST patients (Best response per RECIST, n=37) Use of cf. DNA as pharmacodynamic biomarker (Best response per RECIST, n=19)‡ ‖ +10 fold § § ‖ 1 ‖ ‖ ‖ -10 fold ‖ -100 fold -3 Ex 9 Ex 11 Ex 13 Ex 14 Ex 17 Ex 18 • Use of cf. DNA as pharmacodynamic biomarker shows pan-KIT activity of DCC-2618 in patients with KIT mutant and advanced GIST • DCC-2618 causes MAF reductions in cf. DNA in all resistance-associated exons • Treatment decisions were based on disease control (and not on MAF changes) *66% increase in tumour size; †PR at RP 2 D; ‡MAF reductions from baseline for Exons 9, 11, 13, 14, 17, and 18 – patients with detectable plasma cf. DNA at baseline and at least one follow-up are included §Patient in first dose cohort; ‖Patient with mixed histology MAF, mutation allele frequency Janku F, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1473 O
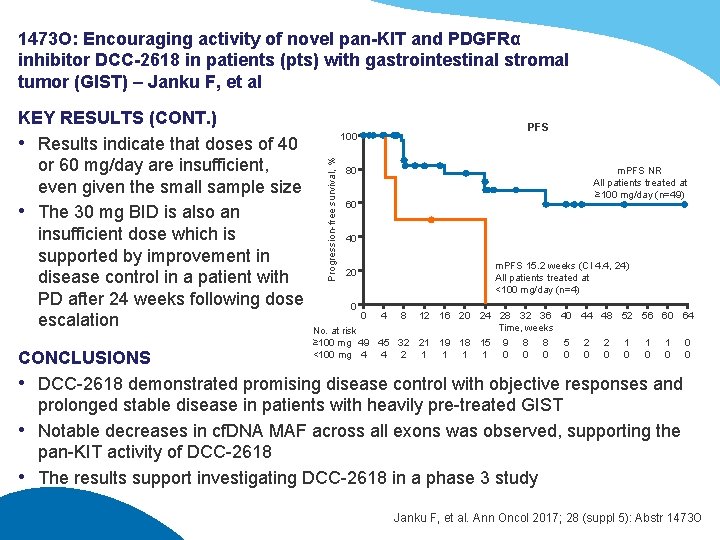
1473 O: Encouraging activity of novel pan-KIT and PDGFRα inhibitor DCC-2618 in patients (pts) with gastrointestinal stromal tumor (GIST) – Janku F, et al PFS 100 Progression-free survival, % KEY RESULTS (CONT. ) • Results indicate that doses of 40 or 60 mg/day are insufficient, even given the small sample size • The 30 mg BID is also an insufficient dose which is supported by improvement in disease control in a patient with PD after 24 weeks following dose escalation m. PFS NR All patients treated at ≥ 100 mg/day (n=49) 80 60 40 m. PFS 15. 2 weeks (CI 4. 4, 24) All patients treated at <100 mg/day (n=4) 20 0 0 4 8 12 16 20 24 28 32 36 40 44 48 52 56 60 64 Time, weeks No. at risk ≥ 100 mg 49 45 32 21 19 18 15 9 8 8 5 2 2 1 1 1 0 <100 mg 4 4 2 1 1 0 0 0 0 0 CONCLUSIONS • DCC-2618 demonstrated promising disease control with objective responses and prolonged stable disease in patients with heavily pre-treated GIST • Notable decreases in cf. DNA MAF across all exons was observed, supporting the pan-KIT activity of DCC-2618 • The results support investigating DCC-2618 in a phase 3 study Janku F, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1473 O
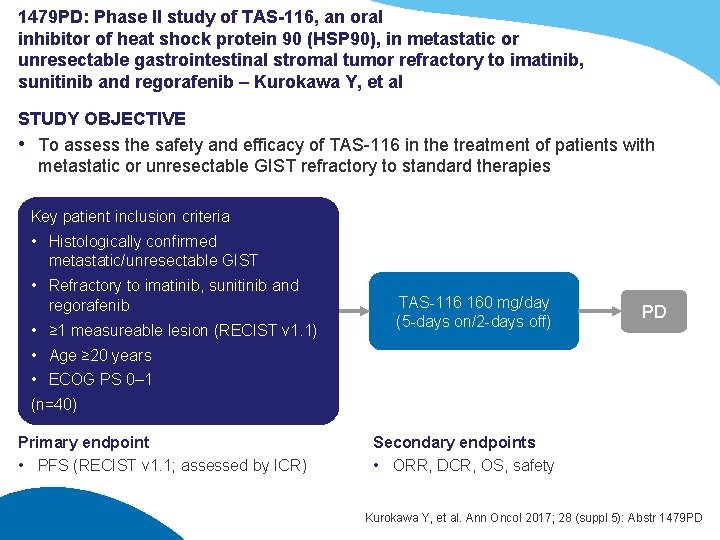
1479 PD: Phase II study of TAS-116, an oral inhibitor of heat shock protein 90 (HSP 90), in metastatic or unresectable gastrointestinal stromal tumor refractory to imatinib, sunitinib and regorafenib – Kurokawa Y, et al STUDY OBJECTIVE • To assess the safety and efficacy of TAS-116 in the treatment of patients with metastatic or unresectable GIST refractory to standard therapies Key patient inclusion criteria • Histologically confirmed metastatic/unresectable GIST • Refractory to imatinib, sunitinib and regorafenib • ≥ 1 measureable lesion (RECIST v 1. 1) TAS-116 160 mg/day (5 -days on/2 -days off) PD • Age ≥ 20 years • ECOG PS 0– 1 (n=40) Primary endpoint • PFS (RECIST v 1. 1; assessed by ICR) Secondary endpoints • ORR, DCR, OS, safety Kurokawa Y, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1479 PD
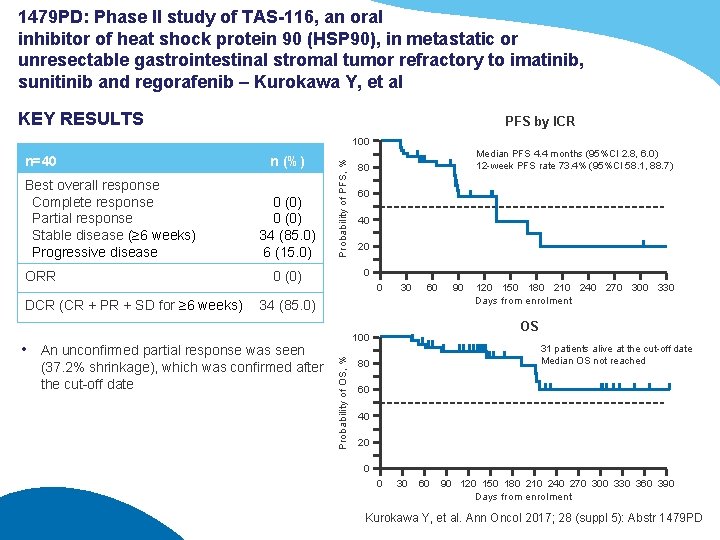
1479 PD: Phase II study of TAS-116, an oral inhibitor of heat shock protein 90 (HSP 90), in metastatic or unresectable gastrointestinal stromal tumor refractory to imatinib, sunitinib and regorafenib – Kurokawa Y, et al KEY RESULTS PFS by ICR Best overall response Complete response Partial response Stable disease (≥ 6 weeks) Progressive disease ORR DCR (CR + PR + SD for ≥ 6 weeks) n (%) 0 (0) 34 (85. 0) 6 (15. 0) Median PFS 4. 4 months (95%CI 2. 8, 6. 0) 12 -week PFS rate 73. 4% (95%CI 58. 1, 88. 7) 80 60 40 20 0 0 (0) 0 34 (85. 0) • An unconfirmed partial response was seen (37. 2% shrinkage), which was confirmed after the cut-off date 30 60 90 120 150 180 210 240 270 300 330 Days from enrolment OS 100 Probability of OS, % n=40 Probability of PFS, % 100 31 patients alive at the cut-off date Median OS not reached 80 60 40 20 0 0 30 60 90 120 150 180 210 240 270 300 330 360 390 Days from enrolment Kurokawa Y, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1479 PD
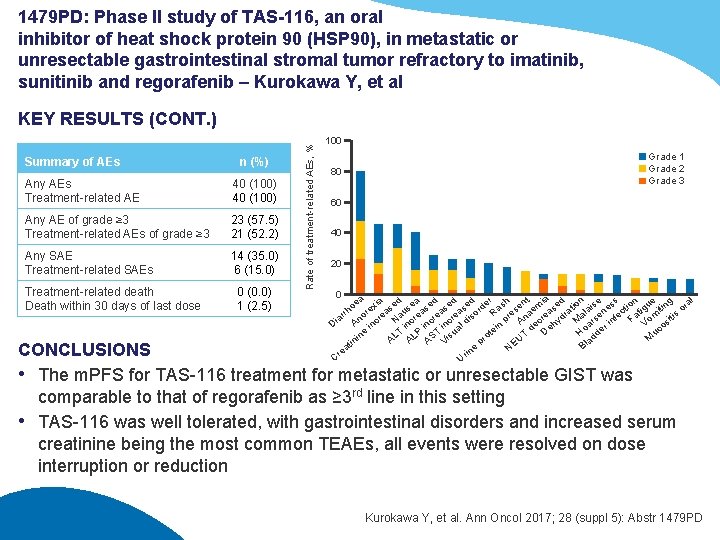
1479 PD: Phase II study of TAS-116, an oral inhibitor of heat shock protein 90 (HSP 90), in metastatic or unresectable gastrointestinal stromal tumor refractory to imatinib, sunitinib and regorafenib – Kurokawa Y, et al Summary of AEs n (%) Any AEs Treatment-related AE 40 (100) Any AE of grade ≥ 3 Treatment-related AEs of grade ≥ 3 23 (57. 5) 21 (52. 2) Any SAE Treatment-related SAEs 14 (35. 0) 6 (15. 0) Treatment-related death Death within 30 days of last dose 0 (0. 0) 1 (2. 5) Rate of treatment-related AEs, % KEY RESULTS (CONT. ) 100 80 Grade 1 Grade 2 Grade 3 60 40 20 0 l t ia d n e s n e g a a d d d r h oe exi se se se rde as sen em se atio lais nes ctio igu itin ora h a a a t r u r o R re na re dr a e fe a m tis ar no cre Na cre cre dis p A c hy M ars in F Vo si A in Di e e o r in in in al in o d e e e t T T u P S is D uc H dd T n o L L i r M U A A A V a p tin Bl e NE ea in r r C U CONCLUSIONS • The m. PFS for TAS-116 treatment for metastatic or unresectable GIST was comparable to that of regorafenib as ≥ 3 rd line in this setting • TAS-116 was well tolerated, with gastrointestinal disorders and increased serum creatinine being the most common TEAEs, all events were resolved on dose interruption or reduction Kurokawa Y, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1479 PD

Locally advanced sarcoma

1474 O: Improved overall and progression free survival after surgery in expert sites for sarcoma patients: a nationwide study of FSG-GETO/NETSARC – Blay J, et al STUDY OBJECTIVE • To investigate the impact of surgery on relapse and survival in a reference centre in the nationwide Net. Sarc/RREPS study METHODS • Net. Sarc is a network of 26 reference sarcoma centres with specialized multidisciplinary tumour boards (MDTB), wherein presentation to the MDTB and second pathological review are now mandatory for patients with sarcoma • Patient characteristics, diagnosis and treatment data, medical decision and disease progression and survival were reviewed using the Net. Sarc network database between January 2010 and December 2014 • Prognostic factors for local relapse-free survival (LRFS), RFS and OS was analysed using univariate and multivariate analysis • 9646 patients with non-metastatic sarcoma (age ≥ 15 years) and a first diagnosis of soft tissue and visceral sarcoma were reviewed Blay J, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1474 O
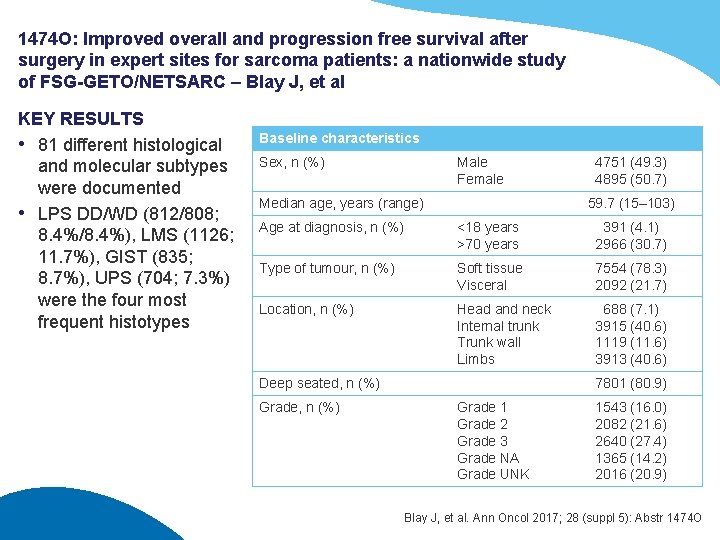
1474 O: Improved overall and progression free survival after surgery in expert sites for sarcoma patients: a nationwide study of FSG-GETO/NETSARC – Blay J, et al KEY RESULTS • 81 different histological and molecular subtypes were documented • LPS DD/WD (812/808; 8. 4%/8. 4%), LMS (1126; 11. 7%), GIST (835; 8. 7%), UPS (704; 7. 3%) were the four most frequent histotypes Baseline characteristics Sex, n (%) Male Female Median age, years (range) 4751 (49. 3) 4895 (50. 7) 59. 7 (15– 103) Age at diagnosis, n (%) <18 years >70 years 391 (4. 1) 2966 (30. 7) Type of tumour, n (%) Soft tissue Visceral 7554 (78. 3) 2092 (21. 7) Location, n (%) Head and neck Internal trunk Trunk wall Limbs 688 (7. 1) 3915 (40. 6) 1119 (11. 6) 3913 (40. 6) Deep seated, n (%) Grade, n (%) 7801 (80. 9) Grade 1 Grade 2 Grade 3 Grade NA Grade UNK 1543 (16. 0) 2082 (21. 6) 2640 (27. 4) 1365 (14. 2) 2016 (20. 9) Blay J, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1474 O

1474 O: Improved overall and progression free survival after surgery in expert sites for sarcoma patients: a nationwide study of FSG-GETO/NETSARC – Blay J, et al KEY RESULTS • During the study period, the proportion of patients undergoing primary surgery within the Net. Sarc network was 37. 1% (range 34. 5– 40. 0; no trend for change) Compliance with CPGS Surgery within the Net. Sarc network Surgery outside the Net. Sarc network p-value MDTB before surgery, % 56. 2 10. 4 <0. 0001 Adequate tumour imaging prior to treatment/surgery, % 84. 7 57. 8 <0. 0001 Biopsy prior to first resection , % 77. 9 32. 5 <0. 0001 • At clinical presentation, patients were of a younger age, more commonly male and less likely to have visceral sarcomas Tumour characteristic p-value Larger tumours: median size 104 vs. 88 mm <0. 0001 More deep seated: 83. 8 vs. 75. 9% <0. 0001 Higher grade: G 2/3 51. 1 vs. 47. 7 <0. 0001 Blay J, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1474 O
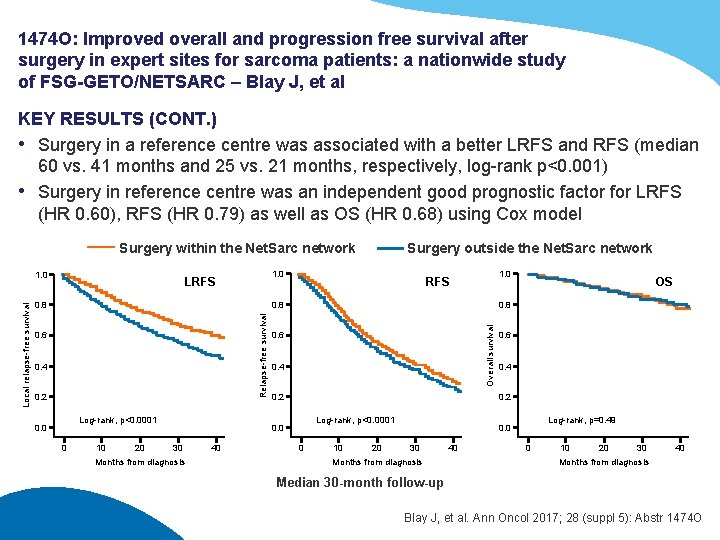
1474 O: Improved overall and progression free survival after surgery in expert sites for sarcoma patients: a nationwide study of FSG-GETO/NETSARC – Blay J, et al KEY RESULTS (CONT. ) • Surgery in a reference centre was associated with a better LRFS and RFS (median 60 vs. 41 months and 25 vs. 21 months, respectively, log-rank p<0. 001) • Surgery in reference centre was an independent good prognostic factor for LRFS (HR 0. 60), RFS (HR 0. 79) as well as OS (HR 0. 68) using Cox model Surgery within the Net. Sarc network 1. 0 LRFS 0. 8 1. 0 RFS 0. 6 0. 4 0. 2 Log-rank, p<0. 0001 0. 0 0 10 20 0. 6 0. 4 0. 2 Months from diagnosis 40 0. 6 0. 4 0. 2 Log-rank, p<0. 0001 0. 0 30 OS 0. 8 Overall survival 0. 8 Relapse-free survival Local relapse-free survival 1. 0 Surgery outside the Net. Sarc network 0 10 20 Log-rank, p=0. 49 0. 0 30 Months from diagnosis 40 0 10 20 30 40 Months from diagnosis Median 30 -month follow-up Blay J, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1474 O
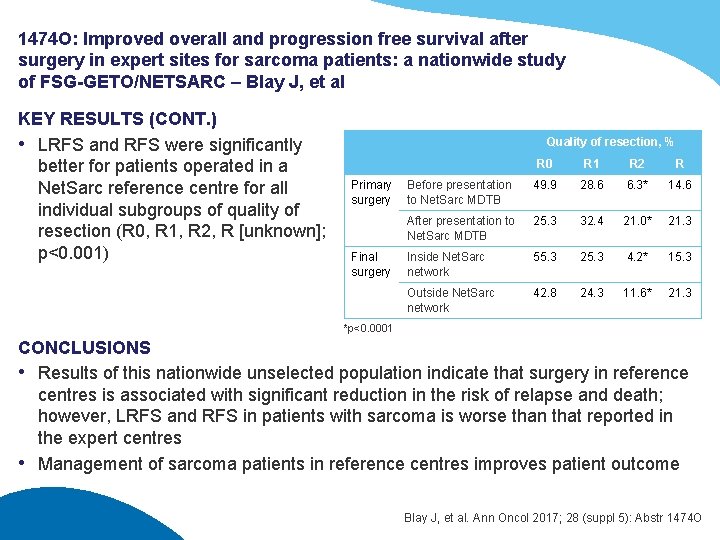
1474 O: Improved overall and progression free survival after surgery in expert sites for sarcoma patients: a nationwide study of FSG-GETO/NETSARC – Blay J, et al KEY RESULTS (CONT. ) • LRFS and RFS were significantly better for patients operated in a Net. Sarc reference centre for all individual subgroups of quality of resection (R 0, R 1, R 2, R [unknown]; p<0. 001) Quality of resection, % Primary surgery Final surgery R 0 R 1 R 2 R Before presentation to Net. Sarc MDTB 49. 9 28. 6 6. 3* 14. 6 After presentation to Net. Sarc MDTB 25. 3 32. 4 21. 0* 21. 3 Inside Net. Sarc network 55. 3 25. 3 4. 2* 15. 3 Outside Net. Sarc network 42. 8 24. 3 11. 6* 21. 3 *p<0. 0001 CONCLUSIONS • Results of this nationwide unselected population indicate that surgery in reference centres is associated with significant reduction in the risk of relapse and death; however, LRFS and RFS in patients with sarcoma is worse than that reported in the expert centres • Management of sarcoma patients in reference centres improves patient outcome Blay J, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1474 O
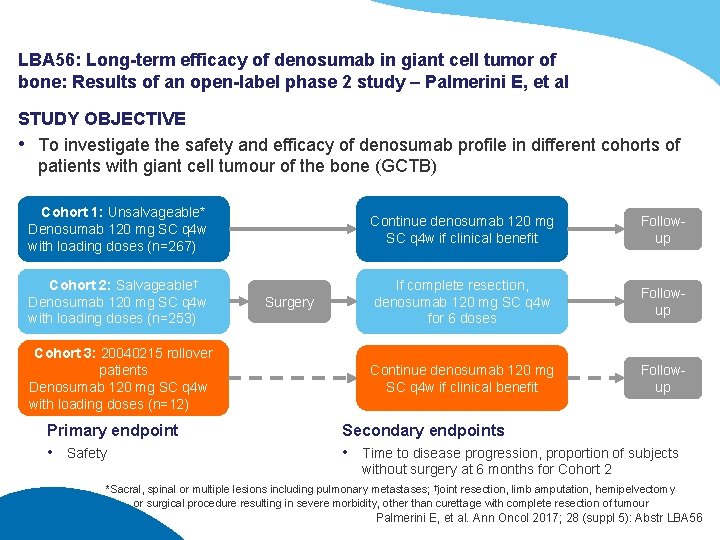
LBA 56: Long-term efficacy of denosumab in giant cell tumor of bone: Results of an open-label phase 2 study – Palmerini E, et al STUDY OBJECTIVE • To investigate the safety and efficacy of denosumab profile in different cohorts of patients with giant cell tumour of the bone (GCTB) Cohort 1: Unsalvageable* Denosumab 120 mg SC q 4 w with loading doses (n=267) Continue denosumab 120 mg SC q 4 w if clinical benefit Followup Cohort 2: Salvageable† Denosumab 120 mg SC q 4 w with loading doses (n=253) If complete resection, denosumab 120 mg SC q 4 w for 6 doses Followup Continue denosumab 120 mg SC q 4 w if clinical benefit Followup Cohort 3: 20040215 rollover patients Denosumab 120 mg SC q 4 w with loading doses (n=12) Primary endpoint • Safety Surgery Secondary endpoints • Time to disease progression, proportion of subjects without surgery at 6 months for Cohort 2 *Sacral, spinal or multiple lesions including pulmonary metastases; †joint resection, limb amputation, hemipelvectomy or surgical procedure resulting in severe morbidity, other than curettage with complete resection of tumour Palmerini E, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 56
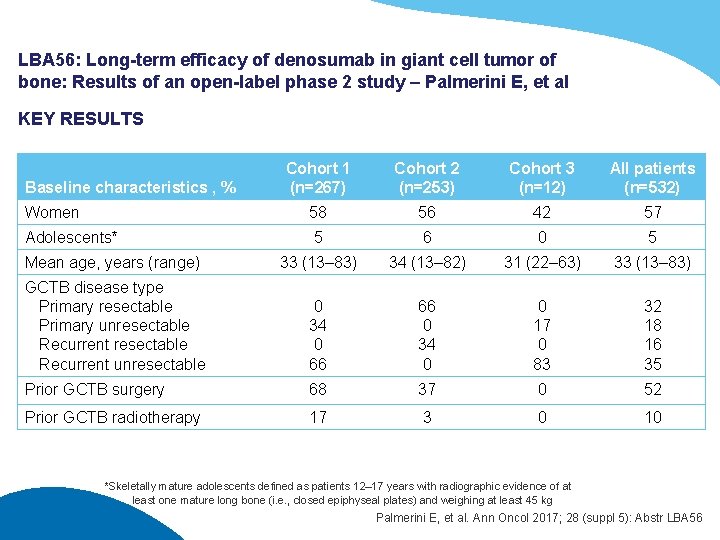
LBA 56: Long-term efficacy of denosumab in giant cell tumor of bone: Results of an open-label phase 2 study – Palmerini E, et al KEY RESULTS Cohort 1 (n=267) Cohort 2 (n=253) Cohort 3 (n=12) All patients (n=532) Women 58 56 42 57 Adolescents* 5 6 0 5 Mean age, years (range) 33 (13– 83) 34 (13– 82) 31 (22– 63) 33 (13– 83) GCTB disease type Primary resectable Primary unresectable Recurrent unresectable 0 34 0 66 66 0 34 0 0 17 0 83 32 18 16 35 Prior GCTB surgery 68 37 0 52 Prior GCTB radiotherapy 17 3 0 10 Baseline characteristics , % *Skeletally mature adolescents defined as patients 12– 17 years with radiographic evidence of at least one mature long bone (i. e. , closed epiphyseal plates) and weighing at least 45 kg Palmerini E, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 56
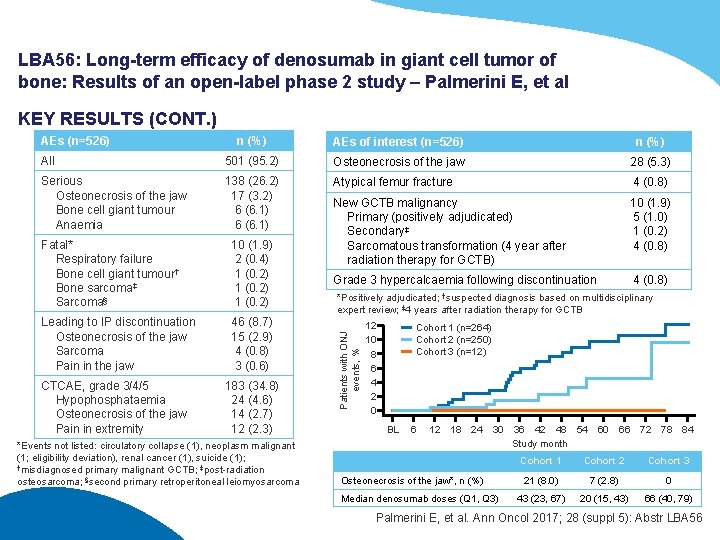
LBA 56: Long-term efficacy of denosumab in giant cell tumor of bone: Results of an open-label phase 2 study – Palmerini E, et al KEY RESULTS (CONT. ) n (%) AEs of interest (n=526) n (%) All 501 (95. 2) Osteonecrosis of the jaw 28 (5. 3) Serious Osteonecrosis of the jaw Bone cell giant tumour Anaemia 138 (26. 2) 17 (3. 2) 6 (6. 1) Atypical femur fracture 4 (0. 8) New GCTB malignancy Primary (positively adjudicated) Secondary‡ Sarcomatous transformation (4 year after radiation therapy for GCTB) 10 (1. 9) 5 (1. 0) 1 (0. 2) 4 (0. 8) Grade 3 hypercalcaemia following discontinuation 4 (0. 8) Fatal* Respiratory failure Bone cell giant tumour† Bone sarcoma‡ Sarcoma§ 10 (1. 9) 2 (0. 4) 1 (0. 2) Leading to IP discontinuation Osteonecrosis of the jaw Sarcoma Pain in the jaw 46 (8. 7) 15 (2. 9) 4 (0. 8) 3 (0. 6) CTCAE, grade 3/4/5 Hypophosphataemia Osteonecrosis of the jaw Pain in extremity 183 (34. 8) 24 (4. 6) 14 (2. 7) 12 (2. 3) *Events not listed: circulatory collapse (1), neoplasm malignant (1; eligibility deviation), renal cancer (1), suicide (1); †misdiagnosed primary malignant GCTB; ‡post-radiation osteosarcoma; §second primary retroperitoneal leiomyosarcoma †suspected *Positively adjudicated; diagnosis based on multidisciplinary expert review; ‡ 4 years after radiation therapy for GCTB Patients with ONJ events, % AEs (n=526) 12 10 8 6 4 2 0 Cohort 1 (n=264) Cohort 2 (n=250) Cohort 3 (n=12) BL 6 12 18 24 30 36 42 48 54 60 66 72 78 84 Study month Osteonecrosis of the jaw*, n (%) Median denosumab doses (Q 1, Q 3) Cohort 1 Cohort 2 Cohort 3 21 (8. 0) 7 (2. 8) 0 43 (23, 67) 20 (15, 43) 66 (40, 79) Palmerini E, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 56
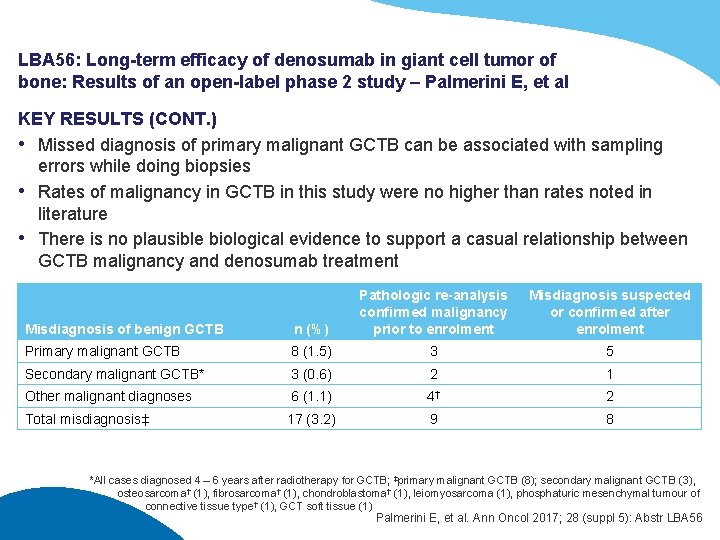
LBA 56: Long-term efficacy of denosumab in giant cell tumor of bone: Results of an open-label phase 2 study – Palmerini E, et al KEY RESULTS (CONT. ) • Missed diagnosis of primary malignant GCTB can be associated with sampling errors while doing biopsies • Rates of malignancy in GCTB in this study were no higher than rates noted in literature • There is no plausible biological evidence to support a casual relationship between GCTB malignancy and denosumab treatment Misdiagnosis of benign GCTB n (%) Pathologic re-analysis confirmed malignancy prior to enrolment Misdiagnosis suspected or confirmed after enrolment Primary malignant GCTB 8 (1. 5) 3 5 Secondary malignant GCTB* 3 (0. 6) 2 1 Other malignant diagnoses 6 (1. 1) 4† 2 Total misdiagnosis‡ 17 (3. 2) 9 8 *All cases diagnosed 4 – 6 years after radiotherapy for GCTB; ‡primary malignant GCTB (8); secondary malignant GCTB (3), osteosarcoma† (1), fibrosarcoma† (1), chondroblastoma† (1), leiomyosarcoma (1), phosphaturic mesenchymal tumour of connective tissue type† (1), GCT soft tissue (1) Palmerini E, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 56
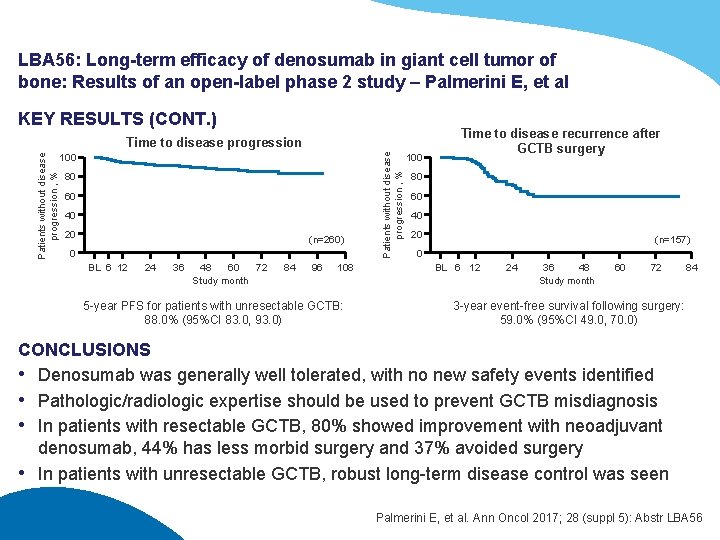
LBA 56: Long-term efficacy of denosumab in giant cell tumor of bone: Results of an open-label phase 2 study – Palmerini E, et al KEY RESULTS (CONT. ) 100 80 60 40 20 (n=260) 0 BL 6 12 24 36 48 60 72 Study month 84 96 108 5 -year PFS for patients with unresectable GCTB: 88. 0% (95%CI 83. 0, 93. 0) Patients without disease progression , % Time to disease progression 100 Time to disease recurrence after GCTB surgery 80 60 40 20 (n=157) 0 BL 6 12 24 36 48 Study month 60 72 84 3 -year event-free survival following surgery: 59. 0% (95%CI 49. 0, 70. 0) CONCLUSIONS • Denosumab was generally well tolerated, with no new safety events identified • Pathologic/radiologic expertise should be used to prevent GCTB misdiagnosis • In patients with resectable GCTB, 80% showed improvement with neoadjuvant denosumab, 44% has less morbid surgery and 37% avoided surgery • In patients with unresectable GCTB, robust long-term disease control was seen Palmerini E, et al. Ann Oncol 2017; 28 (suppl 5): Abstr LBA 56
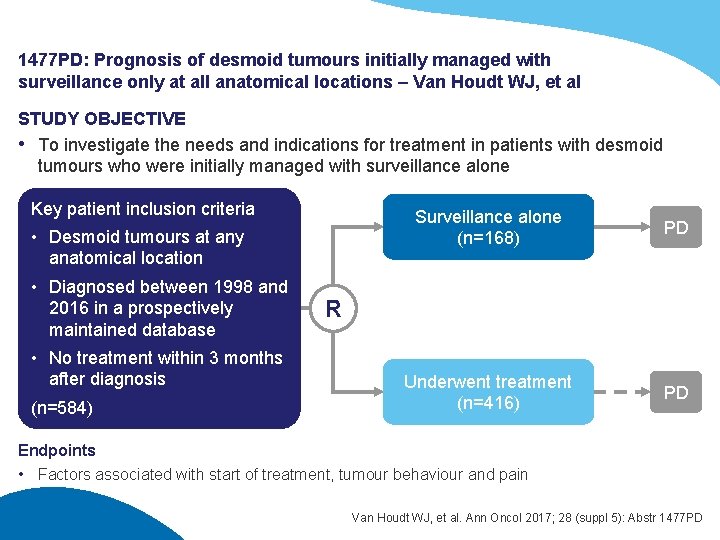
1477 PD: Prognosis of desmoid tumours initially managed with surveillance only at all anatomical locations – Van Houdt WJ, et al STUDY OBJECTIVE • To investigate the needs and indications for treatment in patients with desmoid tumours who were initially managed with surveillance alone Key patient inclusion criteria • Desmoid tumours at any anatomical location • Diagnosed between 1998 and 2016 in a prospectively maintained database • No treatment within 3 months after diagnosis (n=584) Surveillance alone (n=168) PD Underwent treatment (n=416) PD R Endpoints • Factors associated with start of treatment, tumour behaviour and pain Van Houdt WJ, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1477 PD
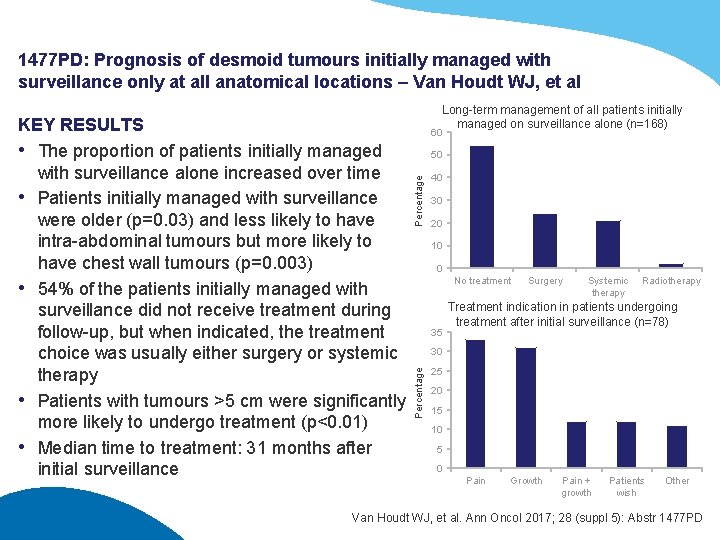
1477 PD: Prognosis of desmoid tumours initially managed with surveillance only at all anatomical locations – Van Houdt WJ, et al 60 Percentage 50 40 30 20 10 0 No treatment 35 Surgery Systemic therapy Radiotherapy Treatment indication in patients undergoing treatment after initial surveillance (n=78) 30 Percentage KEY RESULTS • The proportion of patients initially managed with surveillance alone increased over time • Patients initially managed with surveillance were older (p=0. 03) and less likely to have intra-abdominal tumours but more likely to have chest wall tumours (p=0. 003) • 54% of the patients initially managed with surveillance did not receive treatment during follow-up, but when indicated, the treatment choice was usually either surgery or systemic therapy • Patients with tumours >5 cm were significantly more likely to undergo treatment (p<0. 01) • Median time to treatment: 31 months after initial surveillance Long-term management of all patients initially managed on surveillance alone (n=168) 25 20 15 10 5 0 Pain Growth Pain + growth Patients wish Other Van Houdt WJ, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1477 PD
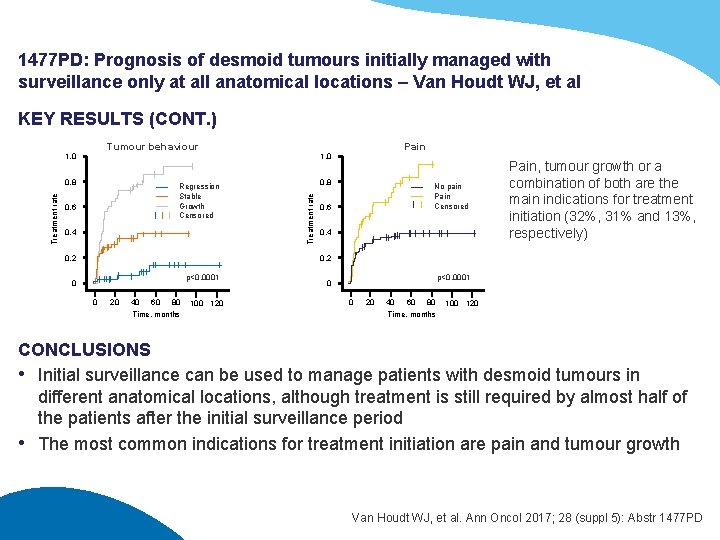
1477 PD: Prognosis of desmoid tumours initially managed with surveillance only at all anatomical locations – Van Houdt WJ, et al KEY RESULTS (CONT. ) Tumour behaviour Treatment rate 0. 8 Regression Stable Growth Censored 0. 6 0. 4 0. 2 Pain 1. 0 0. 8 Treatment rate 1. 0 No pain Pain Censored 0. 6 0. 4 Pain, tumour growth or a combination of both are the main indications for treatment initiation (32%, 31% and 13%, respectively) 0. 2 p<0. 0001 0 0 20 40 60 80 Time, months 100 120 CONCLUSIONS • Initial surveillance can be used to manage patients with desmoid tumours in different anatomical locations, although treatment is still required by almost half of the patients after the initial surveillance period • The most common indications for treatment initiation are pain and tumour growth Van Houdt WJ, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1477 PD

1478 PD: Adult Translocation-related soft tissue sarcomas (TRS): Presentation, management and outcome of 2, 143 cases confirmed by expert pathologists – Penal N, et al STUDY OBJECTIVE • To evaluate the pattern of translocation-related STS compared with other sarcomas METHODS • This was a retrospective multicentre study of 12, 262 patients treated in 22 French Referral Sarcoma Centres and enrolled in the ‘Conticabase’ between January 1980 and December 2013 • Expert pathologists systematically reviewed the diagnoses and classified them according to the 2013 WHO Classification • All cases of GIST were excluded from these analyses Penal N, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1478 PD
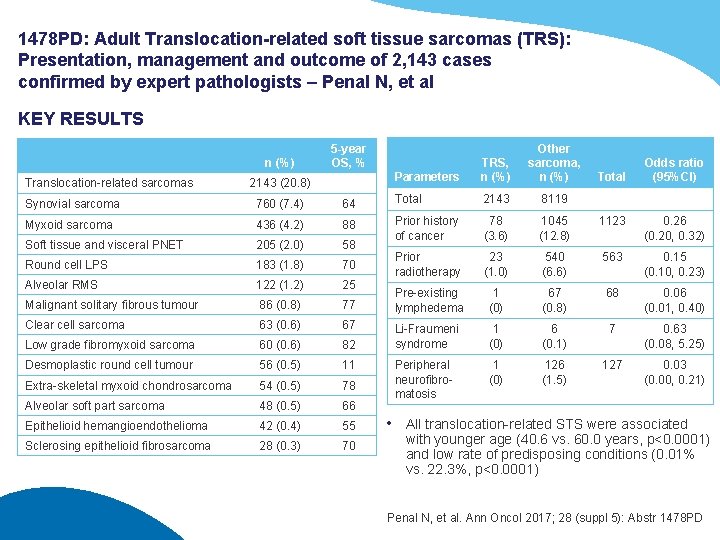
1478 PD: Adult Translocation-related soft tissue sarcomas (TRS): Presentation, management and outcome of 2, 143 cases confirmed by expert pathologists – Penal N, et al KEY RESULTS n (%) Translocation-related sarcomas Parameters TRS, n (%) Other sarcoma, n (%) 5 -year OS, % 2143 (20. 8) Total Odds ratio (95%CI) Synovial sarcoma 760 (7. 4) 64 Total 2143 8119 Myxoid sarcoma 436 (4. 2) 88 205 (2. 0) 58 78 (3. 6) 1045 (12. 8) 1123 Soft tissue and visceral PNET Prior history of cancer 0. 26 (0. 20, 0. 32) Round cell LPS 183 (1. 8) 70 122 (1. 2) 25 23 (1. 0) 540 (6. 6) 563 Alveolar RMS Prior radiotherapy 0. 15 (0. 10, 0. 23) Malignant solitary fibrous tumour 86 (0. 8) 77 Pre-existing lymphedema 1 (0) 67 (0. 8) 68 0. 06 (0. 01, 0. 40) Clear cell sarcoma 63 (0. 6) 67 60 (0. 6) 82 1 (0) 6 (0. 1) 7 Low grade fibromyxoid sarcoma Li-Fraumeni syndrome 0. 63 (0. 08, 5. 25) Desmoplastic round cell tumour 56 (0. 5) 11 54 (0. 5) 78 1 (0) 126 (1. 5) 127 Extra-skeletal myxoid chondrosarcoma 0. 03 (0. 00, 0. 21) Alveolar soft part sarcoma 48 (0. 5) 66 Peripheral neurofibromatosis Epithelioid hemangioendothelioma 42 (0. 4) 55 Sclerosing epithelioid fibrosarcoma 28 (0. 3) 70 • All translocation-related STS were associated with younger age (40. 6 vs. 60. 0 years, p<0. 0001) and low rate of predisposing conditions (0. 01% vs. 22. 3%, p<0. 0001) Penal N, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1478 PD
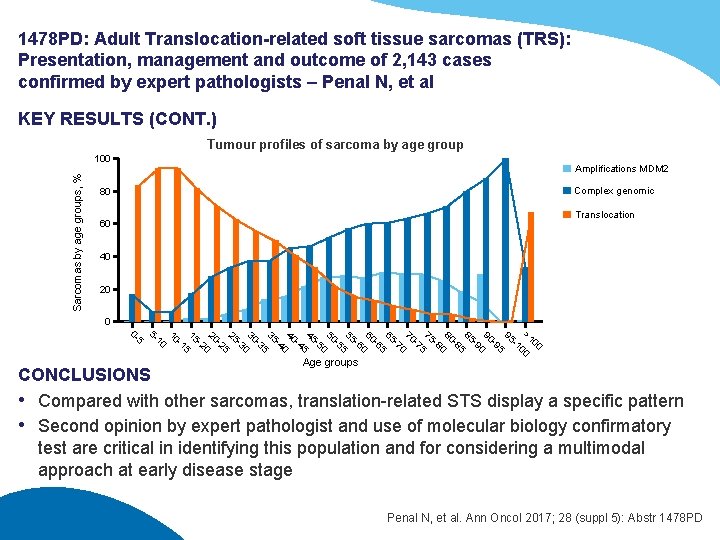
1478 PD: Adult Translocation-related soft tissue sarcomas (TRS): Presentation, management and outcome of 2, 143 cases confirmed by expert pathologists – Penal N, et al KEY RESULTS (CONT. ) Tumour profiles of sarcoma by age group Sarcomas by age groups, % 100 Amplifications MDM 2 Complex genomic 80 Translocation 60 40 20 0 0 - 5 5 - 6 8 2 4 5 5 1 1 2 3 3 4 6 7 8 9 7 9 > 10 0 -1 5 -2 0 -2 5 -3 0 -3 5 -4 0 -4 5 -5 0 -5 5 -6 0 -6 5 -7 0 -7 5 -8 0 -8 5 -9 0 -9 5 -1 10 0 0 5 5 5 0 0 5 5 0 00 0 Age groups CONCLUSIONS • Compared with other sarcomas, translation-related STS display a specific pattern • Second opinion by expert pathologist and use of molecular biology confirmatory test are critical in identifying this population and for considering a multimodal approach at early disease stage Penal N, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1478 PD

Advanced/metastatic sarcoma: chemotherapy, targeted therapy and immunotherapy
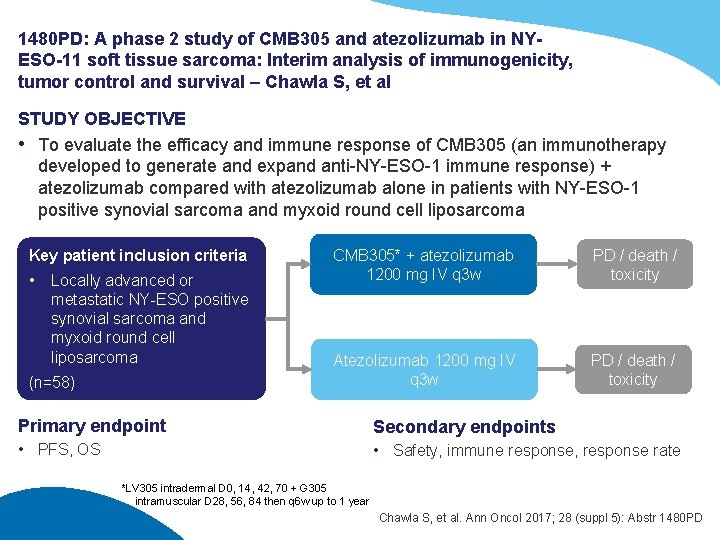
1480 PD: A phase 2 study of CMB 305 and atezolizumab in NYESO-11 soft tissue sarcoma: Interim analysis of immunogenicity, tumor control and survival – Chawla S, et al STUDY OBJECTIVE • To evaluate the efficacy and immune response of CMB 305 (an immunotherapy developed to generate and expand anti-NY-ESO-1 immune response) + atezolizumab compared with atezolizumab alone in patients with NY-ESO-1 positive synovial sarcoma and myxoid round cell liposarcoma Key patient inclusion criteria • Locally advanced or metastatic NY-ESO positive synovial sarcoma and myxoid round cell liposarcoma (n=58) CMB 305* + atezolizumab 1200 mg IV q 3 w PD / death / toxicity Atezolizumab 1200 mg IV q 3 w PD / death / toxicity Primary endpoint • PFS, OS Secondary endpoints • Safety, immune response, response rate *LV 305 intradermal D 0, 14, 42, 70 + G 305 intramuscular D 28, 56, 84 then q 6 w up to 1 year Chawla S, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1480 PD
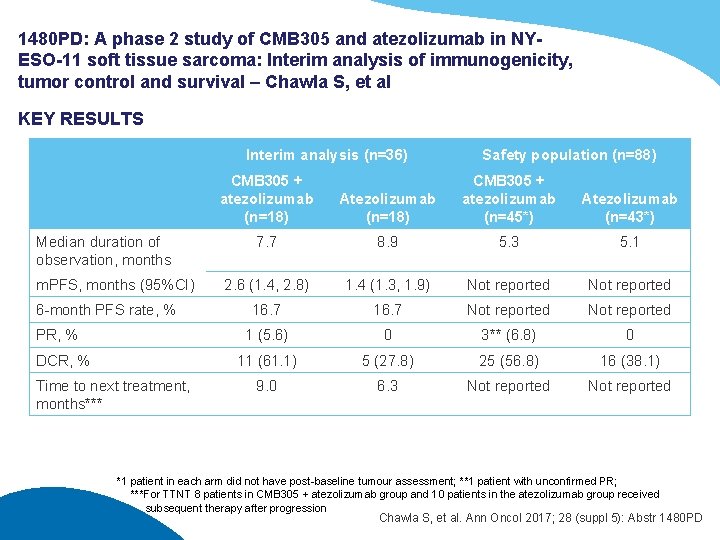
1480 PD: A phase 2 study of CMB 305 and atezolizumab in NYESO-11 soft tissue sarcoma: Interim analysis of immunogenicity, tumor control and survival – Chawla S, et al KEY RESULTS Interim analysis (n=36) Median duration of observation, months m. PFS, months (95%CI) 6 -month PFS rate, % PR, % DCR, % Time to next treatment, months*** CMB 305 + atezolizumab (n=18) Safety population (n=88) Atezolizumab (n=18) CMB 305 + atezolizumab (n=45*) Atezolizumab (n=43*) 7. 7 8. 9 5. 3 5. 1 2. 6 (1. 4, 2. 8) 1. 4 (1. 3, 1. 9) Not reported 16. 7 Not reported 1 (5. 6) 0 3** (6. 8) 0 11 (61. 1) 5 (27. 8) 25 (56. 8) 16 (38. 1) 9. 0 6. 3 Not reported *1 patient in each arm did not have post-baseline tumour assessment; **1 patient with unconfirmed PR; ***For TTNT 8 patients in CMB 305 + atezolizumab group and 10 patients in the atezolizumab group received subsequent therapy after progression Chawla S, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1480 PD

1480 PD: A phase 2 study of CMB 305 and atezolizumab in NYESO-11 soft tissue sarcoma: Interim analysis of immunogenicity, tumor control and survival – Chawla S, et al KEY RESULTS (CONT. ) Patients with at least 1 treatmentrelated treatment-emergent AEs, n (%) Safety population (n=88) CMB 305 + atezolizumab (n=45) Atezolizumab (n=43) Grade 1 18 (40) 13 (30. 2) Grade 2 10 (22) 5 (11. 6) Grade 3 3 (6. 7) 3 (7. 0) Grade 4 1 (2. 2) 1 (2. 3) Grade 5 0 0 CONCLUSIONS • Interim results suggest that CMB 305 + atezolizumab may result in improved clinical efficacy over atezolizumab alone in patients with NY-ESO-1+ STS with low/absent PD-L 1 • CMB 305 + atezolizumab is well tolerated; no new safety signals were detected Chawla S, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1480 PD

1483 PD: Imatinib in combination with everolimus in patients with progressive advanced chordoma: results form an Italian phase 2 clinical trial – Stacchiotti S, et al STUDY OBJECTIVE • To evaluate the anti-tumour activity of imatinib + everolimus in patients with advanced PDGFB and/or PDGFRB+ chordomas with evidence of m. TOR and/or of its effectors (S 6, 4 EBP 1) activation Key patient inclusion criteria • Advanced PDGFB and/or PDGFRB and m. TOR/S 6/4 EBP 1+ chordoma • Disease progression 6 months prior to study entry Imatinib 400 mg/day + everolimus 2. 5 mg/day PD / toxicity (n=43) Primary endpoint • ORR Secondary endpoints • RECIST response, PFS, OS Stacchiotti S, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1483 PD
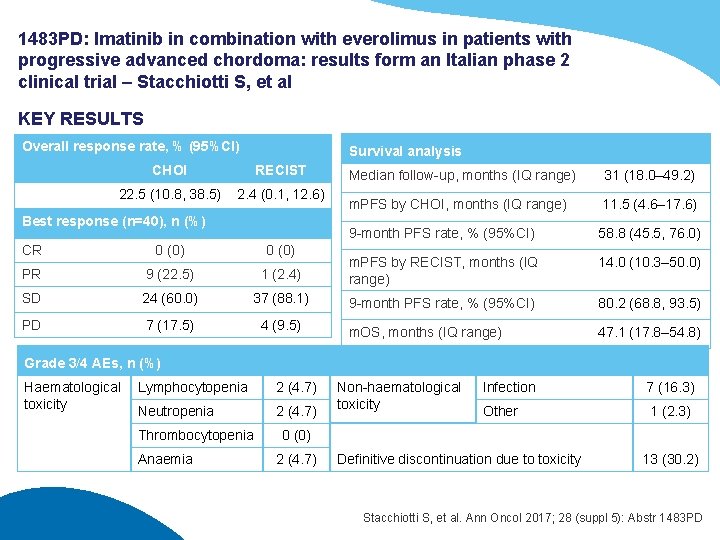
1483 PD: Imatinib in combination with everolimus in patients with progressive advanced chordoma: results form an Italian phase 2 clinical trial – Stacchiotti S, et al KEY RESULTS Overall response rate, % (95%CI) Survival analysis CHOI RECIST 22. 5 (10. 8, 38. 5) 2. 4 (0. 1, 12. 6) Best response (n=40), n (%) Median follow-up, months (IQ range) 31 (18. 0– 49. 2) m. PFS by CHOI, months (IQ range) 11. 5 (4. 6– 17. 6) 9 -month PFS rate, % (95%CI) 58. 8 (45. 5, 76. 0) CR 0 (0) PR 9 (22. 5) 1 (2. 4) m. PFS by RECIST, months (IQ range) 14. 0 (10. 3– 50. 0) SD 24 (60. 0) 37 (88. 1) 9 -month PFS rate, % (95%CI) 80. 2 (68. 8, 93. 5) PD 7 (17. 5) 4 (9. 5) m. OS, months (IQ range) 47. 1 (17. 8– 54. 8) Grade 3/4 AEs, n (%) Haematological toxicity Lymphocytopenia 2 (4. 7) Neutropenia 2 (4. 7) Thrombocytopenia Anaemia Non-haematological toxicity Infection 7 (16. 3) Other 1 (2. 3) 0 (0) 2 (4. 7) Definitive discontinuation due to toxicity 13 (30. 2) Stacchiotti S, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1483 PD
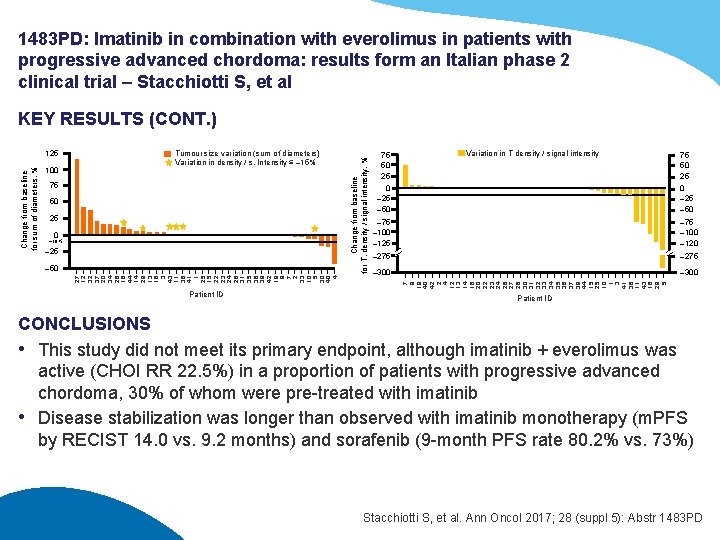
1483 PD: Imatinib in combination with everolimus in patients with progressive advanced chordoma: results form an Italian phase 2 clinical trial – Stacchiotti S, et al 100 Tumour size variation (sum of diameters) Variation in density / s. Intensity ≤ – 15% 75 50 25 0 – 10% – 25 27 12 32 37 20 34 28 16 44 14 29 13 18 3 43 11 38 41 1 25 15 22 23 24 26 31 35 36 39 42 19 9 7 2 33 10 5 30 40 4 – 50 Patient ID 75 50 25 0 – 25 – 50 Variation in T density / signal intensity 75 50 25 0 – 25 – 50 – 75 – 100 – 125 – 75 – 100 – 120 – 275 – 300 7 9 19 40 42 2 4 12 13 14 18 20 22 23 24 26 27 28 30 31 32 33 34 35 36 37 39 44 15 25 10 1 3 41 38 11 43 16 29 5 Change from baseline for sum of diameters, % 125 Change from baseline for T. density / signal intensity, % KEY RESULTS (CONT. ) Patient ID CONCLUSIONS • This study did not meet its primary endpoint, although imatinib + everolimus was active (CHOI RR 22. 5%) in a proportion of patients with progressive advanced chordoma, 30% of whom were pre-treated with imatinib • Disease stabilization was longer than observed with imatinib monotherapy (m. PFS by RECIST 14. 0 vs. 9. 2 months) and sorafenib (9 -month PFS rate 80. 2% vs. 73%) Stacchiotti S, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1483 PD

1484 PD: A matching-adjusted indirect comparison of trabectedin and pazopanib for the treatment of advanced, metastatic, leiomyosarcomas – Jones RL, et al STUDY OBJECTIVE • To explore the clinically relevant differences in efficacy and safety between trabectedin and pazopanib in the treatment of advanced metastatic, leiomyosarcomas after failure of anthracycline or ifosfamide-based regimens METHODS • MAIC (matching-adjusted indirect comparison) was performed using baseline characteristics from two phase 3 trials: SAR 3007 for trabectedin and PALETTE for pazopanib • Patients receiving trabectedin who did not meet the inclusion criteria for PALETTE were excluded, generating a sample size of 372 patients (trabectedin, n=263; pazopanib, n=109) • Using a generalized method of moments (GMM), cohorts were matched optimally to evaluate differences in OS, PFS and safety Jones RL, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1484 PD
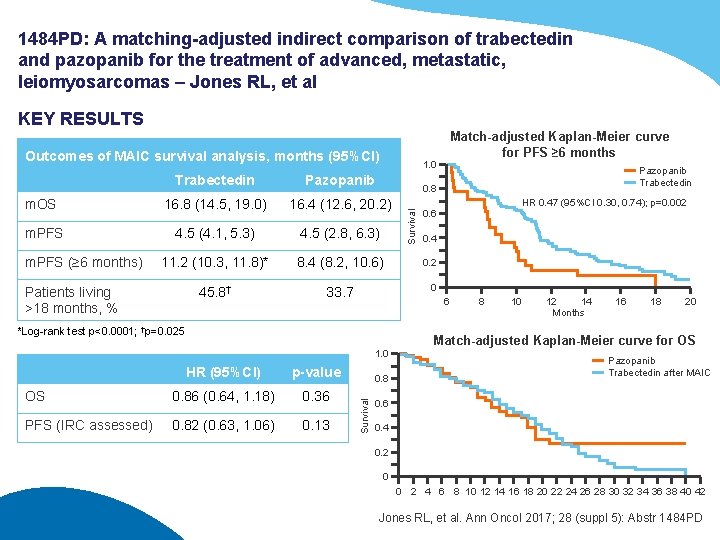
1484 PD: A matching-adjusted indirect comparison of trabectedin and pazopanib for the treatment of advanced, metastatic, leiomyosarcomas – Jones RL, et al KEY RESULTS Outcomes of MAIC survival analysis, months (95%CI) 1. 0 Trabectedin Pazopanib m. OS 16. 8 (14. 5, 19. 0) 16. 4 (12. 6, 20. 2) m. PFS 4. 5 (4. 1, 5. 3) 4. 5 (2. 8, 6. 3) 11. 2 (10. 3, 11. 8)* 8. 4 (8. 2, 10. 6) 45. 8† 33. 7 m. PFS (≥ 6 months) Patients living >18 months, % Match-adjusted Kaplan-Meier curve for PFS ≥ 6 months Pazopanib Trabectedin Survival 0. 8 HR 0. 47 (95%CI 0. 30, 0. 74); p=0. 002 0. 6 0. 4 0. 2 0 6 *Log-rank test p<0. 0001; †p=0. 025 8 10 12 14 Months 16 18 20 Match-adjusted Kaplan-Meier curve for OS 1. 0 p-value 0. 8 OS 0. 86 (0. 64, 1. 18) 0. 36 0. 6 PFS (IRC assessed) 0. 82 (0. 63, 1. 06) 0. 13 Survival HR (95%CI) Pazopanib Trabectedin after MAIC 0. 4 0. 2 0 0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30 32 34 36 38 40 42 Jones RL, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1484 PD

1484 PD: A matching-adjusted indirect comparison of trabectedin and pazopanib for the treatment of advanced, metastatic, leiomyosarcomas – Jones RL, et al KEY RESULTS (CONT. ) • A higher proportion of patients receiving trabectedin than pazopanib underwent subsequent therapies (74. 5% vs. 59%, respectively; p=NS) • AEs that were more common with trabectedin included increased myelosuppression and transient liver enzyme elevation, while those for pazopanib included diarrhoea, hypertension, pulmonary toxicity, pneumothorax and neurotoxicity CONCLUSIONS • Compared with pazopanib, trabectedin significantly increased m. PFS (difference 2. 8 months) in patients who achieved long-term disease control (PFS ≥ 6 months) • The proportion of patients achieving OS >18 months (median relative increment 35. 9%) was also significantly increased with trabectedin • There are differences between the safety profiles of trabectedin and pazopanib Jones RL, et al. Ann Oncol 2017; 28 (suppl 5): Abstr 1484 PD
- Slides: 43



































































